Shape the leaves
Posted by JiyanQI, on 10 January 2018
Before I started my PhD study, I didn’t notice that leaves have two sides: the adaxial side and the abaxial side. When my supervisor Dr. Yuling Jiao first asked me whether I would like to work on this leaf dorsiventral developmental process, I thought I should try, for my own curiosity.
The leaf dorsiventral polarity
For animals, embryonic symmetry is first broken by the dorsal-ventral axis. In seed plants, leaf primordia initiate without polarity at the beginning, but the dorsiventral (adaxial-abaxial) pattern is established quickly afterward. We can see from the pictures below, P1 (the youngest stage) is still a small dome, P2 starts to form curvature, and P3 has already formed epidermal trichomes and a clear adaxial-abaxial difference.

In mature leaves, the side towards the sun is the adaxial side and the other side is the abaxial side. The adaxial sides are greener and have more cuticle than the abaxial sides. The position and structures of the two sides also reflect different functions of the two domains. For instance, we know that leaf is an important photosynthetic organ, and the adaxial side is responsible for light harvest, while the abaxial side is responsible for gas exchange. Thus, proper adaxial-abaxial polarity is important for efficient photosynthesis. Furthermore, the establishment of adaxial-abaxial polarity directs leaf blade expansion, ensuring sufficient space for photosynthesis.
The leaf has long been a good model to investigate polarity patterning (Sussex, 1951). Although extensive molecular genetic research has revealed the genetic regulatory networks of leaf adaxial-abaxial polarity formation, how gene activity ultimately directs organ shape remains unclear.
What did we find?
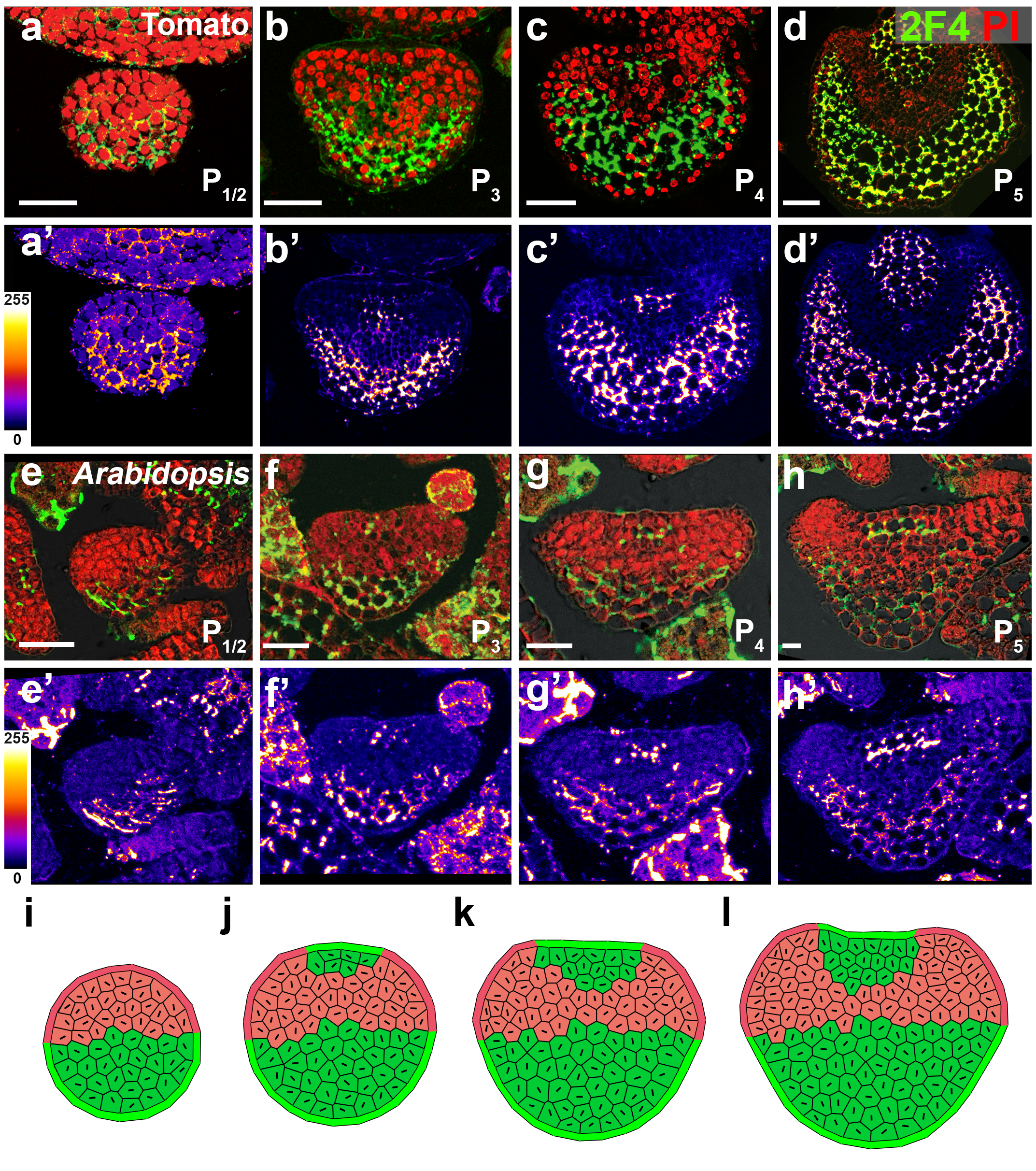
In our previous work, we found there was an auxin gradient in the leaf primordium, and that it was led by PIN1-efflux dependent auxin flow back to the meristem after the leaf initiation (Qi et al., 2014). The acid growth theory suggests that auxin can activate cell wall acidification and triggers cell expansion (Cosgrove, 2005). So different auxin levels within the leaf may lead to different cell wall properties. Therefore, we tested the cell wall thickness with transmission electron microscopy, but did not find any difference in thickness between the adaxial side and the abaxial side in young leaf primordia. We then changed our focus to look at cell wall modification. The major chemical composition of the cell wall are cellulose, hemi-cellulose and pectin. Pectin is complex and heterogeneous group of polysaccharides, one of them is homogalacturonan (HG). HG is mostly methyl-esterified to form stiff cell walls, and however de-methyl-esterification of HG can keep the cell wall pliant and lead it to expansion. Interestingly, we found that there was a clear difference in pectin HG de-methyl-esterification in the leaf primodium, as in the picture shows below. For example, in the P2/3, pectin HG de-methyl-esterification (green signal) is much stronger in the abaxial side than the abaxial side. We were very excited about this finding, because it showed that cell wall modification was different within a leaf, and this could be a reason for the differential growth.
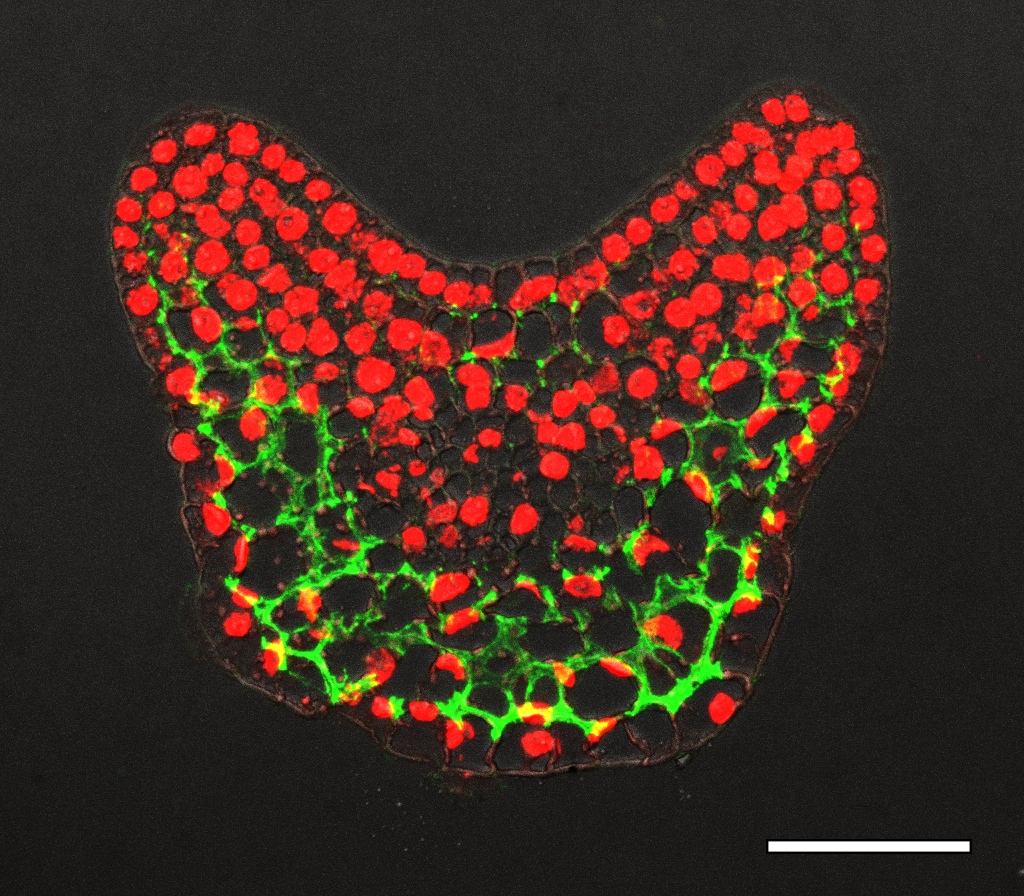
How did we prove our hypothesis?
The difference in pectin modification implies that there would be differences in the mechanics of the cell wall between adaxial and abaxial sides . To this end, we tried to measure the cell wall elastic modulus using atomic force microscopy (AFM). Interestingly, we found that elastic modulus of epidermal cell walls were changing dynamically during leaf asymmetry formation.
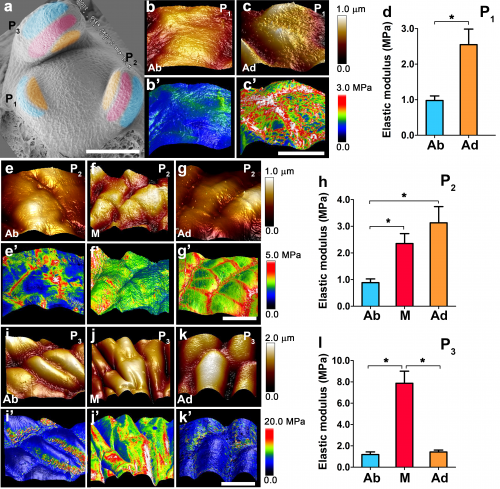
At both P1 and P2 stages, we obtained higher elastic modulus values from adaxial cells than abaxial cells. From the P2 stage, the middle domain starts to stiffen. And then at the P3 stage, the adaxial and abaxial side start to show similar elastic moduli.
Although AFM only measures the upper epidermal cells, the elastic moduli appeared to be consistent with pectin de-methyl-esterification patterns obtained by antibodies in inner cells. Based on direct AFM measurement in tomato and pectin de-methyl-esterification-based inference, the cell wall stiffness may change in the following pattern: at P1/2 stage, cell walls in the abaxial side are relaxed; from the P3 stage, the adaxial side also started to relax, and finally the middle domain joins.
In collaboration with colleagues in the Institute of Mechanics, we then used computational modeling to predict whether the observed dynamics of mechanical properties were sufficient to generate shape changes to produce asymmetric leaves. We applied the model to a 2D template, as a proxy for a leaf cross-section. Starting from a round shape, the morphological evolution of the modeled leaf is governed by energy minimization principle for the inner cells, coordinated with mechanical balance for the epidermis upon various mechanical parameter settings of both inner and epidermal cell walls. Green represents low rigidity, and red represents high rigidity. To mimic the softening behavior in our model setting, as observed experimentally, we allowed a certain number of inner cells and their adjacent epidermal cells to change their properties, when the area of leaf surpassed a threshold value. Such dynamically changing wall mechanics resulted in formation of organ asymmetry that mimics the shape of a P3/4 leaf.

According to what we found, we analyzed the necessity of the mechanical property to the leaf asymmetry. We used microapplication of enzymes and chemicals to decrease cell wall elastic modulus of the adaxial side and to increase cell wall elastic modulus of the abaxial side. Both applications lead to abaxialized symmetry leaves. By connecting gene expression with wall modification and wall elasticity, we were able to link gene activity to leaf adaxial-abaxial growth asymmetry. Difference in wall elasticity based on our AFM results for epidermal cell wall and immunostaining results for inner cells provide a plausible explanation for the observed leaf asymmetric patterning.
Recent work has reported the importance of mechanical regulations in plants and animals, and showed increasing evidence of mechanical influences on development. In our study, we found mechanical dynamic properties changes can account for dynamic shape changes during asymmetric leaf development, providing a simple unifying framework for the control of asymmetric development of organs.
The story behind our recent paper: Jiyan Qi*, Binbin Wu*, Shiliang Feng*, Shouqin Lü, Chunmei Guan, Xiao Zhang, Dengli Qiu, Yingchun Hu, Yihua Zhou, Chuanyou Li, Mian Long★ and Yuling Jiao★ (2017). Mechanical regulation of organ asymmetry in leaves. Nature Plants, 3, 724–733. *: co-first author, ★: correspondent author
References
Cosgrove, D.J. (2005). Growth of the plant cell wall. Nat Rev Mol Cell Biol 6, 850-861.
Qi, J., Wang, Y., Yu, T., Cunha, A., Wu, B., Vernoux, T., Meyerowitz, E., and Jiao, Y. (2014). Auxin depletion from leaf primordia contributes to organ patterning. Proc Natl Acad Sci U S A 111, 18769-18774.
Sussex, I.M. (1951). Experiments on the cause of dorsiventrality in leaves. Nature 167, 651-652.


 (3 votes)
(3 votes)
The adaxial leaf side is darker than the abaxial side simply because the adaxial side faces the sun and has more photosynthesis enhancement than the other side. The adaxial side easily loses its water vapor thereby giving way to plant transpiration and allowing an effective photosynthesis process. The influence of phototropic waves is more at the adaxial side than the abaxial side, thereby causing the adaxial to be darker due to enhanced chlorophyll richness. But the abaxial side is not has dark as the adaxial side because of the effect of gravitropic waves that then pull the leaf’s water down toward the abaxial leaf side. Gravitropic waves can not aid water vapor evaporation from the leaf stomata but the phototropic wave does. We might have an accumulation of water at the abaxial side than the adaxial side. This might even be one of the major reasons why we have discripancies in their spectral signature. The spectral reflectance (NIR) at the adaxial side will be more than the abaxial side. The water accumulation at the abaxial will reflect more blue radiation probably absorbed more (NIR), thereby causing low spectral reflectance of NIR.
The adaxial leaf side is darker than the abaxial side simply because the adaxial side faces the sun and has more photosynthesis enhancement than the other side. The adaxial side easily loses its water vapor thereby giving way to plant transpiration and allowing an effective photosynthesis process. The influence of phototropic waves is more at the adaxial side than the abaxial side, thereby causing the adaxial to be darker due to enhanced chlorophyll richness. But the abaxial side is not has dark as the adaxial side because of the effect of gravitropic waves that then pull the leaf’s water down toward the abaxial leaf side. Gravitropic waves can not aid water vapor evaporation from the leaf stomata but the phototropic wave does. We might have an accumulation of water at the abaxial side than the adaxial side. This might even be one of the major reasons why we have discrepancies in their spectral signature. The spectral reflectance (NIR) at the adaxial side will be more than the abaxial side. The water accumulation at the abaxial will reflect more blue radiation probably absorbed more (NIR), thereby causing low spectral reflectance of NIR.