The people behind the papers – Wael Noor El-Nachef and Marianne Bronner
Posted by the Node Interviews, on 19 December 2020
This interview, the 85th in our series, was published in Development earlier this year.
The enteric nervous system (ENS) derives from the neural crest and innervates the gastrointestinal system, in which it is essential for gut function throughout life. A new paper in Development uses zebrafish to investigate the poorly understood process of post-embryonic ENS neurogenesis, in both development and injury contexts. To find out more, we met the paper’s two authors, Wael Noor El-Nachef, Assistant Clinical Professor of Medicine at UCLA, and Marianne Bronner, Albert Billings Ruddock Professor of Biology and Biological Engineering at Caltech.

Marianne, can you give us your scientific biography and the questions your lab is trying to answer?
MB: When I started graduate school, I thought that I wanted to do structural biology, but then I took a course in developmental biology and found my passion. I learned about Nicole Le Douarin’s work on the neural crest and it blew me away. I immediately knew that I wanted to work on neural crest cells and be just like her. To this day, she remains my role model. I was fortunate enough to get my first faculty position at the University of California Irvine right out of graduate school. I then moved to Caltech in 1996, where my lab grew larger but continued working on the same types of problems, while adapting new and exciting technologies to help solve them.
My lab pursues diverse questions in neural crest biology, ranging from how and when neural crest cells are induced to how they may have arisen and changed during vertebrate evolution. How and why have neural crest cells continued to acquire new traits? How do they migrate, find their proper sites and differentiate into a diverse array of cell types? Most recently, we have become fascinated by their potential role in tissue regeneration. Wael’s work on de novo neurogenesis in the ENS fits perfectly into these areas of interest.
And Wael – how did you come to work in Marianne’s lab and what drives your research today?
WE-N: I have had somewhat of a non-traditional path into basic science research in that I didn’t get serious about research until after medical school. During my clinical training, I was frequently frustrated by so many ‘idiopathic’ diseases in gastroenterology that are difficult to treat and terrible for the patient, and the most challenging of those conditions often involve the ENS. Rather than wait around for someone else to figure out these problems, I decided to pursue research training in regenerative medicine, initially studying cultured enteric neuronal progenitors in a tissue-engineered intestine construct. Although that was a rewarding experience, I quickly realized that there are many fundamental issues that we do not yet understand about ENS development and homeostasis. And without establishing some of these basics, regenerative medicine approaches to enteric neuropathies will be stymied.
My big revelation came when I realized that ‘developmental biology’ is not limited to embryogenesis, but rather includes postnatal development and growth, aging and senescence, and homeostasis and regeneration. Or, in the words of John Wallingford, ‘we are all developmental biologists’. This perspective expanded my views of the field I wished to join and I reached out to Marianne (I cold called her, and she surprisingly took me on). Training in her lab seems like an obvious choice in retrospect, but in our sometimes cloistered research niches it initially felt a bit mavericky. Luckily, UCLA, my home institution, has an established affiliation with Caltech and has been extremely supportive of my research pursuits.
How has your research been affected by the COVID-19 pandemic?
WE-N: Like so many other scientists, our experimental lab work was placed on hold for a few months while we were on lockdown. Luckily, we are currently ramping-up our activity in the lab; we work in shifts, practice physical distancing, wear masks, etc. It’s great to be back in the lab, and I’m sure I’m not the only one who may have gotten a bit misty-eyed pipetting something for the first time in months. I consider myself extremely fortunate in that I didn’t lose anything other than some time; my family and I are healthy, my fish lines are intact, my projects can restart fairly easily, etc. But returning to the lab within this current COVID-19 paradigm, I now realize the importance of those chance collisions with my colleagues when we informally exchange ideas or troubleshoot a problem together during our daily lab tea time. Videoconferencing is no substitute.
Why has the manner (and even the existence) of postnatal enteric neurogenesis been so controversial?
WE-N: It comes down to this: the gastrointestinal tract is difficult to study. It is arguably the largest endocrinologic organ, largest epithelial organ, largest immunologic organ and second largest neurologic organ; it fills up the largest body cavity in a long convoluted bundle; it is constantly moving and is dynamic in several respects; and to top it all off, it is filled with ‘luminal contents’, which include the largest microbiome compartment of our body. Although this amazing complexity is what first attracted me to this field, there are days when I envy those who study the central nervous system.
In model organisms other than zebrafish, one challenge has been how to directly visualize and detect enteric neurogenesis. Simply counting neurons from a representative section and then extrapolating in some way is not reliable. Live imaging the ENS in species such as mouse is possible but technically challenging and often carried out using a few time points rather than a several-hours long time-lapse. Lineage-tracing experiments with murine Cre transgenic lines have been hampered by relatively low recombination rates. Assessing for evidence of enteric neurogenesis with the use of thymidine analogues has also been complicated by unclear optimal pulse/chase duration and timing. Lastly, studies of ENS homeostasis using non-zebrafish models often investigated the intestinal tract divorced from the rest of the body, and thus may have been unable to consider the possibility of a gut-extrinsic source of neuronal precursors using their methods.
Ultimately, new enteric neurons may arise in small numbers over time and over a large area, and thus may be difficult to detect without fully surveying the ENS. It is also important to acknowledge that alternative hypotheses are not necessarily mutually exclusive, and nature may have provided multiple solutions for addressing issues of ENS homeostasis. But, my experience with the zebrafish model has shown me the advantages of using a model that is highly tractable and amenable to live and in toto imaging to allow us to compellingly explore questions of enteric neurogenesis.
Can you give us the key results of the paper in a paragraph?
WE-N & MB: Using a photoconvertible transgenic line to ‘time stamp’ enteric neurons, we found that enteric neurogenesis persists in post-embryonic zebrafish development, despite an apparent absence of enteric neuronal progenitors, including enteric glia. Lineage tracing with carefully timed DiI injections into the neural tube, as well as with an inducible Cre-transgenic line, supported a trunk neural crest origin of these post-embryonic enteric neurons, consistent with them arising from Schwann cell precursors (SCPs). Enteric neurogenesis was also observed after injury, modelled with two-photon laser ablation of individual neurons. The 5-HT4 receptor agonist prucalopride increased enteric neurogenesis during normal development, as well as in pre-treated fish that subsequently underwent neuronal ablation.
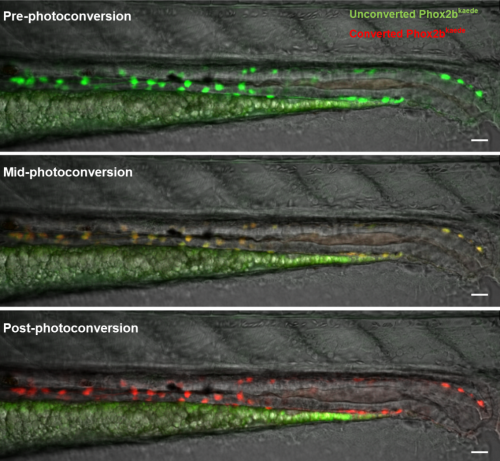
Comparing zebrafish with other species, what can you infer about the contribution of SCPs to the ENS during evolution?
WE-N & MB: In the lamprey, an early jawless vertebrate that appears to lack a vagal neural crest, the ENS originates from the trunk neural crest, suggesting that this may have been the original strategy for providing the basal vertebrate intestine with enteric neurons. However, the evolution of jawed vertebrates was accompanied by the appearance of the vagal neural crest, and the initial formation of the ENS in these animals arose from this newer cell population. We suspect that rather than completely losing the trunk crest contribution to the ENS, later vertebrates adapted this cell population of SCPs to act as a reservoir of ENS progenitor cells that contribute enteric neurons as the intestine grows or to maintain enteric neuron number in the case of injury.
Does your work have any implications for the understanding or treatment of human enteric neuropathies?
WE-N & MB: Certainly. As there is evidence of SCP-derived enteric neurogenesis in teleosts, chicks and mice, it is reasonable to consider that this is also conserved in humans. Many acquired enteric neuropathies are thought to be due (at least in part) to a loss of enteric neurons. In such disease states, promoting enteric neurogenesis with agents such as 5HT4 receptor agonists represents a potential therapeutic approach that is much simpler than trying to rebuild the ENS by injecting precursors that were created in a lab.
Stepping aside from treatment, understanding why a patient’s SCPs are unable to maintain enteric neuron number may lead to a deeper understanding of these pathologies. Is the enteric neuron loss too profound for the SCPs to compensate, or does the disease process involve the SCPs directly? Could enteric neuropathies actually be SCP-opathies?
When doing the research, did you have any particular result or eureka moment that has stuck with you?
WE-N: My first time-lapses that captured new enteric neurons arising in the intestine really energized my efforts. To me, it was exciting, aesthetically pleasing to view, and, simply put, cool. That feeling of seeing something that perhaps no one else has observed before is a very special feeling. I did those experiments early on, and it gave me the confidence that I had the right tools and techniques to explore my research questions in fun and interesting ways.
Seeing something that perhaps no one else has observed before is a very special feeling
And what about the flipside: any moments of frustration or despair?
WE-N: I love coffee. But it turns out that too much coffee on the day you need to dechorionate and microinject zebrafish embryos is not a good thing. Suffice to say that day of experiments was an absolute disaster and had to be rescheduled.
What’s next for you after this paper?
WE-N: I’m very interested in exploring the evolutionary development of enteric glia further. Our results suggest that zebrafish may be missing enteric glia, or at least cells with classical glial properties in the gut. If we can determine when in evolution enteric glia arose, it may help us infer why they arose. Currently, there is some controversy concerning the role of enteric glia in health and disease, and improving our understanding of their function may shine a light on a number of gastrointestinal diseases.
I’m also looking forward to better defining the SCP cell population in zebrafish to aid me in the study of their migration dynamics, and to begin exploring the potential signalling mechanism of 5HT4 receptor agonism in enteric neurogenesis.
Where will this story take the Bronner lab?
MB: We are very interested in evolutionary changes in the ENS and how and when increased complexity arises. Comparing diverse species, such as lamprey, zebrafish and mouse ENS neurons and glia, may offer clues as to how the ENS has become elaborated during the course of vertebrate evolution.
Another direction we are pursuing is using single cell RNA sequencing to examine the heterogeneity of enteric neurons in zebrafish as a function of time. Despite the fact that the zebrafish intestine is comparative simple compared with mouse, there are a diverse array of neurons characterized by different neurotransmitters and neuropeptides. Understanding the molecular underpinnings of how and when these differentiate fascinates me and is a direction we are currently pursuing.
Finally, let’s move outside the lab – what do you like to do in your spare time in California?
WE-N: I have a 1-year-old son, and I’ve recently taken up hiking with him sitting in a special pack that I wear on my back. If I slow down, he’ll bang on my head, so it’s a great workout with very personalized coaching.
MB: I enjoy swimming and hiking, both of which I have been lucky enough to continue during the lockdown. My baking has also improved!


 (No Ratings Yet)
(No Ratings Yet)