Fly-CURE: a nation-wide CURE initiative
Posted by Jacob Kagey, on 11 March 2022
Why CUREs?
The 2011 Vision and Change document called for undergraduate STEM majors to have an authentic research experience during their undergraduate careers(Holm, Carter, & Woodin, 2011). There is evidence that research experiences help to increase retention and persistence in STEM (Estrada et al., 2016). One of the biggest challenges of this charge at a PUI (Primarily Undergraduate Institution) is capacity: there are often too few faculty and too many majors, making it impossible to provide traditional mentored research experiences for every student. One way to address this issue is through the implementation of Course-based Undergraduate Research Experiences (CUREs), which embed authentic hypothesis-driven research projects within a credit-bearing undergraduate (usually laboratory) course. This provides a mechanism for one faculty member to provide authentic research experiences for many (20-30) students all at once.
Students who participate in CUREs show gains in psychosocial metrics similar to those students who participated in traditional undergraduate summer research experiences (Shaffer et al., 2014). Additionally, CUREs can reduce the entrance barrier to research for first-generation, historically minoritized, and non-traditional students. Despite all these advantages, there are barriers of entry to CURE implementation:
- Designing a project that can address an authentic research hypothesis and be collaboratively investigated by a large group of undergraduate researchers within the confines of a teaching laboratory course (typically once a week for 3 hours).
- Ensuring that students who take the CURE have experiences and exposures to topics and techniques typically covered in more traditionally taught laboratory courses.
- The facilities, equipment, and funding available to undertake a large-scale research endeavor.
- A significant time investment and skills required to develop an effective CURE that can be broadly implemented.
- Institutional support for the implementation of CUREs.
The Fly-CURE
The Fly-CURE is a national CURE that simultaneously addresses many of these issues. Fly-CURE was started at the University of Detroit Mercy in 2012 and is based on a genetic screen in Drosophila looking for conditional regulators of developmental signaling, cell growth control, and cell division (Kagey, Brown, & Moberg, 2012). In each class section of the Fly-CURE, students work on a novel mutant from the genetic screen. Throughout the semester the students name their mutant; characterize the mosaic phenotype; genetically map the location of the mutation; sequence potential candidate genes; and present their work in written and oral formats.
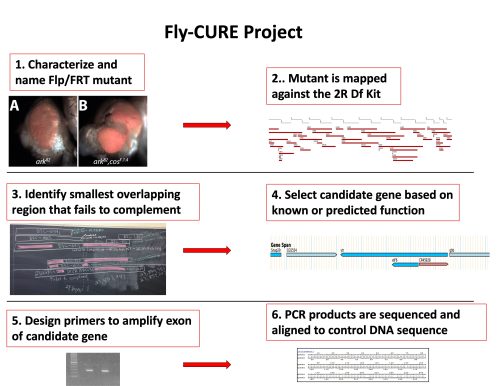
In 2014, the Fly-CURE began to expand to the other institutions. The pilot implementation of the Fly-CURE at the University of Evansville provided evidence that this project could be successfully expanded across the country, dramatically increasing the number of undergraduate STEM majors impacted by this project. Initially, 7 institutions participated in an expanded pilot and in 2021, the Fly-CURE was awarded an NSF IUSE grant to expand the project to 20 institutions. The goals of this grant are to assess the impact of this experience on student attitudes towards science and on key learning objectives in genetics.
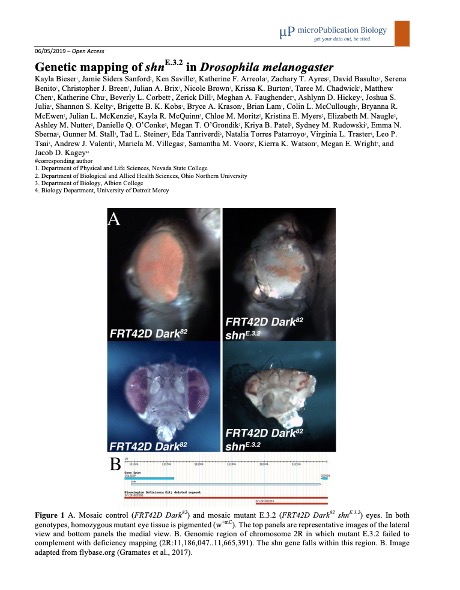
Currently, the Fly-CURE is being taught at sixteen institutions including PUI’s, a CC, and MSI’s. Faculty implementers have a wide range of previous fly experience: some run their own Drosophila research lab, while others had no prior experience with Drosophila before this project. To date, over 500 undergraduate researchers have contributed to the mapping of 14 novel Drosophila mutants, and 6 research papers describing this work have been published with 358 student co-authors across institutions (i.e. (Bieser et al., 2019)).
Adapting to the COVID-19 Pandemic
When traditionally delivered college courses and labs became disrupted during the COVID-19 pandemic, Fly-CURE pivoted to create materials that allowed for different modes of implementation. Faculty taught the Fly-CURE in person, as a hybrid course, or as a fully online virtual CURE. To accommodate hybrid and virtual courses, Fly-CURE faculty collaborated to create virtual datasets of fly crosses typically viewed in person for students to analyze virtually. For other methods, some faculty created videos performing wet-lab protocols for students to continue participation in the project from home. Our assessment data found that students who analyzed virtual datasets were able to map and characterize a novel Drosophila mutant as successfully as those in traditional in-person labs. Our adaptation provides a model for other CURE and inquiry-based undergraduate laboratory courses to generate virtual/online modules of data analysis. The use of virtual datasets not only accommodated differential instruction during the COVID-19 pandemic, but also provides a model for providing virtual research experiences for under-resourced institutions and training materials for new faculty implementers.

Looking Forward
As the Fly-CURE project expands to additional institutions, we have incorporated the expertise and experiences of Fly-CURE faculty to continue improving and expanding the scope of the Fly-CURE project. For example, faculty have added whole genome sequencing and bioinformatics analysis, behavioral assays, and CRISPR analysis of the original mutants found in the genetic screen. We hope to continue to utilize these mutants to bring new techniques and ask new research questions within the classroom setting. Faculty are also conducting new genetic screens and collaborating with the Drosophila community to establish a large pool of mutants for future study by Fly-CURE students.
If you’re interested in learning more about the Fly-CURE, please reach out to one of the Co-PIs:
Kayla Bieser, Nevada State College, kayla.bieser@nsc.edu
Jacob Kagey, University of Detroit Mercy, kageyja@udmercy.edu
Joyce Stamm, University of Evansville, js383@evansville.edu
Alysia Vrailas-Mortimer, Illinois State University, admorti@ilstu.edu
Reference:
Bieser, K., Sanford, J., Saville, K., Arreola, K., Ayres, Z., Basulto, D., . . . Kagey, J. (2019). Genetic mapping of shn(E.3.2) in Drosophila melanogaster. MicroPubl Biol, 2019. doi:10.17912/micropub.biology.000118
Estrada, M., Burnett, M., Campbell, A. G., Campbell, P. B., Denetclaw, W. F., Gutierrez, C. G., . . . Zavala, M. (2016). Improving Underrepresented Minority Student Persistence in STEM. CBE Life Sci Educ, 15(3). doi:10.1187/cbe.16-01-0038
Holm, B., Carter, V. C., & Woodin, T. (2011). Vision and change in biology undergraduate education: Vision and change from the funding front. Biochem Mol Biol Educ, 39(2), 87-90. doi:10.1002/bmb.20502
Kagey, J. D., Brown, J. A., & Moberg, K. H. (2012). Regulation of Yorkie activity in Drosophila imaginal discs by the Hedgehog receptor gene patched. Mech Dev, 129(9-12), 339-349. doi:10.1016/j.mod.2012.05.007
Shaffer, C. D., Alvarez, C. J., Bednarski, A. E., Dunbar, D., Goodman, A. L., Reinke, C., . . . Elgin, S. C. (2014). A course-based research experience: how benefits change with increased investment in instructional time. CBE Life Sci Educ, 13(1), 111-130. doi:10.1187/cbe-13-08-0152


 (1 votes)
(1 votes)