Bridging the gap between the sub-cellular and the tissue-organ scale
Posted by Stefano De Renzis, on 22 August 2013
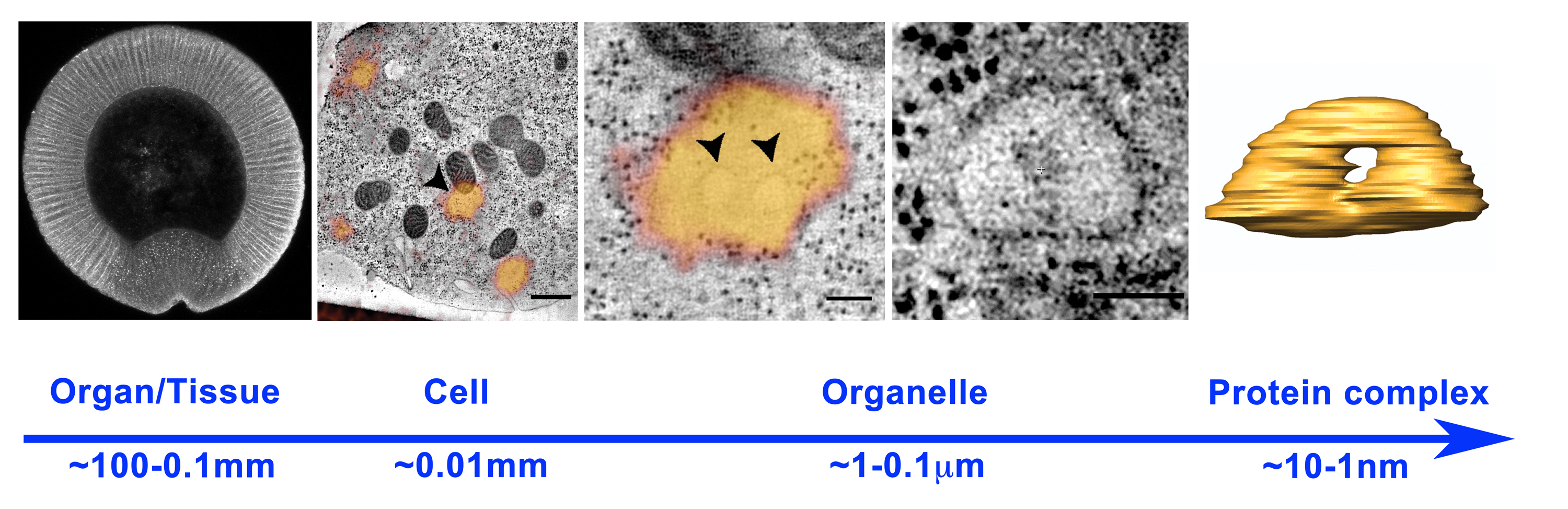
A major challenge when studying the cell biological bases underlying morphogenesis is represented by the gap between the sub-cellular and the tissue-organ scale (Figure 1). Ideally, one would like to follow tissue dynamics with sub-cellular resolution and obtain ultra-structural details about the organization of specific plasma membrane domains, cytoskeletal structures, intracellular compartments and vesicular carriers involved in signaling and adhesion. It is, indeed, the spatio-temporal modulation of membrane and cytoskeletal dynamics in individual cells or group of cells, which ultimately drive tissue remodeling during organismal development. Cell shape changes play, indeed, a fundamental role during embryonic development and are often initiated by expansion or contraction of specific plasma membrane domains. While the role of the cytoskeleton in driving plasma membrane remodeling is well established, the contribution of membrane trafficking remains an open question.
Figure 1. Scales in developmental biology: from organisms (left panel, cross section of a Drosophila m. embryo ~0.5 mm) to organelles (right panel, endosome ~0.5 microns). Tissue and organ morphogenesis depends on the modulation of intracellular machines operating in the nanometer scale.
In my laboratory, for example, we are interested in understanding how endocytosis contributes to shaping cells and tissues during morphogenesis. Although, the role of endocytosis in signaling and cell polarity is well-established, to what extent endocytosis is directly involved in shaping cells is less understood. The cellularization of the early Drosophila embryo provides an ideal system for studying the mechanisms driving plasma membrane remodeling during morphogenesis. Over the course of one hour a syncitium of 6000 nuclei is divided into an equal number of polarized epithelial cells by invagination and growth of the apical plasma membrane (for video see, http://youtu.be/kwN1aNAvTwk). Scanning electron microscopy studies revealed that during this process the apical plasma membrane undergoes a dramatic morphological re-organization characterized by the retraction of villous protrusions and flattening of the apical surface. To date, however, the apical surface of the embryo has proven extremely difficult to visualize in real time using traditional live imaging techniques.
In our recently published paper (Fabrowski P. et al 2013) we adapted Total Internal Reflection Microscopy (TIFR-M) to visualize endocytic dynamics during surface flattening in live Drosophila embryos. This microscopy technique relies upon an evanescent wave that exclusively illuminates a region in close proximity (10-200 nm) to the coverslip. In cell culture, TIRF-M has been successfully employed to visualize vesicle fission and fusion events with the plasma membrane (Toomre D. et al 2000). Furthermore, because of its high sensitivity and low signal to noise ratio, TIRF-M can be applied also to image single molecules dynamics (Jain A. et al 2012).
Imaging living organisms is not as simple as imaging single molecules in solution or cells grown on glass coverslips. In living organisms, such as developing embryos, cells move, are not adhering to coverslips and autofluorescence sometime poses a serious limit to the imaging techniques that can be employed. In the Drosophila embryo, cells are enclosed by the vitelline membrane and are surrounded by peri-vitelline fluid. The vitelline membrane is a proteinaceous waxy layer that tends to be autofluorescent when illuminated with blue (488) light. It seemed therefore unlikely that TIRF-M could be employed to visualize plasma membrane dynamics in live embryos. However, after several failed attempts, we realized that it was possible to direct the laser light with such an angle that an evanescent wave could be generated at the interface between the vitelline membrane and the peri-vitelline fluid. This approach allowed us to visualize, for the first time, the morphogenetic remodeling of the apical surface over the entire course of cellularization (see movie below and also movie S1 in Fabrowski P et al. 2013)
Quantification of endosome dynamics, marked by the small GTPase Rab5 tagged with GFP at its endogenous locus, revealed a massive increase in apical endocytosis that correlates with changes in apical morphology (Figure 2, for video see, http://youtu.be/9X5uM85lBBM).
Figure 2. TIRF-M imaging of apical endocytic dynamics during plasma membrane remodeling in live Drosophila embryo. From left to right, thee snapshots of early, middle and late stages of cellularization showing the up-regulation of endocytic vesicles marked by Rab5 (green). The plasma membrane is labeled by Gap-43 and it is shown in white. Scale bar, 10 microns.
In a series of experiments, which I will not describe here in detail, we demonstrated that endocytosis is required for surface flattening. We, therefore, decided to investigate the endocytic mechanisms driving this morphogenetic process. The large quantities of plasma membrane contained in protrusions together with the relative fast kinetics of flattening (~10 minutes) raised the question of whether clatrhrin coated vesicles mediated endocytosis could, by itself, be sufficient to drive membrane remodeling. To address this question, we employed Correlative Light-Electron Microscopy (CLEM), a powerful, but time-consuming technique, that allows one to correlate fluorescently labeled particles onto a corresponding electron microscope image. By combining CLEM with electron tomography we reconstructed the 3D organization of GFP labeled endocytic membranes. In summary, the results of this analysis revealed that surface flattening is driven by the activation of a prominent tubular endocytic pathway characterized by the formation of tubular plasma membrane invaginations that serve as platforms for the de novo generation of vacuolar Rab5-positive endosomes. Thus, surface flattening is an endocytosis dependent morphogenetic process during which endosomes form directly at the plasma membrane rather than by fusion of incoming clathrin coated vesicles.
In conclusions, the application of these powerful microscopy techniques to multicellular systems will undoubtedly help bridging the gap between the sub-cellular and the tissue-organ scale. The possibility, for example, of visualizing endocytic events during tissue differentiation should help characterizing the molecular regulation and spatial organization of signaling systems in an in vivo context with an unprecedented level of resolution.
References
Jain A, Liu R, Xiang YK, Ha T. (2012) Single-molecule pull-down for studying protein interactions Nat Protoc. 7(3), 445-52. doi: 10.1038/nprot.2011.452.
Toomre D, Steyer JA, Keller P, Almers W, Simons K. (2000) Fusion of constitutive membrane traffic with the cell surface observed by evanescent wave microscopy. J Cell Biol. 149 (1), 33-40.
Fabrowski P, Necakov AS, Mumbauer S, Loeser E, Reversi A, Streichan S, Briggs JA, De Renzis S. (2013) Tubular endocytosis drives remodelling of the apical surface during epithelial morphogenesis in Drosophila. Nat Commun.4:2244. doi: 10.1038/ncomms3244.



 (6 votes)
(6 votes)
You can watch a short video from this paper, narrated by Stefano, in the YouTube channel of EMBL: