The people behind the papers – Vincent Mouilleau, Célia Vaslin and Stéphane Nedelec
Posted by the Node Interviews, on 19 July 2021
This interview, the 96th in our series, was published in Development earlier this year.
As the vertebrate body axis extends, HOX genes are sequentially activated in axial progenitors to specify their identity. A new paper in Development addresses what regulates the tempo of this HOX expression in human progenitors. To hear more about the story, we caught up with the paper’s two first authors, Vincent Mouilleau and Célia Vaslin, and their supervisor Stéphane Nedelec, Group Leader at the Institut du Fer à Moulin in Paris.

Stéphane, can you give us your scientific biography and the questions your lab is trying to answer?
SN: I studied Biology in Rennes and then Paris, where I did my PhD in the Department of Biology of the Ecole Normale Supérieure. I had the chance to work in the group of Alain Prochiantz under the supervision of Alain Trembleau, where I studied local protein synthesis in neurons. To follow up on this research I then joined Hynek Wichterle, who had recently started his lab at Columbia University. Hynek is a pioneer of in vitro differentiation of pluripotent stem cells (PSCs) to study development and diseases. It was very exciting to work in this environment in the early days of the field with great colleagues and so many things to explore, and this time at Columbia had a profound impact on my scientific career since I was still working on spinal cord development using in vitro approaches. I then moved back to France, working again in a very stimulating environment with Cécile Martinat at the I-STEM institute in Evry. There, I started projects aiming at studying human developmental biology using human PSCs (hPSCs). We developed a powerful approach to assess how extrinsic cues control cell fate and discovered pathways sufficient to convert hPSCs into distinct neuronal subtypes, including spinal motor neurons (MNs). Building on this work, I started a new lab in Paris, at the Institut du Fer à Moulin: a very dynamic and collaborative Neuroscience institute. The current story was largely developed there in collaboration with both the Hynek and Cécile groups, which was very satisfying.
We currently use spinal cord development as a model system to address two interrelated questions. First, the mechanisms by which a limited number of extrinsic factors control human spinal neuronal diversity and morphogenesis – in vitro differentiation of hPSCs is a unique model to approach this question. Second, the mechanisms by which mutations in ubiquitous genes perturb developmental programs to impair selective neuronal populations and cause MN diseases – here we take advantage of developmental studies to improve cell and tissue engineering.
Vincent and Célia – how did you come to work in Stéphane’s lab and what drives your research today?
VM: During my bachelor’s degree in Nantes I became fascinated by stem cells and neurodevelopment. I thus decided to join a Master’s program in Paris focusing on these two topics. The possibility of generating, in vitro, a specific subtype of human cells with the right ‘recipes’ fascinated me, and for my internship I joined the I-STEM institute and Stéphane’s lab. I then moved on to do a PhD, and helped Stéphane set up the new lab in Paris while continuing working between the two institutes. It was an intense but enriching experience.
CV: During my undergraduate studies at Sorbonne Université, I quickly became interested in developmental biology and neuroscience. During a first internship in the lab of Jean Livet in Paris, I studied neural lineages in the chick embryo spinal cord, which confirmed my interest in these two fields. This led me to join Stéphane’s lab, first as an intern and then a PhD student, to investigate molecular mechanisms controlling spinal cord development. The power of the in vitro approaches used in the lab allowed me to decipher signalling mechanisms controlling spinal neural diversification – a subject that fascinates me.
How has your research been affected by the COVID-19 pandemic?
VM, CV & SN: Our lab was completely shut down for 2 months last spring. Afterwards, we worked part-time on site to limit the number of people, which obviously significantly delayed the projects. However, working mostly with in vitro models helped, as it was easier to stop and restart the experiments. Also, the lockdown forced us to focus on writing and planning experiments, which was a positive side effect. Overall, this pandemic has most certainly delayed the progress of our research, but we were fortunate to return to the lab fairly quickly and be able to work in good conditions thanks to the heads of our institute, who did a fantastic job in dealing with the situation.
Before this project, what was understood about the relative influence of extrinsic and intrinsic factors on the pacing of the human HOX clock?
VM, CV & SN: We already knew a lot about the extrinsic and intrinsic mechanisms controlling the sequential induction of HOX genes during axial elongation. However, several aspects remain obscure; notably, the mechanisms pacing the clock within axial progenitors, in particular in humans. It was well established that cis-regulatory sequences within and outside the complexes are important for HOX gene sequential induction, and that progressive changes in chromatin structure along the complexes accompany the progression of the HOX clock. On the other hand, extrinsic factors such as retinoic acid (RA), Wnts, FGFs and GDF11 were shown to induce HOX gene collinear expression or modulate HOX gene expression patterns. However, whether these extrinsic factors were pacing the sequential activation within axial progenitors or were actuating an intrinsic timer was unclear.
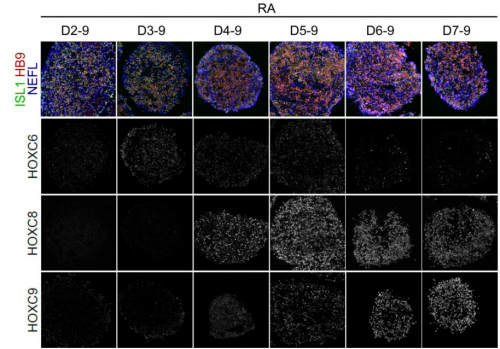
Can you give us the key results of the paper in a paragraph?
VM, CV & SN: We first characterized the expression profile of HOX transcription factors and MN subtype markers in the human embryonic spinal cord, so we could assess the functional consequences of HOX regulation in axial progenitors and properly define the identity of in vitro-generated human MNs. Using MN subtype as a readout, as well as transcriptional analysis of the axial progenitor stages, we showed that HOX genes undergo a temporal collinear activation in hPSC-derived axial progenitors that, upon differentiation, generate MN subtypes found in progressively more caudal regions of the spinal cord. Analysis of the transcriptomic data showed that the sequential activation of HOX genes was paralleled by an increase in FGF ligands and markers of active FGF signalling. This FGF activity was necessary for the HOX clock to proceed, and precociously increasing FGF levels hastened the expression of HOX genes expressed normally later on. The HOX clock was further accelerated with a rapid rise of the very caudal HOX10 genes when FGF was combined with GDF11, another extrinsic factor known to control the expression pattern of caudal thoracic and lumbar HOX genes in mouse and chick embryos. Slowing down or accelerating the clock in axial progenitors was always paralleled by a shift in MN subtype specification within the same time line of differentiation. These results demonstrated that the pace of HOX gene activation within axial progenitors is regulated by sequences of extrinsic factors. This observation argues against a solely intrinsic, chromatin-based, pacing mechanism. However, even in the most accelerating/caudalizing conditions, HOX genes are still expressed in a largely collinear sequence, which suggests that cell-intrinsic mechanisms likely ensure the order of expression. In addition, our work provides for the first time a method to efficiently generate well-defined MN subtypes for basic and translation approaches.
The pace of HOX gene activation within axial progenitors is regulated by sequences of extrinsic factors
Do you have any idea what controls the onset and duration of FGF signalling in hPSC cultures and in the embryo?
VM, CV & SN: The onset and duration of FGF signalling in axial progenitors are certainly controlled by extrinsic factors both in vivo and in vitro. Work from different labs has indicated that FGF and Wnt signals, provided in vivo by the primitive streak and the surrounding epiblast, and in vitro by addition of agonists in the medium, specify axial progenitors, which in turn induce different FGF and Wnt ligands. Thereby, a positive-feedback loop is generated, which likely contributes to an increase in FGF signalling overtime. Accordingly, in this study we observed a temporal induction of FGF ligands and of well-recognized downstream target genes in hPSC-derived axial progenitors. Then, in the neuronal lineage, the duration of FGF signalling in axial progenitors depends on the rate of their differentiation in neural progenitors. In vivo, neurogenesis-promoting RA from the abutting somites can repress FGF gene expression and pathway activity. In our study, the duration of FGF signalling is also likely controlled by the moment at which axial progenitors are exposed to RA. Of note, we also showed that FGF concentration can be integrated by axial progenitors so, in addition to the duration of FGF signalling, a progressive increase in FGF concentration might play a role in pacing the HOX clock. Whether this occurs in embryos is currently unclear.
The dynamics of intracellular signalling downstream of FGFs might also play an important role in rostro-caudal patterning. We showed that FGF activity on HOX genes requires activation of the MAPK pathway. In other models, this pathway adopts distinct signalling activity dynamics in response to variations in concentration or in duration of extrinsic factors. Whether changes in intracellular signalling dynamics downstream of FGFs play a role in HOX clock regulation is an interesting avenue to pursue.
How do you think your findings will impact clinical or bioengineering efforts?
VM, CV & SN: It’s another important aspect of the paper. Studying developmental principles using hPSC differentiation helps optimize differentiation strategies so specific cell or tissues types can then be used for disease modelling, drug screening or cell therapy approaches.
In our case, one consequence of the discovery of the HOX pacing mechanisms is the ability to efficiently and synchronously generate MN subtypes found at different positions in the human spinal cord. MN diseases, such as amyotrophic lateral sclerosis or spinal muscular atrophies, differentially impact MN subpopulations, but the basis of this differential vulnerability remains largely unknown. Providing the community with the ability to generate these MN subtypes might stimulate research on these currently incurable diseases.
Finally, considering the iterative use of HOX transcription factors to induce cell diversity in many lineages, it will be interesting to explore whether our strategy could help refine the production of other cell types, such as somite or neural crest derivatives.
When doing the research, did you have any particular result or eureka moment that has stuck with you?
VM: First seeing the efficient induction of caudal HOX genes while preserving MN induction was a particularly important moment. I’m also happy that we characterized HOX expression patterns and MN subtype markers in human embryos in collaboration with Gist and Mackenzie, who initiated that at Columbia. I think it will be an important resource.
CV: I tested different concentrations of Wnt agonist and I was particularly thrilled when I finally understood why a specific cell line required higher concentration of this agonist to generate caudal MNs. It’s a result that is a bit hidden in the manuscript but might have important consequences when people want to use these protocols with their favourite cell line. I also remember when I assembled images and graphs for the first time to organize the figures, I realized the accomplished work, even if much remained to be done at this time, and I was very proud of all the work we did together with Vincent.
And what about the flipside: any moments of frustration or despair?
CV: Experimentally, it has not always been smooth and easy: hPSCs can be tricky to deal with and are always demanding. However, I had the chance to work very closely with Vincent and constantly support each other, which helped a lot, both scientifically and personally. And finally, discovering such interesting results always cheered us up.
VM: As Celia said, working with hPSCs has its pros and cons as cells need to be taken care of almost every day, and all products and reagents need to be carefully calibrated. We had coating issues at some points and still unexplained cell death at another. These periods were very frustrating.
What next for you two after this paper?
CV: I am currently exploring the signalling mechanisms downstream FGF using reporters of signalling pathway activities. I will defend my thesis in a few weeks. Then I’m favouring a career in a biotech or industry.
VM: After defending my thesis I wanted to look for jobs abroad but the pandemic delayed this project. In the meantime, I’m helping with the national effort to test and track Covid patients while exploring future plans.
Where will this story take the Nedelec lab?
SN: On one hand, as we always try to combine developmental biology with cell engineering, we are exploring what’s downstream of the extrinsic factors that pace the HOX clock, the mechanisms by which they signal to the genome to induce distinct cell fates, and how they control spinal cord morphogenesis (using a new type of organoid model). 3D in vitro differentiation provides an experimentally accessible model for fine modulations of signalling pathways that can be coupled to genomic analysis while tracking consequences on cell fate and tissue shape. In collaboration, we are implementing optogenetic approaches and genomic approaches to address these questions.
On the other hand, we use the products of these developmental studies to study the basis of the differential vulnerability of MN subtypes in different forms of paediatric MN diseases called spinal muscular atrophies. For that, we have created a very stimulating network of collaborators, including clinicians and cell biologists.
Finally, let’s move outside the lab – what do you like to do in your spare time in Paris?
CV: A long time ago, before the pandemic, I really enjoyed living in Paris and often went to the cinema or museum, and I loved to discover new restaurants. But nowadays, it’s more biking or walking in the city and around, when the weather is nice!
VM: I like to walk randomly and get lost in the maze of Paris, discovering new streets and monuments randomly. When it was still possible, I particularly enjoyed waking up early on weekends to go to the Louvre to walk around the museum with almost nobody around. I also really enjoyed going to bars with co-workers to share problems and discuss projects. While I reduced this activity during my PhD, I’m also a big fan of Aikido.
SN: As Célia and Vincent mentioned, Paris will, hopefully, soon be Paris again, so we can enjoy the theatres, museums and the terraces. I also like rock climbing and, while it might sound surprising, Paris is not such a bad place for it. The nearby forest of Fontainebleau is a fantastic bouldering spot with endless possibilities. As my daughter has started to really enjoy it as well, I try to go as much as I can.


 (No Ratings Yet)
(No Ratings Yet)