Uncovering Non-canonical Roles of E-cadherin Beyond Cell Adhesion
Posted by anups11, on 14 February 2017
Broad perspective
Successful division was an essential criterion for establishing the cell as the basic unit of life on earth. Later, cell-cell adhesion made possible the evolution of multicellular life forms. These two fundamental cellular processes co-function throughout the life of an organism, during development, wound healing and tissue regeneration. In epithelial tissues this results in a curious situation. Epithelial cells, to fulfill their barrier function, need to attach strongly with one another via E-cadherin mediated cell junctions and not leave any gaps between them. However, to divide, cells must round up and form an ingression furrow and therefore detach from their neighbors.
Therefore a ‘cell division conundrum’ exists in the multicellular scenario, wherein the dividing cell is trying to furrow inwards by assembling a contractile actomyosin ring, while the neighboring cells are attempting to keep the tension intact at the boundary by forming cell-cell junctions. In such a scenario an inhibitory regulation between cell-adhesion and cell division machineries would enable the cells to defuse this ‘tug of war’ situation between the two forces.
Convergence on the idea
After completing my Ph.D. with Prof. Mohan Balasubramanian studying cytokinesis in the single cell S.pombe[1], I was eager to address the interplay between cell-cell adhesion and cytokinesis in a multicellular organism. I joined the group of Dr. Ronen Zaidel-Bar, who had recently moved to the Mechanobiology Institute, Singapore and setup his lab to study cell and tissue morphogenesis in mammalian cells and in C. elegans. Co-incidentally, during his post-doc Ronen had observed a genetic interaction between cell-adhesion and cell division mutants in C. elegans embryogenesis and had wondered about possible connections between the two force-generating cellular machineries. However, he never followed up on those results. Therefore, he was extremely receptive to my idea despite the fact that I had never seen a C. elegans nematode before then.
The ride
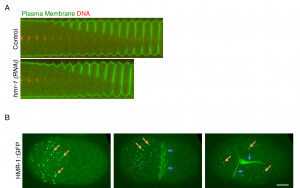
We decided to focus on the early embryo, where the cells are large and divisions are rapid. Additionally, the absence of cell-matrix adhesion in early embryos meant we could focus solely on cytokinesis and cell-cell adhesion. The ortholog of E-cadherin in C. elegans is called HMR-1 [2, 3], and so my first experiment was to deplete HMR-1 by RNA interference (RNAi) and measure the time it took for cytokinetic furrows to ingress during the first, second and third cell divisions (Fig. 1A)[4]. We were really excited to observe faster furrow closure upon HMR-1 depletion during these divisions. This suggested that HMR-1-mediated cell adhesion inhibited cytokinetic furrow ingression and in the absence of HMR-1, this inhibition was released allowing embryonic cells to accomplish faster cytokinesis.
Around the same time, three papers, published back to back in Developmental Cell, reported the existence of a ‘tug-of-war’ mechanism in Drosophila epithelial cells between E-cadherin-mediated cell adhesion and contractile forces of the cytokinetic ring [5-7]. On the one hand, we were pleased our hypothesis received strong reinforcement from Drosophila, but on the other hand we couldn’t help feeling “scooped”.
The First Division Problem
While most of our results were consistent with our hypothesis and the newly published papers from Drosophila, there was one inconsistent result: furrow ingression was faster in the first cell division when HMR-1 was depleted. How could the “tug-of-war between forces” model explain the speeding up of furrow ingression in the 1-cell zygote lacking any cell contacts? The fact was it couldn’t.
To answer this question I analysed the localisation and dynamics of HMR-1 in these early embryonic stages, by imaging Green Fluorescent Protein (GFP) fused to the cytoplasmic domain of HMR-1 protein [8]. As expected, HMR-1 localised to cell-cell junctions in the 2-cell and 4-cell stage (Fig. 1B, blue arrows). Additionally, I also detected HMR-1::GFP as distinct spots or clusters along the non-junctional surfaces in these stages as well as the 1-cell stage (Fig. 1B, orange arrows) [4, 9]. Thus, we concluded, it must be these non-junctional E-cadherin/HMR-1 clusters, which, even though not involved in cell-cell adhesion, slow down cytokinesis. But how do they do it?
Non-Junctional E-cadherin/HMR-1 Dictate a Change in Course
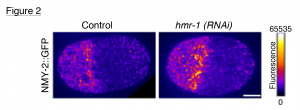
Our unexpected findings in the zygote led to a new question: how do non-junctional E-cadherin/HMR-1 clusters regulate cytokinesis? The first clue to this puzzle emerged when I examined type-II myosin NMY-2, in hmr-1 depleted embryos and control embryos. I detected a significant increase in the levels of NMY-2 at the cortex (Fig. 2) upon HMR-1 depletion and also observed that HMR-1 and NMY-2 excluded each other. Depletion of HMR-1 also resulted in up regulation of cortical RHO-1, the global contractility regulator, which acts upstream of NMY-2. At this juncture we realised that although our analysis of the 1-cell stage might not be along the ‘tug of war’ model, there definitely is an inhibitory relationship between the adhesion and cytokinesis machineries of the cell.
As the zygote initiates first cleavage by assembling a contractile zone bisecting the separating chromosomes, the actomyosin cortex rotates. I found this cortical rotation to be faster in embryos lacking HMR-1 compared to the control. Given that NMY-2 is the major force generator in cells, it seemed natural to attribute the faster cortical rotation as well as the accelerated cytokinesis to the increase in cortical NMY-2. However, when I tested this hypothesis by reducing the amount of NMY-2 recruited to the cortex in both control and hmr-1 (RNAi) embryos, I observed that even when NMY-2 levels were reduced to similar levels, hmr-1 (RNAi) depleted embryos furrowed faster than control, indicating that HMR-1 could influence cytokinesis independently of NMY-2 regulation.
The Discovery of an Unexpected Mechanism
The second clue came to light when I examined the relationship between HMR-1 clusters and cortical F-actin. I detected transient interactions between F-actin at the cortex and HMR-1 clusters embedded in the membrane, and showed that this transient interaction slows the cortical movement akin to stapler pins holding paper sheets from sliding. Since furrow ingression during cytokinesis is clearly a form of cortical deformation, one possible mechanism by which HMR-1 slows down cytokinesis independent of NMY-2 is by physically resisting cortex deformations.
Our studies uncovered a hitherto unknown facet of E-cadherin/HMR-1, wherein it regulates the actomyosin cortex through interactions of its cytoplasmic intra-cellular domain and not the adhesive abilities of the extracellular domain. The intracellular domain – cortex association maintains the structural integrity of the cortex and slows its movement. Compromising this association leads to weakening of the cortex, which under the influence of cellular contractile forces results in faster cytokinetic furrow ingression and in extreme cases can result in splitting of the cortex (Movie1).
Movie 1 – F-actin cortex splitting in hmr-1(RNAi) embryos
Wider Implications
In parallel with my work in the C. elegans zygote, other members of our group working with mammalian epithelial cells have found that non-junctional clusters of E-cadherin can be found all over the cell surface[10, 11]. Whether non-junctional cadherin in mammals regulates the cortex in the same way they do in C. elegans remains to be tested. Nevertheless, given the high frequency of loss of E-cadherin in cancer the possibility that it could affect cellular processes other than cell adhesion, such as cell division and migration, is tantalising.
References
- Padmanabhan, A., Bakka, K., Sevugan, M., Naqvi, N.I., D’Souza, V., Tang, X., Mishra, M., and Balasubramanian, M.K. (2011). IQGAP-related Rng2p organizes cortical nodes and ensures position of cell division in fission yeast. Curr Biol 21, 467-472.
- Costa, M., Raich, W., Agbunag, C., Leung, B., Hardin, J., and Priess, J.R. (1998). A putative catenin-cadherin system mediates morphogenesis of the Caenorhabditis elegans embryo. J Cell Biol 141, 297-308.
- Armenti, S.T., and Nance, J. (2012). Adherens junctions in C. elegans embryonic morphogenesis. Subcell Biochem 60, 279-299.
- Padmanabhan, A., Ong, H.T., and Zaidel-Bar, R. (2017). Non-junctional E-Cadherin Clusters Regulate the Actomyosin Cortex in the C. elegans Zygote. Curr Biol 27, 103-112.
- Herszterg, S., Leibfried, A., Bosveld, F., Martin, C., and Bellaiche, Y. (2013). Interplay between the dividing cell and its neighbors regulates adherens junction formation during cytokinesis in epithelial tissue. Dev Cell 24, 256-270.
- Guillot, C., and Lecuit, T. (2013). Adhesion disengagement uncouples intrinsic and extrinsic forces to drive cytokinesis in epithelial tissues. Dev Cell 24, 227-241.
- Founounou, N., Loyer, N., and Le Borgne, R. (2013). Septins regulate the contractility of the actomyosin ring to enable adherens junction remodeling during cytokinesis of epithelial cells. Dev Cell 24, 242-255.
- Chihara, D., and Nance, J. (2012). An E-cadherin-mediated hitchhiking mechanism for C. elegans germ cell internalization during gastrulation. Development 139, 2547-2556.
- Munro, E., Nance, J., and Priess, J.R. (2004). Cortical flows powered by asymmetrical contraction transport PAR proteins to establish and maintain anterior-posterior polarity in the early C. elegans embryo. Dev Cell 7, 413-424.
- Guo, Z., Neilson, L.J., Zhong, H., Murray, P.S., Zanivan, S., and Zaidel-Bar, R. (2014). E-cadherin interactome complexity and robustness resolved by quantitative proteomics. Sci Signal 7, rs7.
- Wu, Y., Kanchanawong, P., and Zaidel-Bar, R. (2015). Actin-delimited adhesion-independent clustering of e-cadherin forms the nanoscale building blocks of adherens junctions. Dev Cell 32, 139-154.


 (4 votes)
(4 votes)