Seeing a future for retinal regeneration
Posted by Erin M Campbell, on 14 June 2013
 Regeneration is a superpower not just reserved for superheroes—salamanders and newts are able to regenerate lost limbs and tails, and fish can regenerate new retinal neurons after injury to the eye. Mammals have limited ability to regenerate retinal neurons, but a recent paper in Development finds that a single transcription factor may be able to change that.
Regeneration is a superpower not just reserved for superheroes—salamanders and newts are able to regenerate lost limbs and tails, and fish can regenerate new retinal neurons after injury to the eye. Mammals have limited ability to regenerate retinal neurons, but a recent paper in Development finds that a single transcription factor may be able to change that.
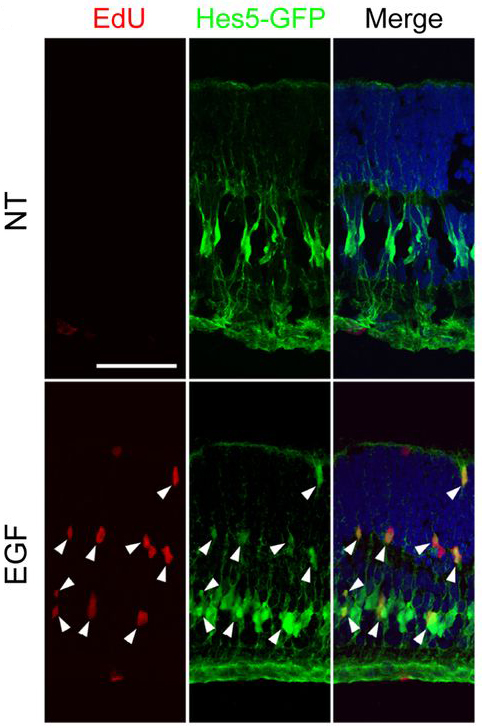
In fish, chemical- or light-induced damage to the eye’s retina drives retinal neuron regeneration. In this pathway, Müller glial cells re-enter the cell cycle and de-differentiate into multipotent progenitor cells able to differentiate into any type of retinal neuron. The transcription factor Ascl1 (Mash1 in mammals) is upregulated shortly after injury, and is required for retina regeneration. Mammals do not upregulate Ascl1 after injury, and have a limited ability to regenerate injured retinal neurons. A recent study in Development investigated if ASCL1 alone could induce the retinal neuron regeneration pathway in mammals. Pollak and colleagues overexpressed Ascl1 in mouse Müller glial cells and intact retinal explants, and found that ASCL1 upregulated retinal progenitor genes and downregulated glial genes. ASCL1 remodeled chromatin at the transcription factor’s targets to a more active configuration. These ASCL1-reprogrammed cells have several characteristics of neurons, including morphology and physiological response to neurotransmitters. In the images above, Müller glial cells (green) in entire retina explants treated to overexpress Ascl1 (bottom row) re-entered the cell cycle (red, arrowheads). Control retina explants are in the top row. From these results, Pollak and colleagues suggest that ASCL1 overexpression may provide a strategy for repair of the retina after injury or disease in humans.
For a more general description of this image, see my imaging blog within EuroStemCell, the European stem cell portal.
![]() Pollak, J., Wilken, M., Ueki, Y., Cox, K., Sullivan, J., Taylor, R., Levine, E., & Reh, T. (2013). ASCL1 reprograms mouse Muller glia into neurogenic retinal progenitors Development, 140 (12), 2619-2631 DOI: 10.1242/dev.091355
Pollak, J., Wilken, M., Ueki, Y., Cox, K., Sullivan, J., Taylor, R., Levine, E., & Reh, T. (2013). ASCL1 reprograms mouse Muller glia into neurogenic retinal progenitors Development, 140 (12), 2619-2631 DOI: 10.1242/dev.091355


 (1 votes)
(1 votes)