Towards a mechanistic understanding of branching innovations in plant evolution.
Posted by Jill Harrison, on 29 April 2015
Jill Harrison and Yoan Coudert.
The conquest of land by plants was one of the most significant events in our planet’s history, and was underpinned by a series of innovations in plant architecture. Amongst these, the innovation of branching stands out in allowing plants to colonize new volumes of space in the subaerial environment.
Unlike most plants, living bryophyte representatives of the earliest land plants have a biphasic life cycle with multicellular forms in both the haploid (gametophyte) and diploid (sporophyte) life cycle stages. The dominant photosynthetic phase of the life cycle is the gametophyte, and the sporophyte typically comprises a single ephemeral stem capped in a spore-bearing reproductive structure1.
Sporophytic branching forms are thought to have evolved once, contributing to the radiation of our dominant vascular plant flora (c. 260,000 species). In contrast, distinct gametophytic branching forms have evolved in each bryophyte lineage (c. 16,000 species)2.
Mosses are the most speciose bryophyte lineage (c. 10,000)2,3. Although all mosses are relatively small, having leaves that are a single cell thick, their branching habits are diverse and contribute to their ecology4 (Figure 1).
Figure 1: The diversity of branching forms in mosses. (A-E) Photographs of herbarium specimens of (A) Braithwaitea sulcata, (B) Hypopterygium arbuscula, (C) Cyatophorum bulbosum, (D) Ancistroides genuflexa and (E) Hymenodontopsis stresemannii showing variation in the vertical and radial distribution of lateral branches on the leafy gametophyte. The distribution of the slender leafless sporophytic stems also varies between species. In the species with erect gametophytic forms (A-C), sporophytes are preferentially localised at the top of the shoot, whereas in a species with a pendant form (D), the sporophytes are dispersed. (E) has sporophytes with a lateral and basal position. Dr Yoan Coudert is collaborating with colleagues at the Royal Botanic Garden, Edinburgh and the Natural History Museum in London to characterise evolutionary trajectories between these and other forms using a character mapping approach. Photos by Dr Yoan Coudert, with thanks to NHM for access to specimens. (click to see a bigger image)
There is also an interplay between the gametophytic branching habit and the arrangement of sporophytes on the stem, such that some forms have a single sporophyte at the tip, some forms have a cluster of sporophytes towards the top of the shoot, and others have sporophytes that are dispersed over the plant.
The functional basis and significance of these differences in architecture is not yet known.
Our recent work on the basis of branching patterns in the model moss, Physcomitrella patens, provides a starting point to identify the genetic mechanisms that underpinned the radiation of branching forms in mosses5,6. As there were no previous reports showing how branches arise in Physcomitrella, we started the project by characterising initiation. Using SEM and histology, we found that branches arise spontaneously from the epidermis with a patterned distribution6. Data from flowering plants7, other mosses8, other labs9 and other unpublished projects in our lab led us to believe that a hormonal interplay between auxin, cytokinin and strigolactone could contribute to branching patterns. We used a combination of computational modelling, genetics and pharmacology to show that the integrated action of these three plant hormones determines the distribution of branches up the gametophytic shoot6.
By varying the scope of contributions of each of hormone, we now aim to reproduce the diversity of branching forms in mosses in silico, and will use modelling to generate predictions that allow us to identify the basis of variation between species in future functional work.
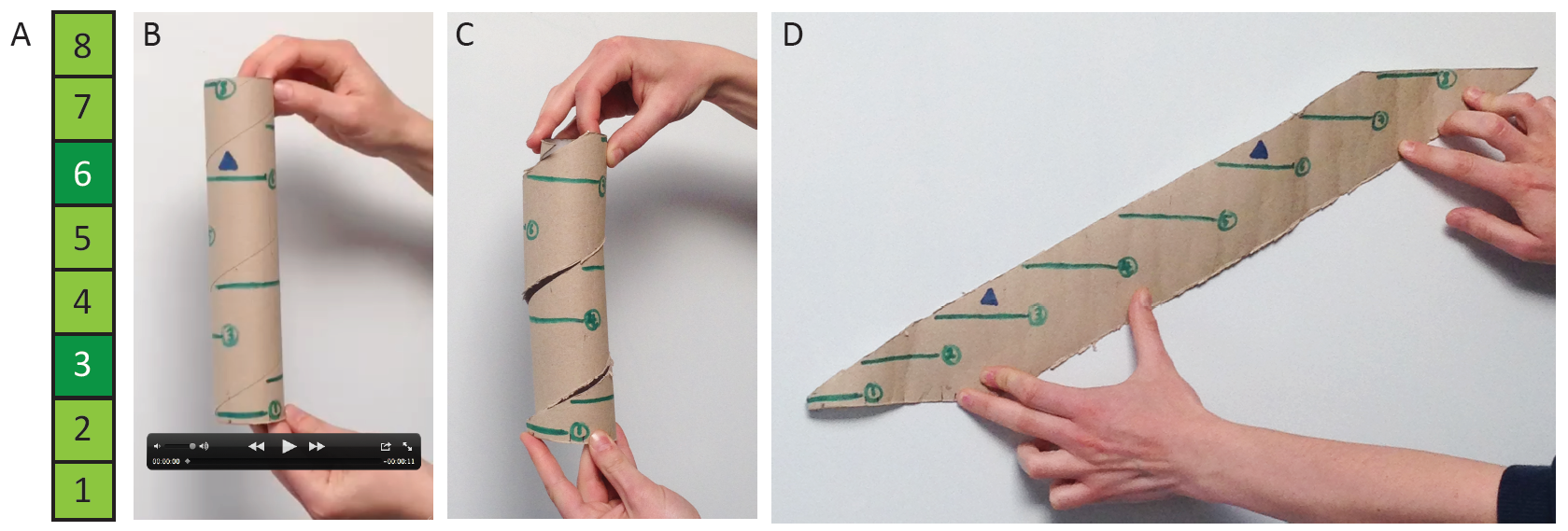
The distribution of branches around the shoot is a key component of moss architecture that we have not yet taken into account (Figure 2), and several studies have indicated that the epidermis of Physcomitrella may be the primary site of auxin response5,10,11.
Figure 2: Gametophytic branching distributions (A) as represented in Coudert et al. (2015), and (B-D) incorporating radial position. (A) Leaves were removed in a numbered series from gametophytic shoots, and if a branch was revealed, the position was recorded with dark green shading. (B) Movie of a rotating kitchen roll holder with green lines representing leaves ascending the shoot with a 137˚ divergence angle, and blue triangles representing a recorded branch distribution. (C) Photograph showing a cut that allowed us to unravel the kitchen roll holder to see (D) the radial distribution of branched represented in 2D. Photos by Dr Jill Harrison and hands from Dr Yoan Coudert. (click to see a bigger image. You can watch the movie below)
As branch initiation is an epidermal phenomenon, we will adapt our 2D modelling approach to analyse 3D branching architectures including radial patterning. We aim to analyse the level and distribution of each plant hormone in relation to the branching distribution with new fluorescent reporter systems in the future.
The work opens the door to mechanistic understanding of the transitions in form that happened during the evolution of branching- one of the defining features of our dominant land plant flora.
Further reading:
1 Langdale & Harrison (2008). ‘Developmental changes during the evolution of plant form‘ in Evolving Pathways: Key Themes in Evolutionary Developmental Biology (ed A. Minelli and G. Fusco) p.299-315.
2 Shaw, A., Szovenyi, P., & Shaw, B. (2011). Bryophyte diversity and evolution: Windows into the early evolution of land plants American Journal of Botany, 98 (3), 352-369 DOI: 10.3732/ajb.1000316
3 Laenen, B., Shaw, B., Schneider, H., Goffinet, B., Paradis, E., Désamoré, A., Heinrichs, J., Villarreal, J., Gradstein, S., McDaniel, S., Long, D., Forrest, L., Hollingsworth, M., Crandall-Stotler, B., Davis, E., Engel, J., Von Konrat, M., Cooper, E., Patiño, J., Cox, C., Vanderpoorten, A., & Shaw, A. (2014). Extant diversity of bryophytes emerged from successive post-Mesozoic diversification bursts Nature Communications, 5 DOI: 10.1038/ncomms6134
4 Farge-England, C. (1996). Growth Form, Branching Pattern, and Perichaetial Position in Mosses: Cladocarpy and Pleurocarpy Redefined The Bryologist, 99 (2) DOI: 10.2307/3244546
5 Bennett, T., Liu, M., Aoyama, T., Bierfreund, N., Braun, M., Coudert, Y., Dennis, R., O’Connor, D., Wang, X., White, C., Decker, E., Reski, R., & Harrison, C. (2014). Plasma Membrane-Targeted PIN Proteins Drive Shoot Development in a Moss Current Biology, 24 (23), 2776-2785 DOI: 10.1016/j.cub.2014.09.054
6 Coudert, Y., Palubicki, W., Ljung, K., Novak, O., Leyser, O., & Harrison, C. (2015). Three ancient hormonal cues co-ordinate shoot branching in a moss eLife, 4 DOI: 10.7554/eLife.06808
7 Domagalska, M., & Leyser, O. (2011). Signal integration in the control of shoot branching Nature Reviews Molecular Cell Biology, 12 (4), 211-221 DOI: 10.1038/nrm3088
8 von Maltzahn, K. (1959). Interaction between Kinetin and Indoleacetic Acid in the Control of Bud Reactivation in Splachnum ampullaceum (L.) Hedw. Nature, 183 (4653), 60-61 DOI: 10.1038/183060a0
9 Proust, H., Hoffmann, B., Xie, X., Yoneyama, K., Schaefer, D., Yoneyama, K., Nogue, F., & Rameau, C. (2011). Strigolactones regulate protonema branching and act as a quorum sensing-like signal in the moss Physcomitrella patens Development, 138 (8), 1531-1539 DOI: 10.1242/dev.058495
10 Bierfreund, N., Reski, R., & Decker, E. (2003). Use of an inducible reporter gene system for the analysis of auxin distribution in the moss Physcomitrella patens Plant Cell Reports, 21 (12), 1143-1152 DOI: 10.1007/s00299-003-0646-1
11 Jang, G., Yi, K., Pires, N., Menand, B., & Dolan, L. (2011). RSL genes are sufficient for rhizoid system development in early diverging land plants Development, 138 (11), 2273-2281 DOI: 10.1242/dev.060582



 (4 votes)
(4 votes)