A day in the life of an onychophoran lab
Posted by Sandra Treffkorn, on 7 August 2019
What are onychophorans and why do we study them?
My name is Sandra Treffkorn, and I recently finished my PhD in the department of zoology lead by Georg Mayer at the University of Kassel, Germany. In our research group, we focus on studying the evolution of animal diversity by investigating two very interesting but largely understudied taxa, the Onychophora and the Tardigrada. Tardigrades, commonly known as “water bears”, are well known to scientists and non-scientists alike due to their ability to survive extreme environmental conditions including exposure to space [1-3]. In contrast to tardigrades, onychophorans comprise a less known animal group even among biologists. Hence, I will use this opportunity to introduce you to these exciting animals and give you a short overview of our research.
When we get asked what onychophorans are, we sometimes tend to describe them as worms with legs and a smooth velvety skin because this is basically what onychophorans look like (Fig. 1). This is also reflected in their common name “velvet worms”, which refers to the velvety appearance of their skin. But actually, they are not worms at all. Onychophorans are terrestrial invertebrates that belong to the Ecdysozoa – the clade of molting animals [4, 5]. Together with the tardigrades, they are the closest living relatives of the arthropods (spiders, myriapods, crustaceans and hexapods), which is the most abundant and diverse animal group on our planet. Compared to arthropods, however, the anatomy of onychophorans has changed little since the Early Cambrian period (~520 million years ago) [6], and they resemble fossil lobopodians of that time, which represent stem-group panarthropods (onychophorans + tardigrades + arthropods).
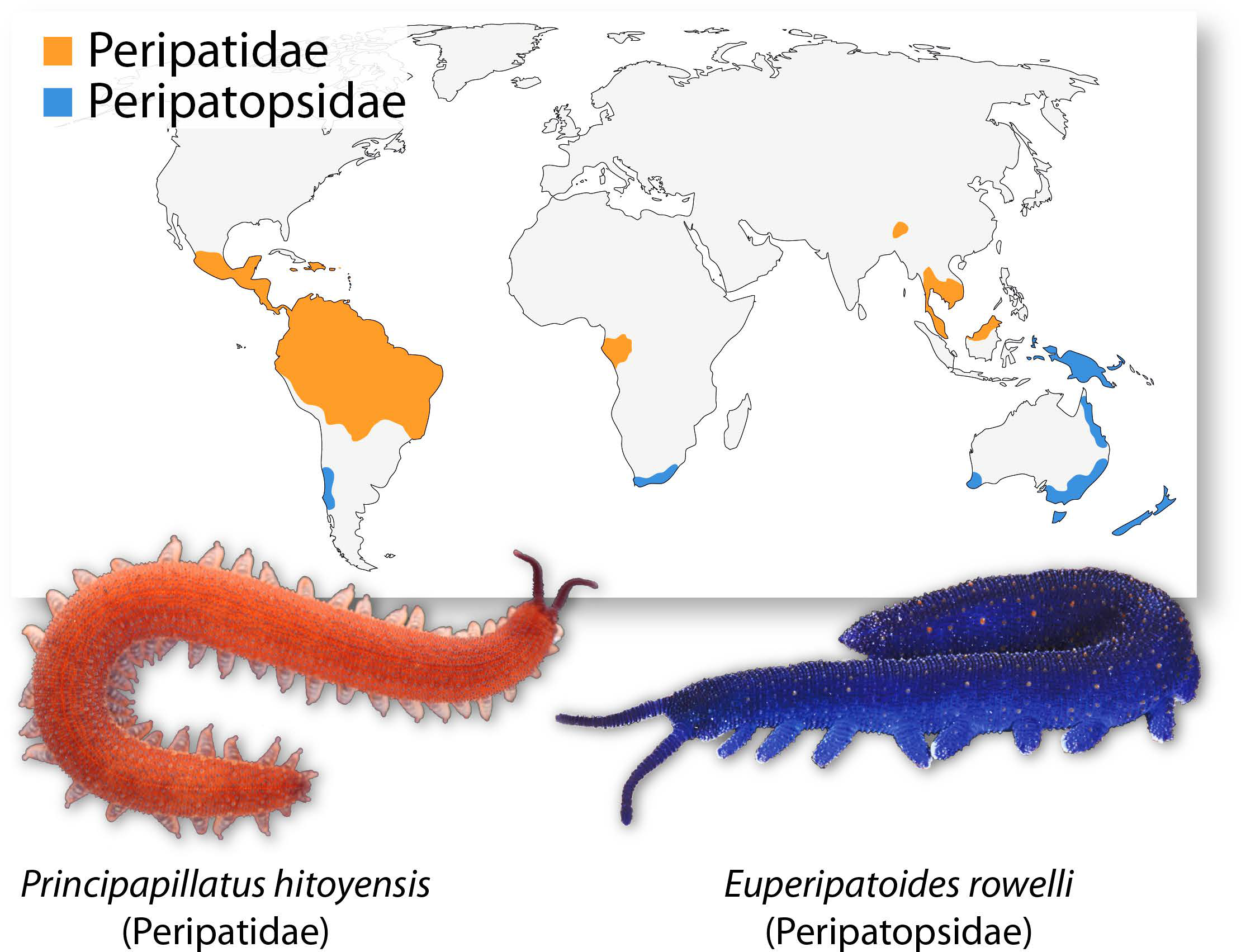
The onychophoran body comprises a head, a uniform worm-shaped trunk bearing a variable number of unjointed limbs, and a limbless posterior anal cone (Fig. 1). The head is composed of three segments, the limbs of which have been modified into three pairs of specialized appendages: the antennae, the jaws, and the slime papillae [7, 8]. The slime papillae are used in a fascinating way to capture the prey: two jets of a sticky secretion are ejected by the slime papillae, which immediately immobilizes the prey (woodlice, crickets and other small invertebrates) [6, 9]. Thus, even though onychophorans are relatively slow, they are very effective hunters. However, the slime is not only used for prey capture but also for defense against predators [6, 9].
Onychophorans typically inhabit humid microhabitats such as leaf litter, soil and decaying logs in tropical and temperate forests. There are currently about 200 described species of Onychophora which are classified into two major groups, the Peripatidae and the Peripatopsidae (Fig. 1) [6, 10]. While the Peripatidae can be found in equatorial areas such as the Neotropics, West Africa and South East Asia, the distribution areas of the Peripatopsidae are mainly restricted to the southern hemisphere, including Chile, South Africa and Australasia (Fig. 1) [6, 10].
But why do we study onychophorans? What’s so interesting about them? The close phylogenetic relationship of onychophorans to two of the most extensively studied model organisms – the nematode Caenorhabditis elegans and the fruit fly Drosophila melanogaster – makes them an attractive model for studying the evolution of animal diversity. Studies of onychophorans as an outgroup to arthropods can be used to polarize morphological and developmental characters of the arthropod ancestor. By extending this comparative approach to cycloneuralians (nematodes, priapulids, kinorhynchs and allies), we are able to make assumptions about the panarthropod ancestor, which in turn might help to clarify the ancestral characters in the ecdysozoan lineage when taking comparative data from spiralians and deuterostomes into account. Hence, onychophorans are a key taxon for understanding the evolution of animal diversity.
Our fields of research
In our lab, we study onychophorans in a wide variety of research fields, including taxonomy, phylogeography, population genetics, species conservation, biochemistry, physiology, behavior, evolutionary developmental biology, neuroanatomy and reproductive biology. Our major goal is to identify and understand the evolutionary changes that have taken place since the divergence of onychophorans from arthropods during the Cambrian radiation over 540 million years ago. Thus, even though working on an emerging model organism can be tricky, and we usually have to develop new methods and optimize existing protocols, the work is quite rewarding since we get the opportunity to look back in time and reconstruct evolutionary events that had taken place hundreds of millions of years ago.
A typical day in an onychophoran lab
Animal collection
For most of our work, we use the onychophoran species Euperipatoides rowelli from Australia, which is one of the most extensively studied onychophorans so far and has recently been established as a model for developmental studies (Figs 1; 2) [6]. Adults of E. rowelli are typically dark-blue colored with more or less prominent, reddish terracotta pattern (Figs 1; 2). Females reach up to 6 cm in length while males are usually smaller.

Before we can start our lab work, however, we actually have to go out and collect animals. Since E. rowelli lives in Australia but our lab is located in Germany, we are not able just to go out and collect animals from the wild whenever we need them. Thus, we have to go on collection trips from time to time to collect animals in Australia, which we then keep and breed in culture in our lab. We usually collect the animals in the Tallaganda State Forest situated about an hour’s drive southeast of Australia’s capital, Canberra (Fig. 2).
Like arthropods, onychophorans possess tracheae as respiratory organs. Unlike arthropods, however, they are not able to close their tracheal openings, which is why they prefer humid environments to avoid drying out (Fig. 2). Typically, onychophorans can be found in the humid leaf litter or under stones. The easiest way to collect them, however, is actually during dry periods when the leaf litter is too dry for the animals and they are all hiding in the decaying logs, which retain a decent level of humidity (Fig. 2). Instead of crawling around on the ground, turning every stone and searching the leaf litter for animals, we can just pry open the decaying logs and collect the animals inside. So, armed with crowbars, screwdrivers, shovels and other tools useful for prying open decaying wood, we head out and collect the animals, which we then put into plastic jars filled with some earth and damp paper towels to keep them safe for the journey around the world to their new home.
Maintenance in the lab
When back in Germany, the animals are cultured in plastic jars containing a layer of peat and damp paper towels, which we keep in climate cabinets under a constant temperature of 18 °C and 60% humidity (Fig. 3).

The first thing to do after a collection trip is to separate the animals. We keep around six specimens – usually three males and three females – together in each jar. And then the most laborious part of our work begins. Of course, we have to take care of the animals to keep them happy and thriving. The paper towels have to be kept damp at all times in order to provide a nice humid environment (Fig. 4). However, the humidity promotes fungal growth, which can harm the animals. So, in addition to maintaining a constant humidity, we replace the paper towels once a week (Fig. 4). Every three weeks, the animals are fed with crickets, which we leave in the jars for two days. Afterwards, we transfer the onychophorans to new jars with fresh peat and paper towels, again to avoid fungal growth due to the decaying cricket remains. Taken together, animal care has to be done at least twice a week and takes several hours each time. This is especially laborious when a new batch of babies is born, which we separate from the parents into new containers. But with a lot of teamwork, it is doable.

Collecting embryos
One major part of our research includes studying embryonic development of onychophorans. For this, we apply a variety of different methods, including in situ hybridization, immunohistochemistry, fluorescence and confocal microscopy, and scanning electron microscopy [11-14]. During my PhD, I specifically analyzed expression patterns of different developmental genes by using in situ hybridization. So, the next step for me was to obtain as many embryos as possible, which is much trickier than for the commonly used model organisms, such as D. melanogaster and C. elegans. In contrast to these model organisms, where embryos are available constantly throughout the year [15, 16], E. rowelli has an annual reproductive cycle with females usually giving birth between November and February under laboratory conditions [17]. Furthermore, the entire embryonic development takes place within the uterus of the mother [6]. In order to obtain embryos of different developmental stages for the experiments (Fig. 5), they have to be dissected from the females manually – usually between September and January – to cover all developmental stages. Thus, we have to plan ahead and get as many embryos as possible during this time, which we then fix and store for long term usage. But since each female typically bears 20 to 40 embryos of different developmental stages, it is possible to cover most stages by dissecting only a few females.
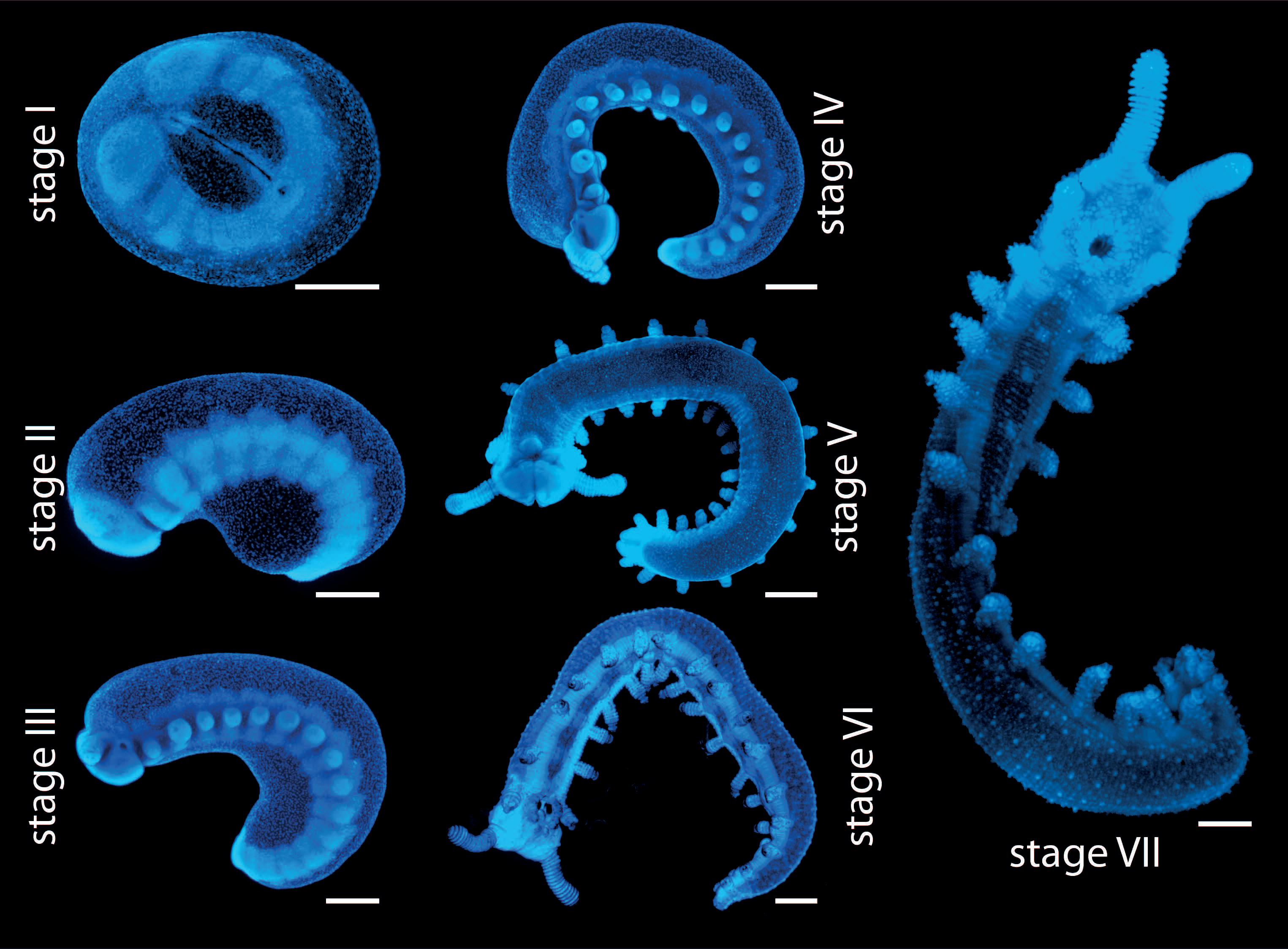
To dissect the embryos, the female is first anaesthetized with chloroform vapor, the entire genital tract is then removed from the female, and the embryos are dissected from the uterus. The yolky embryos of E. rowelli are surrounded by two envelopes: an inner vitelline membrane and an outer chorion, which both persist until birth [6]. These envelopes are removed manually using fine forceps without damaging the embryo. This is especially difficult for the early developmental stages, which are extremely fragile, but with a bit of practice this becomes easier over time. After removing the envelopes, the embryos are fixed using different fixatives (depending on the experiments they are used for) and then stored until they are needed. Treated this way, the embryos can be stored for years.
Establishing a permanent culture of E. rowelli in the lab
Another problem with collecting embryos is that, thus far, we were unable to establish a stable culture in the lab that would reproduce over several generations. Newly collected females usually give birth for two or three years in culture and then stop reproducing. The F1 generation that is born in the lab from the females collected from the wild might also give birth in the lab, but beyond this, reproduction by the following generations has never been reported under laboratory conditions. So, in contrast to other model organisms, we need to collect new animals from the wild every couple of years for our research on embryonic development. One of our future goals is to establish a permanent culture of E. rowelli, which would reproduce in the lab by experimenting with different culturing conditions. However, due to the long gestation period of onychophorans, this is a long-term task. For now, our best option is to go on collection trips to Australia every once in a while.
I hope I could show you what a typical day in an onychophoran lab looks like. If you are interested in onychophorans, or are looking for further information about these fascinating animals, please feel free to check out our webpages: www.onychophora.com and http://www.uni-kassel.de/go/zoologie.
References
- Boothby TC, Tenlen JR, Smith FW, Wang JR, Patanella KA, Osborne Nishimura E, Tintori SC, Li Q, Jones CD, Yandell M, et al: Evidence for extensive horizontal gene transfer from the draft genome of a tardigrade. Proc Natl Acad Sci USA 2015, 112:15976–15981.
- Jönsson KI, Rabbow E, Schill RO, Harms-Ringdahl M, Rettberg P: Tardigrades survive exposure to space in low Earth orbit. Curr Biol 2008, 18:R729–R731.
- Bemm FM, Burleigh L, Foerster F, Schmucki R, Ebeling M, Janzen C, Dandekar T, Schill R, Certa U, Schultz J: Draft genome of the Eutardigrade Milnesium tardigradum sheds light on ecdysozoan evolution. bioRxiv
- Aguinaldo AMA, Turbeville JM, Linford LS, Rivera MC, Garey JR, Raff RA, Lake JA: Evidence for a clade of nematodes, arthropods and other moulting animals. Nature 1997, 387:489–493.
- Giribet G, Edgecombe GD: Current understanding of Ecdysozoa and its internal phylogenetic relationships. Integr Comp Biol 2017, 57:455–466.
- Mayer G, Franke FA, Treffkorn S, Gross V, Oliveira IS: In Evolutionary Developmental Biology of Invertebrates 3: Ecdysozoa I: Non- Tetraconata. Edited by Wanninger A. Wien: Springer; 2015: 53–98
- Storch V, Ruhberg H: In Microscopic Anatomy of Invertebrates: Onychophora, Chilopoda, and Lesser Protostomata. Volume 12. Edited by Harrison FW, Rice ME. New York: Wiley-Liss; 1993: 11–56
- Ruhberg H, Mayer G: Onychophora, Stummelfüßer. In Spezielle Zoologie Teil 1 Einzeller und Wirbellose Tiere. Edited by Westheide W, Rieger G. Berlin: Springer; 2013: 457–464
- Baer A, Mayer G: Comparative anatomy of slime glands in Onychophora (velvet worms). J Morphol 2012, 273:1079–1088.
- Oliveira IS, Read VMSJ, Mayer G: A world checklist of Onychophora (velvet worms), with notes on nomenclature and status of names. ZooKeys 2012, 211:1- 70.
- Treffkorn S, Kahnke L, Hering L, Mayer G: Expression of NK cluster genes in the onychophoran Euperipatoides rowelli: Implications for the evolution of NK family genes in nephrozoans. EvoDevo 2018, 9.
- Treffkorn S, Mayer G: Conserved versus derived patterns of controlled cell death during the embryonic development of two species of Onychophora (velvet worms). Dev Dyn 2017, 246:403–416.
- Franke FA, Mayer G: Controversies surrounding segments and parasegments in Onychophora: Insights from the expression patterns of four “segment polarity genes” in the peripatopsid Euperipatoides rowelli. PLOS ONE 2014, 9:
- Oliveira IS, Tait NN, Strübing I, Mayer G: The role of ventral and preventral organs as attachment sites for segmental limb muscles in Onychophora. Front Zool 2013, 10:1–18.
- Sommer RJ: In Evolutionary Developmental Biology of Invertebrates 3: Ecdysozoa I: Non-Tetraconata. Edited by Wanninger A. Wien: Springer; 2015: 15–33
- Ashburner M, Roote J: Culture of Drosophila: The Laboratory Setup. Cold Spring Harbor Protocols 2007, 2007:ip34.
- Walker MH, Tait NN: Studies on embryonic development and the reproductive cycle in ovoviviparous Australian Onychophora (Peripatopsidae). J Zool 2004, 264:333–354.
- Oliveira IS, Franke FA, Hering L, Schaffer S, Rowell DM, Weck-Heimann A, Monge- Nájera J, Morera-Brenes B, Mayer G: Unexplored character diversity in Onychophora (velvet worms): A comparative study of three peripatid species. PLOS ONE 2012, 7:
- Reid AL: Review of the Peripatopsidae (Onychophora) in Australia, with comments on peripatopsid relationships. Invertebr Taxon 1996, 10:663–936.
Check out the rest of our ‘A Day in the Life’ series here, and let us know if your model hasnn’t yet been featured!


 (9 votes)
(9 votes)