Does (brain) size matter?
Posted by beckymcintosh, on 30 October 2015
Chris Puhl and Rebecca McIntosh
As a part of a team of students from the MRC Centre for Developmental Neurobiology, Kings College London we commissioned and edited an issue of The Biochemical Society’s magazine, The Biochemist. The issue is entitled ‘What makes us human’ and is a discussion of the evolutionary steps that lead to modern humans. The articles in the issue were written by a fantastic set of authors. The potential role of connectivity in the evolution of humans was one of many topics we were interested in but didn’t include in the end. Here we share some of our ideas on the topic.
Is a large or highly connected brain more important for human intelligence?
Whenever anyone talks about what makes humans special compared to other animals on earth, our intelligence and self-consciousness is often the first thing mentioned. The cognitive abilities held by humans are usually attributed to the size of our cortex, which is the most rostral, outermost and highly folded region part of the brain. The mammalian cortex is generated by a varied progenitor pool that has expanded and diversified through evolution1,2.
A large number of studies have tried to elucidate the human specific genes and regulatory elements that are involved in generating and regulating mammalian neuronal progenitors in order to make a larger brain during development3,4. Together these studies suggest that as brains evolve to be larger, the stem and progenitor cells that make the brain show increased heterogeneity in both cell biology and gene transcription1-4. This results in blurred lines between stem/progenitor cells and differentiating/committed cells in the developing human brain3,4. It is still unknown whether a larger brain containing more neurons alone can have a subsequent effect on intelligence or cognitive ability. In fact, even authors of these studies have suggested that the evolution of human-like cognitive abilities would have required more than a big brain full of a large number of neurons. Is it possible (even likely) that something else has to change in the brain inorder to evolve consciousness and levels of learning and memory that are considered human traits.
So if not the size of the brain, what other candidate do we have for explaining our unique cognitive abilities? One possibility is that the critical factor lies in how the neurons connect to each other. Greater amounts of cortical connectivity allow for a greater number of neural circuits to exist and thus potentially allow for a more plastic, adaptable brain. This line of thinking can be summarized by: ‘it’s not the size of your brain but what you do with it that matters’.
Increased connectivity
One human-specific genetic change that has been shown to boost synaptic connectivity is the duplication of SRGAP2. Around the transition from Australopithecus to Homo (approximately 3.4 million years ago) an initial duplication event generated SRGAP2B5. This initial duplication was followed by secondary duplication events which generated SRGAP2C ~2.4mya and SRGAP2D ~1mya. Interestingly, these gene duplication events (summarised in figure 1) occur around the time that the neocortex expands, use of stone tools begins, and more complex culture and behaviour emerges6. All of these duplication events were incomplete and were found to generate truncated proteins that assume a dominant negative function and antagonise the ancestral SRGAP2 protein5. Additional quantification of the paralogues found that of the three duplicated genes, only SRGAP2C is expressed in any significant amount in the human brain.
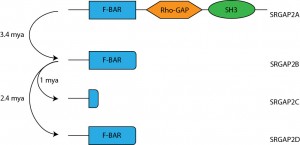
and the evolutionary duplication events generated that them. Adpated from 1.
Charrier et al. 2012 examined the functional consequences of the SRGAP2 gene duplication and the inhibitory interaction with its paralog SRGAP2C7. Inhibition of SRGAP2 led to much faster radial migration of neurons in the cortex with less leading process branching. They also found that expression of SRGAP2C in mouse pyramidal neurons in vitro led to drastically altered morphology of dendritic spines. Dendritic spines receive most of the excitatory inputs for a neuron, and thus any changes to their morphology can have drastic effects on neuronal output. They are also thought to allow neurons to sample inputs over a greater area of nearby space and thus allow for additional connectivity without a corresponding increase in brain size8.
In mice, SRGAP2 promotes spine maturation and limits density. On the other hand, coexpression with SRGAP2C led to increased spine density, lengthened dendritic necks and ultimately led to marginally larger spine heads (figure 2). These ‘mutant’ spines also required more time to fully mature. These results are intriguing as human spines are known to be more dense in the cortex, have longer necks and larger heads, and also to develop more slowly (termed neoteny)9 -11. This experiment then provides an anatomical readout of what may have been occurred in the human brain after the gene duplication event5.
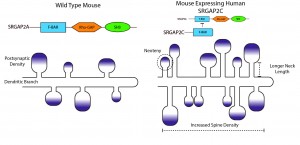
While these studies have shown that a human-specific gene can alter neuronal anatomy, the physicological and functional outcome of the expression of this gene is still not clear. Do the cells expressing both SRGAP2 and SRGAP2C have different electrical properties from those that do not? Do they integrate their inputs differently? Are the rules governing plasticity different or altered for cells with different dendritic spine sizes?
These experiments suggest a plethora of future directions for research. There are a number of human-specific duplicate genes that are incomplete or missing segments12,13. If these genes are important in neurodevelopment or neuronal structure and behavior, then they could assume roles analogous to that played by SRGAP2C to its parent gene. And of course, linking these genetic changes to anatomical and ultimately behavioral changes in neurons would be the ultimate goal in explaining what it is that makes us human.
References
- http://www.biochemist.org/bio/03705/0016/037050016.pdf.
- Gotz M, Huttner WB (2005) The cell biology of neurogenesis. Nat Rev Mol Cell Biol. 2005 Oct;6(10):777-88.
- Florio M, Albert M, Taverna E, Namba T, Brandl H, Lewitus E, Haffner C, Sykes A, Wong FK, Peters J, Guhr E, Klemroth S, Prufer K, Kelso J, Naumann R, Nusslein I, Dahl A, Lachmann R, Paabo S, Huttner WB (2015) Human-specific gene ARHGAP11B promotes basal progenitor amplification and neocortex expansion. Science 347(6229):1465-70.
- Johnson MB, Wang PP, Atabay KD, Murphy EA, Doan RN, Hecht JL, Walsh CA. (2015) Single-cell analysis reveals transcriptional heterogeneity of neural progenitors in human cortex. Nat Neurosci. 18(5):637-46.
- Dennis MY, Nuttle X, Sudmant PH, Antonacci F, Graves TA, Nefedov M, Rosenfield JA, Sajjadian S, Malig M, Kotkiewicz H, Curry CJ, Shafer S, Shaffer LG, de Jong PJ, Wilson RK, Eichler EE (2012) Evolution of Human-Specific Neural SRGAP2 Genes by Incomplete Segmental Duplication. Cell, 149:912-922.
- Jobling M, Hurles M, Tyler-Smith C (2004) Human Evolutionary Genomics. New York: Garland Science.
- Charrier C, Joshi K, Coutinho-Budd J, Kim J-E, Lambert N, de Marchena J, Jin W-L, Vanderhaeghen P, Ghosh A, Sassa T, Polleux F (2012) Inhibition of SRGAP2 Function by Its Human-Specific Paralogs Induces Neoteny during Spine Maturation. Cell 149: 923-935.
- Yuste, R (2011) Dendritic Spines and Distributed Circuits. Neuron, 71:772-781.
- Elston GN, Benavides-Piccione R, DeFelipe J (2001) The pyramidal cell in cognition: a comparative study in human and monkey. Neurosci. 21: RC163.
- Benavides-Piccione R, Ballesteros-Yanez I, DeFelipe J, Yuste R (2002) Cortical area and species differences in dendritic spine morphology. Neurocytology 31:337-346.
- Petanjek Z, Judas M, Simic G, Rasin MR, Uylings HB, Rakic P, Kostovic I (2011) Extraordinary neoteny of synaptic spines in the human prefrontal cortex. Natl. Acad. Sci. USA, 108:13281-132886.
- Sudmant PH, Kitzman JO, Antonacci F, Alkan C, Malig M, Tsalenko A, Sampas N, Bruhn L, Shendure J, Eichler EE ; 1000 Genoms Project (2010) Diversity of human copy number variation and multicopy genes. Science, 330:641-646.
- Fortna A, Kim Y, MacLaren E, Marshall K, Hahn G, Meltesen L, Brenton M, Hink R, Burgers S, Hernandez-Boussard T, Karimpour-Fard A, Glueck D, McGavran L, Berry R, Pollack J, Sikela JM (2004) Lineage-specific gene duplication and loss in human and great ape evolution. PLoS Biol. 2(7):E207.


 (1 votes)
(1 votes)