Loving zebrafish with all my heart
Posted by Erin M Campbell, on 8 June 2012
I would have thought that all organisms heal a broken heart the same way humans do (bad movies and cheap wine), but I was wrong. Some organisms, such as zebrafish and newts, are able to regenerate heart tissue where injury, such as myocardial infarction, occurs. Understanding tissue regeneration is a necessary leap in generating successful stem cell therapies. A recent paper in the Development describes the role of TGFβ signaling in zebrafish heart regeneration.
Mammals respond to myocardial infarctions by forming scar tissue at the site of the injury. Zebrafish form scar tissue at the site of an infarction, but simultaneously begin a complex regenerative process to replace the scar tissue with healthy cardiac muscle. This regeneration process involves the temporal and spatial coordination of both cardiac and non-cardiac cells, but the molecular players that regulate this process were unknown. A recent paper by Chablais and Jaźwińska shows that TGFβ signaling is active during heart regeneration. By timing and reversing inhibition of the pathway using a type I receptor inhibitor, Chablais and Jaźwińska found that TGFβ signaling is key for several steps throughout regeneration—scar deposition, tissue remodeling, and cardiomyocyte proliferation.
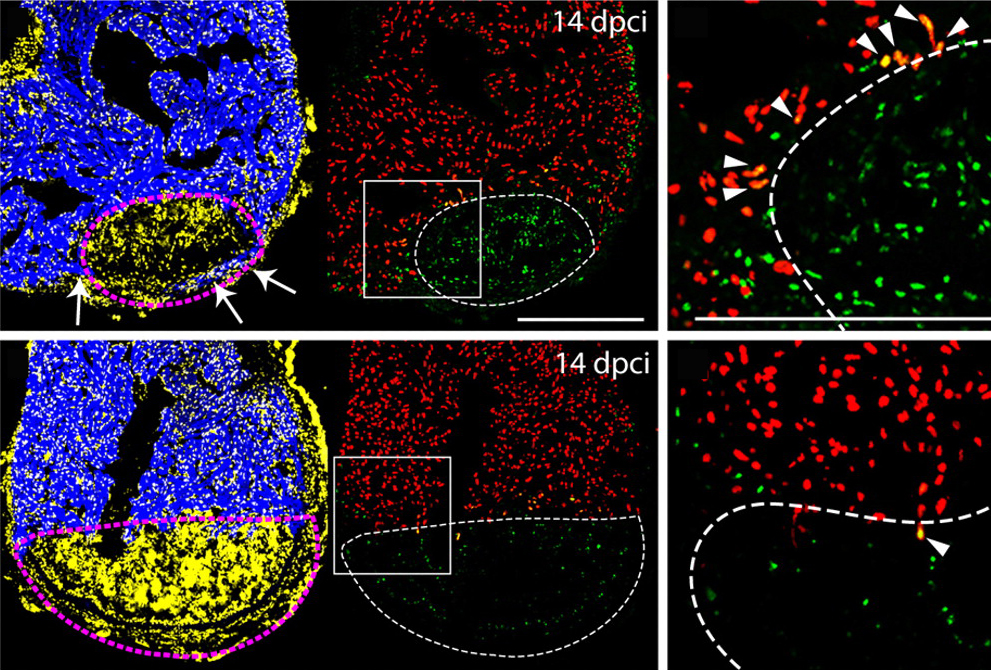
The images above show zebrafish heart sections after cryoinjury, which mimics myocardial infarction (left column: injured area in yellow, healthy tissue in blue). In control sections (top row), cells both within and at the boundary of (arrowheads in zoomed image, seen in cardiomyocytes) the injured area show staining for phospho-Smad3 (green), a direct downstream TGFβ signal transducer. New myocardium can be seen at the boundary of the injury (arrow in top left). Treatment with the type I receptor inhibitor (bottom row) successfully suppressed pSmad3 staining and the invasion of cardiomyocytes, and eventually caused ventricular deformation.
For a more general description of this image, see my imaging blog within EuroStemCell, the European stem cell portal.
![]()
Chablais, F., & Jazwinska, A. (2012). The regenerative capacity of the zebrafish heart is dependent on TGF signaling Development, 139 (11), 1921-1930 DOI: 10.1242/dev.078543


 (3 votes)
(3 votes)