Epigenetic inheritance of acquired traits via stem cells dedifferentiation/differentiation or transdifferentiation cycles
Posted by Arshak Alexanian, on 8 February 2026
Do cells carry memories of the whole body into the next generation?
For more than two millennia, biologists and philosophers have debated whether traits acquired during life can be inherited. From Hippocrates and Aristotle to Lamarck and Darwin, this idea repeatedly surfaced but was ultimately set aside due to the absence of a convincing biological mechanism.
Recent advances in epigenetics have reopened this question.
In a recent paper, DOI: 10.1016/j.cdev.2024.203928 I propose a conceptual framework for how environmentally induced epigenetic information might be transmitted from somatic tissues to germ cells—not solely through diffusible molecules, but through cellular movement combined with fate plasticity.
A traveler stem cell hypothesis
The central idea is that certain pluripotent or highly plastic adult stem cells—potentially including germline-associated stem cells—may act as epigenetic travelers. These cells could circulate through the body, enter developmentally active or regenerating tissues, and undergo cycles of differentiation or transdifferentiation in response to local cues. During these transitions, they would acquire tissue-specific epigenetic modifications.
Importantly, these cells would not remain terminally committed. Through dedifferentiation or further transdifferentiation, they could revert to an uncommitted state while retaining accumulated epigenetic information. During gametogenesis, such cells might be recruited back to the gonads, where they ultimately contribute to germ cells—carrying with them epigenetic memory collected across multiple somatic environments.
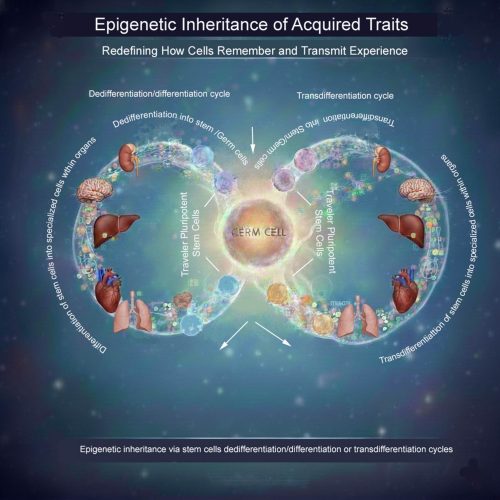
What the image illustrates
The accompanying schematic visualizes this concept: pluripotent “traveler” stem cells move between tissues, repeatedly cycling through differentiation, dedifferentiation, and transdifferentiation. Over time, they integrate epigenetic inputs from diverse organs before re-entering the germline, offering a potential cellular route for soma-to-germline information transfer.
Existing biological foundations
Crucially, elements of this process are not purely hypothetical. Across many multicellular organisms—including plants, invertebrates, and vertebrates—intergenerational and transgenerational epigenetic inheritance has already been experimentally demonstrated. Epigenetic information can persist through extensive developmental reprogramming events and across multiple generations, indicating that biological systems possess robust mechanisms for preserving epigenetic memory.
Moreover, it has been shown that germ cells or germline-associated stem cells are not irreversibly restricted to reproductive fate. Under specific developmental or experimental conditions, germ cells have been observed to generate diverse somatic cell types. Conversely, somatic or pluripotent stem cells can be induced to acquire germ cell identity and contribute to functional gametes. These bidirectional fate transitions challenge a strict interpretation of the soma–germline barrier and establish that germline and somatic identities are more plastic than traditionally assumed.
Together, these observations provide a biological foundation for considering mobile, fate-plastic cells as integrators and carriers of epigenetic information across tissues.
Why this matters
This framework does not contradict existing models of epigenetic inheritance involving small RNAs or other molecular mediators. Instead, it complements them by addressing a key unresolved problem: how complex, tissue-specific epigenetic states accumulated across an organism’s lifetime might be integrated and transmitted coherently to the next generation.
If experimentally validated, this idea could have implications for developmental biology, evolution, aging, regenerative medicine, and disease inheritance.
A question for the community
If highly plastic stem cells can act as mobile carriers of epigenetic memory, how might we experimentally trace their movements, fate transitions, and epigenetic histories across tissues and generations?
I would welcome thoughts on experimental strategies—or alternative interpretations—that could test or challenge this hypothesis.


 (No Ratings Yet)
(No Ratings Yet)