Stem cell decisions and the cell cycle
Posted by Erin M Campbell, on 15 February 2013
 A lot of things cycle in life, even down to the cellular level. In the developing central nervous system, regulators of the cell cycle play important roles in maintaining the balance between stem cell self-renewal and differentiation. A recent paper in the journal Development describes a cell cycle regulator in stem and progenitor cells in the nervous system.
A lot of things cycle in life, even down to the cellular level. In the developing central nervous system, regulators of the cell cycle play important roles in maintaining the balance between stem cell self-renewal and differentiation. A recent paper in the journal Development describes a cell cycle regulator in stem and progenitor cells in the nervous system.
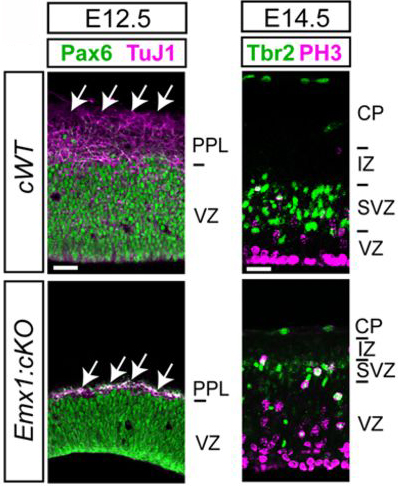
The developing central nervous system depends on the divisions of neural stem cells (NSCs) and intermediate neural progenitor cells (NPCs). In early development, NSCs continuously self-renew in the ventricular zones of the early neuroectoderm. After neuroectoderm specification, NSCs give rise to NPCs, which divide and differentiate into many different types of neurons. Proteins that regulate the progression through different phases of the cell cycle have been shown to regulate NSC and NPC divisions, specifically affecting the balance between proliferative and neurogenic cell divisions. A recent paper in the journal Development identified the role of a zinc-finger transcription factor specificity protein 2 (Sp2) in regulating cell cycle progression in NSCs and NPCs. Liang and colleagues found Sp2 expression in NSCs and NPCs in the embryonic and postnatal CNS. Conditional Sp2-null mice had mitosis-arrested NSCs and NPCs in vivo. In addition, conditional deletion of Sp2 caused a decrease in the number of NPCs and neurons in developing and postnatal brains, as seen in the images above. Compared with normal cerebral cortex tissue in the early mouse embryo (E12.5), Sp2-deleted tissue (bottom) had a reduced number of postmitotic neurons (purple, arrows, left column). NSCs (green, left column) occupied the entire thickness of the Sp2-null cerebral cortex, whereas NSCs occupy only the ventricular zone (VZ) in control tissue. In addition, NPCs (green, right column) were less dense in Sp2-null tissue a bit later in development (E14.5), compared with control cerebral cortex.
For a more general description of this image, see my imaging blog within EuroStemCell, the European stem cell portal.
Liang, H., Xiao, G., Yin, H., Hippenmeyer, S., Horowitz, J., & Ghashghaei, H. (2013). Neural development is dependent on the function of specificity protein 2 in cell cycle progression Development, 140 (3), 552-561 DOI: 10.1242/dev.085621


 (1 votes)
(1 votes)