BSDB Gurdon/The Company of Biologists Summer Studentship Report #23 – Oliver Beaven
Posted by BSDB, on 17 December 2018
Established by the British Society for Developmental Biology in 2014, The Gurdon/The Company of Biologists Summer Studentship scheme provides financial support to allow highly motivated undergraduate students an opportunity to engage in practical research during their summer vacation. Each year, ten successful applicants spend eight weeks in the research laboratories of their choices, and the feedback we receive is outstanding. You can read accounts from previous years here.
Our second report from the 2018 group of student awardees comes from Oliver Beaven (student at Durham), who undertook his project with Colin Jahoda, also at Durham.
Do embryonic mouse tails regenerate?
This summer, with the incredible help of the Gurdon/The Company of Biologists Summer Studentship, I was lucky enough to work under the supervision of Prof. Colin Jahoda in his lab at Durham University. The project I worked on aimed to determine whether epimorphic regeneration occurs in the tips of embryonic mouse tails.
Epimorphic regeneration has scarcely many examples within mammalian systems. The most famous cases among animals are found in the limbs of salamanders, which can regrow fully functional limbs at any stage of their life cycle (Godwin & Rosenthal, 2014). Mammals are far more restricted, with cases found in nail buds, and ear pinnae (Seifert & Muneoka, 2018), but none in hugely complex developmental organs, such as limbs.
The process of epimorphic regeneration begins as the wound site re-epithelializes, and the surrounding tissue dedifferentiates to form a proliferative regeneration blastema. This is then followed by the re-initiation of developmental growth and patterning (morphogenesis). Late last year, the Jahoda lap reported both rapid wound sealing and growth in E13.5 embryonic mouse tails (being studied for other purposes). These highly novel observations therefore correspond with the definition of epimorphic regeneration. Curious, we decided to take a closer look.
The main bulk of my project worked towards elucidating whether the growth which had been observed previously could be classified as actually proliferative, regenerative growth, or simply growth by cellular expansion (not regeneration). I examined this using EdU proliferation markers – a thymidine analogue incorporated into DNA molecules during replication – along with immunohistochemical methods.
The experiments took place with mouse tails removed from E13.5 embryos. Most of the culturing took place on collagen filters, with an EdU pulse three hours before they were frozen down for sectioning. An issue we faced with the collagen filters is the tendency for the wounded end of the ablated tails to adhere to their surface. This would mean of each litter, most tails fail to close fully, blocking us from observing any regeneration in these tails. We tried overcoming this design limitation through the use of 3D hanging drops. While this generally got good results for shorter cultures, long term tail cultures appeared unhealthy. By the end, we tried merging the two methods of short and long-term culture, which seemed to work (unfortunately my time ended before we could generate a refined organ culture method).
Most tails which survived from culture to sectioning contained an EdU profile and an immunohistochemical stain of either fibronectin, or collagen IV, allowing us to identify the position of the EdU stain relative to the basement membrane. We were looking for staining behind the basement membrane, with pronounced upregulation at the tip, which would correspond to the forward growth just beneath the wound site (i.e. within the regeneration blastema). There were several good examples of this taking place when the sections were cut in the middle of the tail (fig. 1 – note that this tail did not have an additional stain). Something notice in this figure is the horizontal orientation of cells right beneath the tip – this is synonymous with patterns seen within regeneration blastema’s, giving support to our hypothesis that epimorphic regeneration was taking place at the tip.
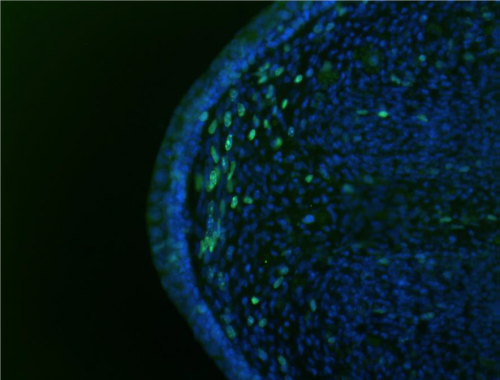
Unfortunately, my finite time in the lab, combined with the harrowing challenge of producing perfect sections at the very centre of the tail, meant our dataset was limited. Consequently, we were unable to confidently state whether the proliferation was congruous with regeneration. This became something of a theme throughout my project; but something I have since come to respect about the nature of research.
We briefly attempted to find whether dedifferentiation occurred on any large scale towards the tip. Our methods again applied immunohistochemistry to observe this epigenetic phenomenon. We used three separate, global histone methylation markers, alongside our EdU analysis. Unfortunately, our markers were too general to notice any significant patterns of dedifferentiation within our samples. It seems the restricted extent to which tissues de-differentiate was matched in their extent of epigenetic repatterning.
Although we are still a way away from positively characterising these phenomena as regenerative processes, it has opened many exciting questions to be explored and debated before a firm statement can be made. Personally, this experience has been exceptionally beneficial for my understanding of research, and has taught me to appreciate new ways to carefully interpret results from a critical perspective. It was also a lot of fun! I would like to send the warmest thanks to Colin and Adam for being endlessly helpful, patient and welcoming, and look forward to hearing about what more results come through in the future.
References
Seifert AW, Muneoka K (2018) The blastema and epimorphic regeneration in mammals. Developmental Biology 433(2):190-199
Godwin JW, Rosenthal N (2014) Scar-free wound healing and regeneration in amphibians: Immunological influences on regenerative success. Differentiation 87(1-2):66-75



 (No Ratings Yet)
(No Ratings Yet)