A new role of an insect steroid hormone: The link between mating and germline stem cells
Posted by Tomotsune Ameku, on 11 August 2016
Tomotsune Ameku, Ryusuke Niwa’s lab, University of Tsukuba, Japan.
Steroid hormones have crucial roles in regulating a broad range of biological processes in most multicellular organisms. They are produced in specialized endocrine organs and act as ligands for the nuclear receptor family of transcription factors. In mammals, sex steroid hormones, such as estrogen and testosterone, regulate sex maturation, reproductive physiology and behavior in both sexes. In insects, the major insect steroid hormones are ecdysteroids, including the most biologically active form 20-hydroxyecdysone (20E). Ecdysteroids are called the “molting hormones”, as they are well-known for coordinating developmental transitions, such as molting and metamorphosis. In contrast, little attention has been paid to “post-developmental” roles of ecdysteroids until recently. However, studies in the past several years have demonstrated that ecdysteroids are also present in adult tissues and regulate diverse biological processes such as reproduction, learning, memory, stress resistance, and lifespan (Uryu et al., 2015).
In 2010, Ables and Drummond-Barbosa at Johns Hopkins University beautifully demonstrated that ecdysone signaling directly controls female germline stem cells (GSCs) (Figure 1), which give rise to mature eggs in adults of the fruit fly Drosophila melanogaster (Ables and Drummond-Barbosa, 2010). Interestingly, their data showed that ecdysteroid signaling components including EcR and its downstream gene cascades regulate GSC maintenance with intrinsic chromatin remodeling factors. In addition, some other important studies have also showed that ecdysteroid signaling is required for GSC regulation (König et al., 2011; Morris and Spradling, 2012). I was an undergraduate student at the time, and very impressed by these works, because their findings supported a link between systemic steroid hormones and adult stem cell self-renewal. On the other hand, I realized that all of these works did not examine whether ecdysteroid biosynthesis is regulated by any environmental factors in the ovaries. It seems to me that this question is fundamental, as ecdysteroid biosynthesis during the larval stages is influenced by external environmental conditions (Niwa and Niwa, 2014a). In addition, I also wondered whether and how ecdysteroid “biosynthesis” in the adult ovaries contributed to the GSC regulation.

Figure 1. Germline stem cells (GSCs) give rise to mature eggs
In 2012, I joined Dr. Ryusuke Niwa’s laboratory at the University of Tsukuba and wanted to tackle my questions stated above using Drosophila. Ryusuke and I spent several months thinking about the direction of our research, and eventually focused on the stimulating effect of mating. The reason why is because it has been known that mating dramatically induces behavioral changes, increased egg laying and decreased receptivity in mated females (Kubli, 2003; Wolfner, 2009). To ask whether mating may affect ecdysteroid biosynthesis, we first measured ovarian ecdysteroid levels in virgin and mated females. I was very glad that our expectation was correct, as we found that mated females showed higher ovarian ecdysteroid levels compared to virgin females in our recent PLoS Genetics paper(Ameku and Niwa, 2016). Moreover, we have confirmed that the post-mating ovarian ecdysteroid production is induced by a peptide from male’s seminal fluid called Sex Peptide, and its specific receptor Sex Peptide receptor, is expressed in the female neurons in the same way as the canonical post mating response in mated females (Ameku and Niwa, 2016).
The next question we raised was about the function of mating-induced ecdysteroid biosynthesis. Taking previous studies into consideration, naturally we decided to investigate the relationship between biosynthesis and the regulation of GSC activity. We indeed found that mating stimulates GSC proliferation in mated females. We concluded that this mating-induced GSC proliferation requires ovarian ecdysteroid production, as GSC proliferation did not occur when we inhibited ecdysteroid production in the ovary by knocking down genes required for ecdysteroid biosynthesis (Niwa and Niwa, 2014b) (Figure 2). Interestingly, our data revealed that ovarian ecdysteroid biosynthesis is regulated by Sex Peptide signaling and mediates mating-induced GSC proliferation. This is the first study to show that GSC is under the control of a characterized neuroendocrine system in response to the mating stimulus (Ameku and Niwa, 2016).
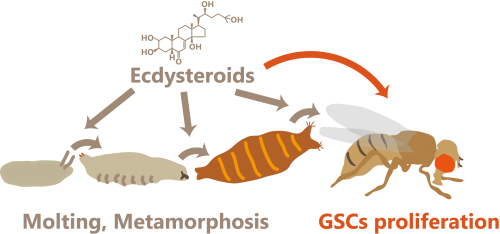
Figure 2. The roles of ecdysteroid during developmental and post-developmental stages
Currently, we are investigating how neuronal input is transmitted to reproductive tissue to regulate GSC activity, which is required for reproductive success. What I have learned from this study is the importance of focusing on biological questions at different levels, such as behavior, neuron, endocrine and stem cells. I hope I will become a researcher who can see things from various perspectives after completing my Ph.D., by making use of my experiences in carrying out this work.
References:
Ables, E. T. and Drummond-Barbosa, D. (2010). The steroid hormone ecdysone functions with intrinsic chromatin remodeling factors to control female germline stem cells in Drosophila. Cell Stem Cell 7, 581–592.
Ameku, T. and Niwa, R. (2016). Mating-Induced Increase in Germline Stem Cells via the Neuroendocrine System in Female Drosophila. PLoS Genet. 12, e1006123.
König, A., Yatsenko, A. S., Weiss, M. and Shcherbata, H. R. (2011). Ecdysteroids affect Drosophila ovarian stem cell niche formation and early germline differentiation. EMBO J. 30, 1549–1562.
Kubli, E. (2003). Sex-peptides: seminal peptides of the Drosophila male. Cell. Mol. Life Sci. 60, 1689–1704.
Morris, L. X. and Spradling, A. C. (2012). Steroid signaling within Drosophila ovarian epithelial cells sex-specifically modulates early germ cell development and meiotic entry. PLoS One 7, e46109.
Niwa, Y. S. and Niwa, R. (2014a). Neural control of steroid hormone biosynthesis during development in the fruit fly Drosophila melanogaster. Genes Genet. Syst. 89, 27–34.
Niwa, R. and Niwa, Y. S. (2014b). Enzymes for ecdysteroid biosynthesis: their biological functions in insects and beyond. Biosci. Biotechnol. Biochem. 78, 1283–1292.
Uryu, O., Ameku, T. and Niwa, R. (2015). Recent progress in understanding the role of ecdysteroids in adult insects: Germline development and circadian clock in the fruit fly Drosophila melanogaster. Zool. Lett. 1, 32.
Wolfner, M. F. (2009). Battle and ballet: molecular interactions between the sexes in Drosophila. J. Hered. 100, 399–410.


 (3 votes)
(3 votes)
prof premraj pushpakaran writes — 2017 marks the 100th birth year of Robert Burns Woodward !!!