Zebrafish transgenes go ubiquitous
Posted by Christian Mosimann, on 7 December 2010
The Node’s staff has kindly asked me to write a little “behind the scenes” on our zebrafish paper released today in Development, “Ubiquitous transgene expression and Cre-based recombination driven by the ubiquitin promoter in zebrafish” (http://dev.biologists.org/content/138/1/169).
The spark to pursue the project were the first conversations I had in spring 2008 with senior postdocs in Leonard Zon’s lab at the Children’s Hospital Boston right when I started my postdoc to study hematopoietic cell fate control using zebrafish. Coming straight out of my graduate work on Wnt and Hh signaling in Drosophila, my thinking was centered on mutants, transgenes, and recombinase-mediated manipulations. The zebrafish is still a relatively new, yet increasingly popular, model organism with compelling imaging possibilities and malleable genetics. It was no small shock however to learn how some key molecular tools were not working well or even totally missing in zebrafish. A reoccurring theme was the lack of a truly ubiquitously expressing promoter for transgenes, in particular one that is active in red blood cells and adult organs. I realized that I would need Cre/loxP tools for my project ideas, all of which depend on a ubiquitous promoter such as Rosa26 transgenes in mice, to permanently express a lineage tracer transgene in the cells we wish to track.
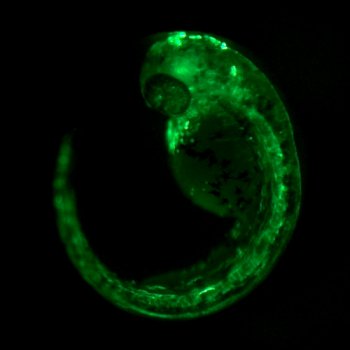
As one of my first practical things in the lab, I therefore assembled the zebrafish ubiquitin (ubi) locus through database searches and ordered primers to amplify the 5’ region of the gene. Why ubi? ubi:GFP are the most reliable transgenic markers in Drosophila for ubiquitous labeling of cells, and I used plenty of such strains in my past projects. After injecting zebrafish ubi promoter-driven EGFP reporter vector into zebrafish embryos, I saw ubi expressed transgenes at all developmental stages and a multitude of adult organs, including cell types such as red blood cells that have so far been missed by quasi-ubiquitous zebrafish promoters such as beta-actin or ef1alpha. The picture below is a mosaic ubi:EGFP embryo from one of these initial injections.
Shortly after these first tests, Charles Kaufman joined the lab as a postdoc. While we discussed during our very first chat his ideas to tackle melanoma formation using transgenic zebrafish, he mentioned requiring a ubiquitous promoter. “There really isn’t a good ubiquitous promoter in zebrafish”, I said. Charles originally trained in mice and was thus used to luxurious molecular genetics tools; so he replied, astonished, “So how are we supposed to do anything then?” “Well, maybe we now have a ubiquitous promoter” I replied and outlined my preliminary data. The rest unfolded as a team effort to first elucidate if ubi is truly that ubiquitous and to subsequently create a tool box for genetic lineage tracing and Cre/loxP-regulated transgenes in zebrafish.
To confirm if ubi truly expresses ubiquitously also in our lab’s favorite tissue, blood, Pulin Li and Emily Pugach successfully carried out adult zebrafish blood transplantation assays and characterized ubi expression in hematopoietic cells. As if timed for the project, Owen Tamplin brought a batch of the original CreERt2 plasmid to the lab when he joined as a postdoc. CreERt2 is a version of the Cre recombinase that is inducible by 4-hydroxytamoxifen (4-OHT). With the precise developmental staging possible in zebrafish embryos, timed addition of 4-OHT to the dish allows for precise temporal activation of a given CreERt2 driver, a principle that has already been successfully established in zebrafish. We therefore generated ubi:creERt2 transgenic zebrafish as a source of ubiquitous inducible CreERt2 recombinase activity and confirmed its sensitivity to 4-OHT at various developmental stages.
To harness the full lineage tracing potential of ubi, we also created ubi:loxP-EGFP-loxP-mCherry, or ubi:Switch as we call it. This transgene initially expresses EGFP ubiquitously, but any cell with active Cre will cut out the EGFP cassette and put mCherry under ubi control, thus indelibly marking this cell and its descendents with fluorescent red throughout development. ubi:Switch now allows simple lineage tracing experiments where ubi:Switch transgenics are crossed to any tissue-specific Cre- or CreERt2-expressing transgenic zebrafish strain of choice, many of which are currently under development in labs around the world.
Future versions of ubi:Switch can easily be cloned to express different fluorescent color combinations tailored to specific experiments. Furthermore, ubi:creERt2 facilitates the future creation of tissue-specific loxP lineage tracing transgenics, which can then be universally tested by crossing to this ubiquitous CreERt2 source. The time spent building these tools was not only great training, but now enables us – and hopefully other zebrafish researchers in the field – to perform lineage tracing experiments and to create new exciting transgenics. The lack of gene knockouts or RNAi technology remains a heavy burden on the zebrafish community. Maybe ubi as new ubiquitous transgene driver resource will now assist the development of these methods. Is ubi the final word on ubiquitous zebrafish transgene promoters? Probably not. But until we find something even more potent, ubi adds another lure to the growing tackle box of zebrafish methods to catch exciting new biology.


 (15 votes)
(15 votes)