Uncovering the Regenerative Capacity of Araneus diadematus
Posted by Georgia Henry, on 25 December 2021
Regeneration is the mechanism by which a species can restore damaged or missing cells, tissues, organs, or body parts. Different living organisms have vastly differing regenerative capabilities. Humans, while capable of regenerating some organs following damage or disease, most notably the liver, are very limited in their capacity. Other organisms, however, have remarkable regenerative capacities that facilitate re-growth of entire limbs, parts of nervous systems, or even whole-body regeneration like planarians.
Despite the relative prevalence of regeneration and the potential contributions of its research to modern medicine, the field is widely understudied, especially from a comparative evolutionary perspective (Lai and Aboobaker 2018 Dev Bio).
This summer, the BDSB Summer Studentship gave me the opportunity to undertake an exciting research project, further studying the regenerative abilities of the European Garden Spider, Araneus diadematus, within the McGregor Laboratory at Oxford Brookes, under the supervision of Research Fellow and spider expert, Anna Schönauer.
Fritz Vollrath’s work (Vollrath, 1990) describes the remarkable ability of juvenile A. diadematus (Figure 1, A) to quickly regenerate functional legs from the coxa-trochanter joint following autotomy (Figure 1, B). This functionality is crucial to their survival, as the spiders rely on the limbs’ sensory-motor abilities for weaving their geometrically complex webs, which are intricately designed to facilitate capture of prey (Reed et al 1965).
Regeneration is understood to be an ancestral trait in arachnid species (Goss, 1992) and although some lineages have lost this ability (Vollrath, 1990), the evolutionary relationships giving rise to these differences remain unclear. Research into the development of closely related spider species, with and without regenerative capabilities, offers potential insight into the changes that have led to the loss of this trait. Further, as outlined in Karl Ernst von Baer’s Laws of Embryology (Wanninger, 2015), studying the early development of a species offers essential contributions to the uncovering of evolutionary patterns and relationships of characteristics.

The aim of my project was to improve understanding of the regeneration walking legs in the Araneus diadematus. Initially my Objectives comprised:
- Generating a time series to document the species’ post-embryonic development
- Comparing the regeneration of the first walking legs between different instars
- Analysing the leg transcriptome of an A. diadematus leg, to compare with a species unable to regenerate lost limbs – Parasteatoda tepidarioum.
Due to impediments imposed by Covid19, in addition to further challenges thrown up by the unpredictable nature of scientific research, the outcomes and overall procedure of my project ultimately deviated significantly from the parameters of my original aims. Covid regulations limited my lab access early enough in the year, and as a result, I was unable to accurately document the beginning of the time series for the species, obstructing my first aim.
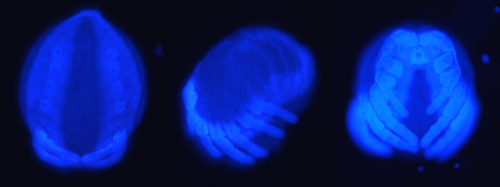
However, acquisition of A. diadematus embryos, contributed an additional branch to the project and supplemented my first objective. Embryos were frozen at different times in development, prior to being peeled, DAPI stained, and microscopically imaged (Figure 2). This contributed to the creation of a partial embryonic time-series, in place of the initially intended post-embryonic time-series.
The examination and comparison of the imaged A. diadematus embryos, with pre-existing images of the P. tepidarioum (Mittman and Wolff 2012) also presents the opportunity to identify significant disparities within the development of the two species, with the potential to propose relevant evolutionary relationships of regeneration, signposted through embryonic development.
The findings from the second objective, analyzing and comparing the regeneration and emergent legs (Figure 3) of individuals within the third and fourth instar, prompted a secondary investigation, examining the effects and implications of the leg regeneration of individuals within the fifth instar. Throughout these analyses, I thoroughly enjoyed the weekly imaging and recording of my experimental subjects’ regenerative development, but nothing quite compared to the excitement of discovering the emergence of a new regenerated leg on my visits to the lab’s spider room first thing in the morning.

Figure 3: The Regenerated Left, First Walking Leg of a Third Instar Female, 21 Days (and 1 molt) After Leg Loss
The final objective (obtaining, analyzing, and comparing the A. diadematus leg transcriptome) is still ongoing and I am learning a lot about the patience required for bioinformatics!
My experiences in the lab have emphasized the fluid and unpredictable nature of scientific study, reinforcing the importance of patience, open mindedness, and flexibility. The Gurdon Summer Studentship has taught me so much that I would not otherwise have had access to in the ordinary course of my undergraduate program. I’ve been involved in lab meetings and journal clubs, worked alongside an amazing group of PhD students, and gained experience using cutting-edge equipment and techniques. I’ve developed lab skills, learned specialized spider husbandry techniques, and grown as a scientist, thanks to the lab team I have been privileged to be a small part of.
I did not underestimate the incredible opportunity being afforded to me and was excited before the project started, but I could not have imagined just how much I would enjoy the process. The unpredictable nature of the field of developmental biology has made for an exciting summer and I owe an enormous debt of gratitude to the BDSB. Further I am beyond grateful to Alistair McGregor and to Anna Schönauer for this incredible opportunity and for their support, encouragement, and training. Their passion for their subject is infectious and would inspire anyone to study further in the field of developmental biology!
References
Goss, R. J. (1992) “The Evolution of Regeneration: Adaptive or Inherent?,” Journal of theoretical biology, 159(2), pp. 241–60.
Lai, A. G. and Aboobaker, A. A. (2018) “Evoregen in Animals: Time to Uncover Deep Conservation or Convergence of Adult Stem Cell Evolution and Regenerative Processes,” Developmental Biology, 433(2), pp. 118–131. doi: 10.1016/j.ydbio.2017.10.010.
Mittmann, B. and Wolff, C. (2012) “Embryonic Development and Staging of the Cobweb Spider Parasteatoda Tepidariorum C. L. Koch, 1841 (syn.: Achaearanea Tepidariorum; Araneomorphae; Theridiidae),” Development genes and evolution, 222(4), pp. 189–216. doi: 10.1007/s00427-012-0401-0.
Reed, C. F. Witt, P. N. and Jones, R. L. (1965) “The Measuring Function of the First Legs of Araneus Diadematus Cl,” Behaviour, 25(1-2), pp. 98–119.
Vollrath, F. (1990) “Leg regeneration in web spiders and its implications for orb weaver phylogeny”. Basel (Suiza): Zoologisches Institute.
Wanninger, A. (2015) “Evolutionary developmental biology of invertebrates”. Wien: Springer (Online access: Springer (t). doi: 10.1007/978-3-7091-1868-9.


 (2 votes)
(2 votes)