Friendly hello and a bit about stress & adult hippocampal neurogenesis
Posted by cathlau, on 27 June 2015
This is my first post for the Node, so I thought I would introduce myself a little bit…
I just finished my MSc in Experimental Psychology (Behavioural Neuroscience) and now I am striving towards becoming a science communicator. Although, I would like to share the research that I am interested in and was involved in, so here’s a glimpse:
I have an interest in the mechanisms underlying stress regulation, especially adult hippocampal neurogenesis. There is plenty of work published on the detrimental effects of stress on neurogenesis (See Review – Schoenfeld & Gould, 2012) in addition, at least one functional purpose for neurogenesis is stress regulation (Snyder et al., 2011). Chronic stress almost always suppresses neurogenesis (there are exceptions – see Hanson et al., 2011). However, acute stress is interesting in that there is conflicting evidence on its effects on neurogenesis. These differences may ultimately lie in the type of stressor used, although different results have manifested from the same protocol (Tanapat et al., 2001; Thomas et al., 2006).
Let’s take a step outside of the cellular/neuroscience world and into the more behavioural realm for just a moment:
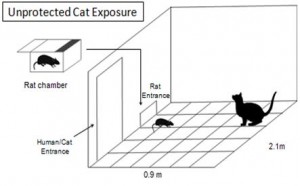
I used an animal model for post traumatic stress disorder (PTSD) that was created by the late Bob Adamec which produces prolonged anxiety-like behaviour following the stressor. This model was unique in that very few labs in the world expose their rat subjects to an unprotected cat exposure (Adamec & Shallow, 1993)!
Image created by Catherine Lau
Much of the chronic stress research and neurogenesis work applies to stress and mood disorders. I wondered if there would be any effect on neurogenesis following a stressor that produces long lasting anxiety-like behaviour. In addition, an interesting review by Kheirbek et al., 2012 shows a potential link between PTSD and neurogenesis through pattern separation (differentiating highly similar contexts). In short, the review talks about how PTSD patients may have less newly born neurons available to differentiate similar episodes and therefore generalize, contributing to symptoms including the re-experience of the traumatic event in their mind. My results revealed that despite an evident stress response (as measured by corticosterone levels), neurogenesis was not different in the hippocampus between controls and predator stressed (Lau et al., manuscript in progress).
What’s happening? Here are some thoughts:
One immediate thought is adaptation, as a certain amount of stress is necessary to cope in our environments. But, how does this manifest in the brain? Kirby et al., 2013 reported an interesting result following acute stress (immobilization and foot shock). Instead of finding a decrease in neurogenesis, an increase in hippocampal neurogenesis was reported in the dorsal hippocampus due to increased secretion of astrocytic FGF2 (necessary proliferative factor for neural progenitor cells). This evidence demonstrates that acute stress may define a life-saving adaptation compared to long term pathology, by stimulating plasticity through increasing neurogenesis. Thus, new neurons in the hippocampus are perhaps needed during acute stress.
A difficult aspect to overcome in any experiment and also in mental health disorders is individual differences. The severity of the disorder may be different as well as how individuals respond to treatment. Some labs are tackling this problem by screening subjects before hand to group a vulnerable and resilient group. Rats displaying extreme behaviour in supposedly exploratory environments, such as the elevated plus maze (no time spent in open arms vs. closed arms) would be classified as the vulnerable group (Cohen et al., 2006).
In addition, there has also been interesting work done by Michael Meaney at McGill University attempting to look at the epigenetics behind depression and stress disorders. Individual differences were characterized by using the frequency of mother and pup interaction (licking/grooming behaviour). Offspring who had mothers giving a high amount of licking/grooming showed a reduced corticosterone response, as well as decreased startle and increased exploratory behaviour compared to those offspring who received less mother to pup interaction. By increasing grooming during their first week of life, rat mothers alter the DNA structure of the glucocorticoid receptor gene promoter in the hippocampus of their offspring (Meaney & Szyf, 2005). This evidence paves the way towards understanding risk factors in mental health, especially early life stressors and how they connect to mental disorders following childhood.
Although I am no longer involved in this area of research, I am trying my best to keep up to date with new experiments that will help elucidate the mechanisms underlying mental health disorders, such as depression and PTSD.
If anyone has interesting articles or thoughts about this, please let me know. Thank you!
References:
Adamec, R., & Shallow, T. (1993). Lasting effects on rodent anxiety of a single exposure to a cat Physiology & Behavior, 54 (1), 101-109 DOI: 10.1016/0031-9384(93)90050-P
Cohen, H., Zohar, J., Gidron, Y., Matar, M., Belkind, D., Loewenthal, U., Kozlovsky, N., & Kaplan, Z. (2006). Blunted HPA Axis Response to Stress Influences Susceptibility to Posttraumatic Stress Response in Rats Biological Psychiatry, 59 (12), 1208-1218 DOI: 10.1016/j.biopsych.2005.12.003
Hanson, N., Owens, M., Boss-Williams, K., Weiss, J., & Nemeroff, C. (2011). Several stressors fail to reduce adult hippocampal neurogenesis Psychoneuroendocrinology, 36 (10), 1520-1529 DOI: 10.1016/j.psyneuen.2011.04.006
Kheirbek, M., Klemenhagen, K., Sahay, A., & Hen, R. (2012). Neurogenesis and generalization: a new approach to stratify and treat anxiety disorders Nature Neuroscience, 15 (12), 1613-1620 DOI: 10.1038/nn.3262
Kirby, E., Muroy, S., Sun, W., Covarrubias, D., Leong, M., Barchas, L., & Kaufer, D. (2013). Acute stress enhances adult rat hippocampal neurogenesis and activation of newborn neurons via secreted astrocytic FGF2 eLife, 2 DOI: 10.7554/eLife.00362
Meaney MJ, & Szyf M (2005). Environmental programming of stress responses through DNA methylation: life at the interface between a dynamic environment and a fixed genome. Dialogues in clinical neuroscience, 7 (2), 103-123 PMID: 16262207
Schoenfeld, T., & Gould, E. (2012). Stress, stress hormones, and adult neurogenesis Experimental Neurology, 233 (1), 12-21 DOI: 10.1016/j.expneurol.2011.01.008
Snyder, J., Soumier, A., Brewer, M., Pickel, J., & Cameron, H. (2011). Adult hippocampal neurogenesis buffers stress responses and depressive behaviour Nature, 476 (7361), 458-461 DOI: 10.1038/nature10287
Tanapat, P., Hastings, N., Rydel, T., Galea, L., & Gould, E. (2001). Exposure to fox odor inhibits cell proliferation in the hippocampus of adult rats via an adrenal hormone-dependent mechanism The Journal of Comparative Neurology, 437 (4), 496-504 DOI: 10.1002/cne.1297
Thomas, R., Urban, J., & Peterson, D. (2006). Acute exposure to predator odor elicits a robust increase in corticosterone and a decrease in activity without altering proliferation in the adult rat hippocampus Experimental Neurology, 201 (2), 308-315 DOI: 10.1016/j.expneurol.2006.04.010


 (2 votes)
(2 votes)
Thanks a lot for sharing your views and your expert advice, I really hope that this will guide me to face my stress at work.