Multiple stem cells, population asymmetry and position-dependent heterogeneity emerge as common features of a niche for Drosophila Follicle Stem Cells and mammalian Intestinal Stem Cells
Posted by Amy Ruth Reilein, on 16 June 2017
A discussion of “Alternative direct stem cell derivatives defined by stem cell location and graded Wnt signalling,” Nat Cell Biol, 2017. 19(5): p. 433-444.
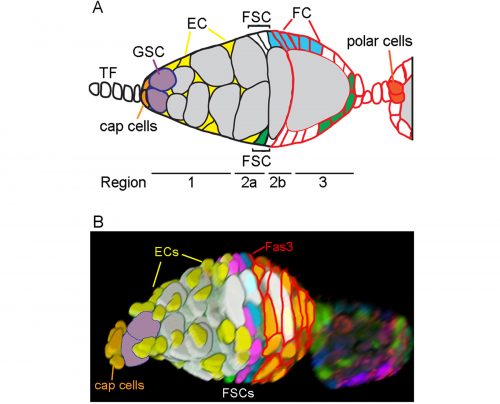
We have recently revised the model of Follicle Stem Cell (FSC) organization in the Drosophila ovary, showing that there is a much larger population of stem cells than formerly realized, that these FSCs exhibit population asymmetry, and that they give rise to Escort Cells as well as Follicle Cells [1]. Ovarian Germline Stem Cells have long been used as model stem cells, benefiting from easy recognition by virtue of their characteristic location at the anterior of the germarium, morphology, and functional markers that reflect the actions of a key BMP (bone morphogenetic protein) niche signal. FSCs, which are required for the development of germline cells into a mature oocyte, have been more difficult to investigate. No marker specific to these stem cells has been discovered, they are not in an easily recognized location and are not morphologically conspicuous among their neighbors.
In 1995 Margolis and Spradling reported the identification of FSCs (originally named Somatic Stem Cells), which they located midway through the germarium by BrdU labeling and lineage analysis [2]. An FSC is defined by lineage analysis as a cell that can produce Follicle Cells (FCs) but remains in the germarium while germline cysts pass through. FSC derivatives that associate with a germline cyst in the posterior half of the germarium all initially proliferate. A few quickly become specialized post-mitotic polar cells and stalk cells to allow egg chamber budding from the germarium, while the rest form an expanding epithelium around the germline cyst, cease divisions in mid-size egg chambers (stage 6) and adopt position-specific fates about a day later (stage 9). We refer here to all of these FSC derivatives including the earliest precursors (often termed prefollicle cells) associated with a germline cyst as Folicle Cells (FCs). Marked lineages that originate in an FC have a predictable, limited lifespan because all FC progeny move through the germarium and ovariole along with the associated cyst at an established rate. It takes the earliest FC about two days to exit the germarium and five days to exit the ovariole. If a marked cell remains in the germarium or the ovariole for longer than this time then we can deduce that the lineage must have originated in an FC precursor, namely an FSC. Although this definition has been used since 1995, our perception has changed from expecting most FSCs to reside in the germarium much longer than an FC, to realizing that many FSCs are in fact very short-lived before becoming FCs.
History of FSC numbers
The number of stem cells was first calculated as the reciprocal of the fraction of the FC epithelium covered by a marked FSC clone and was measured at 9-11 days after clone induction to be sure that all transient FC clones had exited the ovariole [2]. Care was taken that the FSC clones examined likely originated from a single marked cell by inducing clones at low frequency with a heat-shock inducible recombination event, but an implicit assumption was made that FSCs divide asymmetrically (to produce one stem cell and one non-stem cell at each division) and that there would therefore be one marked FSC throughout the history of generating marked FCs. At the time, this was a common assumption for stem cells, inspired in part by studies of ovarian Germline Stem Cells. We found that FSCs do not repeatedly undergo asymmetric divisions but rather exhibit frequent amplification or loss of individual lineages (see later). An accurate count of FSCs deduced from FC contributions cannot therefore be made at 9 days after clone induction because by this time a single surviving clone will include an unknown number of FSCs. Earlier results therefore reflect the FC contribution of multiple FSCs, leading to a large underestimate of the total number of FSCs.
In 2001, Zhang and Kalderon used 3 genetic lineage labels and detected up to 3 different FSC lineage colors in a single ovariole 8 days after clone induction, concluding that there were likely three FSCs [3]. Identification of an FSC lineage was constrained by the requirement to identify an FC-contributing, surviving FSC in a location roughly consistent with the two FSC positions mapped by Margolis and Spradling; any marked FSCs that were lost prior to 8d, that occupied an unexpected position or did not contribute any FCs over the last five days would not have been scored. In 2007 Nystul and Spradling also used 3-genotype labeling and postulated 2 fixed niches of FSCs on opposite sides of the germarium, stating that the germarium is bilaterally symmetric and lies down on the slide in a particular orientation such that the two FSC niches are on either side of the germarium [4]. They did sometimes see three FC genotypes in an ovariole, but postulated that an FSC daughter could temporarily occupy one of the niches and act as an FSC.
In our more recent experiments we used the same strategies as before (counting the contribution of a single FSC lineage to the FC epithelium or counting the number of distinctly colored FSC lineages that can be generated in a single epithelium) but concluded that there are many more FSCs. The biggest reason for the very different outcome was the consideration that some FSC lineages might be lost very quickly while others amplify, necessitating looking at outcomes at a variety of times after marking. Other important differences included using more colors for lineage marking; consideration of more potential locations for FSCs and the possibility that a marked FSC may only occasionally give rise to an FC; and thorough analysis of all cells in a set of ovarioles selected without bias. We developed a lineage tracing method that yields 6 different combinations of GFP, RFP and β-galactosidase. Our strategy was to induce as many recombinations as possible in dividing stem cells in order to see the maximum number of FSC lineages. Using the enduring definition of a surviving marked FSC (a cell that remains in the 2a/b region and has given rise to at least one FC patch in the same ovariole), we found up to six (the maximum we could score) surviving FSC lineages in a single ovariole and inferred from the number of colors seen in a large sample of ovarioles that there were about five surviving FSC lineages on average.
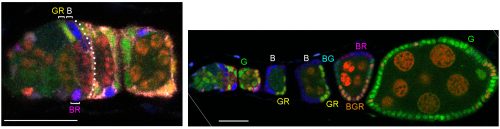
We also found that a single FSC lineage contributed to about 15% of the FC epithelium in a highly mosaic fashion with zero contribution to several egg chambers. Most important, counting surviving FSC lineages at different times after marking showed that many more lineages were present at 5d, while there were decreasing numbers at later times. We estimated that there may be about ten surviving FSC lineages at 5d, but we would need more than six color combinations to count more accurately (five or six colors were present in almost all ovarioles). Consequently, our extrapolation that there may have originally been as many as sixteen FSC lineages at day zero is also just an approximation. In these experiments we did not change the basic methodology or logic of deducing FSC numbers: every one of the cells we called an FSC remained in the germarium for longer than an FC can and had produced at least one FC, defining it unequivocally as an FSC.
Even after taking stem cell loss and amplification into account, it turns out that there is a further limitation on what was counted as a functional FSC because we required proof that an FSC produced an FC. It turns out that some of the roughly sixteen FSCs in a germarium, all of which we believe to have the potential to produce FCs, do not produce any FCs in the 4-5 day time span displayed on a fixed ovariole. This stems in essence from the apparently stochastic behavior of FSCs and positional heterogeneity within the FSC population.
FSCs are radially distributed in layers
In order to identify positions of FSCs we had to examine only those FSC clones with a single candidate stem cell (a single FSC of a certain color that matched an FC patch). This approach allowed us to define multiple locations for an FSC anywhere around the circumference of the germarium and in any of three anterior-posterior (A/P) layers adjacent to the Fasciclin 3-expressing FCs.
One of the challenges we faced as we started to realize that there were many more FSCs than expected was how to score them in sufficient detail. Eventually we developed spreadsheets to record, for each germarium and ovariole, every cell in the 2a/b region by A/P position, and the proportion of each egg chamber occupied by each color. This level of detail was essential to see where FSCs are located and to measure contributions to the FC epithelium. Tabulating the total number of cells in the 2a/b region where FSCs could be found, “the FSC region,” (16 cells on average) provided a second means to deduce the total number of FSCs. The frequency of clones with a single FSC in a given layer was very similar to the distribution of all somatic cells in the FSC region (about 8/16 in layer 1, 6/16 in layer 2 and 2/16 in layer 3, on average), suggesting that all of the roughly 16 cells are FSCs.
At the same time that we started scoring FSCs around the entire circumference, we were surprised to find by live imaging that cells in the FSC region do not stay in fixed locations but undergo back-and-forth radial movements. In addition, the ring of cells around the circumference is indistinguishable when examined by many Gal4 expression patterns and markers [5]. The existence of apparently identical interchangeable cells around the circumference of the germarium at a given A/P position suggests that each ring of FSCs constitutes a pool of equivalent cells.
Population asymmetry
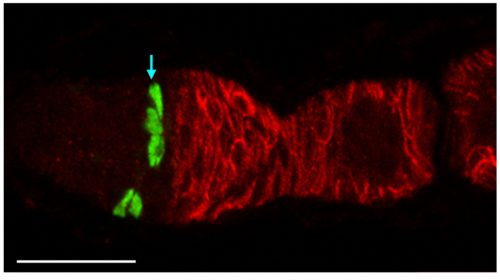
Fundamental to revising our ideas about FSC numbers and behavior is the realization that they are governed by population asymmetry. Population asymmetry is generally understood to mean that the daughters of a stem cell do not necessarily have different fates (one stem and one non-stem cell). Instead, within a homogeneous stem cell population of fixed size, individual stem cells undergo neutral competition during which a stem cell is frequently and stochastically duplicated or lost. Short-lived stem cells are not pre-determined or different from long-lived stem cells—they are selected by chance. If you look at the starting population you can say that only a small subset of those cells will survive as lineages for long time periods but you don’t know which ones. Hence, all of the cells are equivalent and have the potential to survive for a long time and are appropriately given the same name—stem cells. One would have to look very early after marking stem cells to capture them all before any are lost, which is often not practical. Counting the lineages after an arbitrary delay yields the number of stem cell lineages that survived for that time, and eventually only a single lineage will remain. Population asymmetry is by now a well-recognized arrangement that applies to many stem cells, including mammalian gut stem cells[6, 7]. In the mouse small intestine there are around 16 stem cells per crypt but several weeks after labeling individual cells with multiple colors only 1-3 lineages remain in most crypts[7]. We have exactly analogous findings. In both cases researchers first looked long after stem cell marking and, accordingly, thought there were a smaller number of stem cells. The appreciation of the dynamics of population asymmetry resolves these apparent contradictions in both cases.
Our data counting colored patches in ovarioles with no corresponding FSC at 9d clearly showed that many FSC lineages are lost rapidly. By 30 days the majority of ovarioles are monoclonal (we found an average of 1.5 lineages out of 60 ovarioles scored). There is a corresponding increase in the number of FSCs per lineage over time. This demonstration of rapid FSC lineage loss along with a corresponding increase in the number of 2a/b cells per surviving lineage over time shows that FSCs are maintained by population asymmetry.
Origin of Escort Cells—why had it been difficult to see the relationship to FSCs?
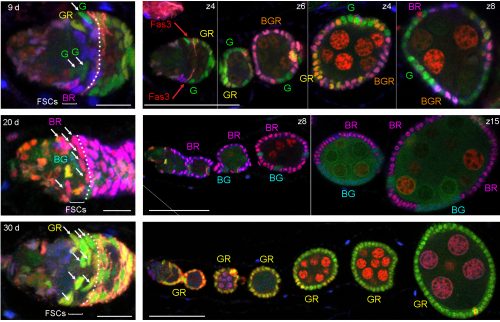
ECs are also labeled in lineage experiments in adults, showing that they are renewed in adult ovaries. However, Margolis and Spradling concluded that there is no lineage relationship between FSCs and ECs [2]. Kirilly et al. concurred but found that the source of new ECs is region 2a/b, very close to FSCs, postulating that the dividing cells are themselves ECs [8]. In these and other lineage studies, including ours, one could readily see ovarioles with marked FCs but no ECs, or marked ECs but no FCs, but there were also many ovarioles containing marked ECs and FCs (as well as FSCs). It was challenging to unravel the basis of these different outcomes.To prove that an FSC can give rise to both ECs and FCs it is essential to prove that a marked lineage containing all three cell types derived from a single cell. That is normally accomplished by inducing marked cells at very low frequency. We found it difficult to label single FSCs using the MARCM technique even by using the mildest of heat-shocks. It is understandable that it is quite difficult to target a single FSC for recombination by chance, given that there are about 16, rather than 2-3 per germarium. We eventually accomplished a satisfactorily low clone induction by additionally taking advantage of multicolor labeling to produce a very low frequency of clones with a specific color combination.
We also became convinced that all adult-born ECs derived from FSCs because the locations of cells in region 2a/b that proliferate, according to EdU labeling, is the same as FSC locations deduced from our multicolor labeling; it was previously thought that most of these cells were ECs. We can now categorize an EC not only by location and morphology but also by the unifying property of exhibiting no proliferation. However, it was puzzling that many lineages contained only ECs or only FCs if all of their progenitors can give rise to both ECs and FCs. We resolved this paradox by a combination of careful analysis of FSC lineages over time and the appreciation that the FSC population is heterogeneous. FSCs can produce only FCs for a few days in succession but a longer time-course shows that virtually all FC-producing lineages eventually produce ECs. Conversely, FSCs can produce only ECs for a few days; ovarioles only reveal FC production over the past 4-5 days, so a constant fraction of FSCs (around 20%) are not associated with marked FCs even though FC production may have been in their past or future. The explanation for periods of producing only ECs or only FCs lies in positional heterogeneity of FSCs.
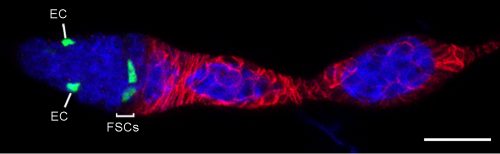
Heterogeneity of FSCs
We found graded cell division rates and graded marker expression across FSC layers. Most important, we found that recent FC production was tightly correlated with FSC location in the most posterior layer (layer 1), leading us to postulate that FCs come directly only from layer 1 FSCs. The process of associating irreversibly with a germline cyst passing through the FSC region has never been visualized, so there was no clear prior indication of where an FC is first “specified.” ECs are anterior to FSCs, so it is almost inevitable that they must derive directly from anterior FSCs, and we also observed movement of anterior FSCs to the EC region by live imaging. Thus, the direct precursors of FCs and ECs are in different FSC layers and as long as an FSC lineage is confined to a single layer it will produce only ECs or only FCs, as often observed at short times after FSC marking. At later times, most surviving FSC lineages amplify and occupy more than one layer, followed by production of both ECs and FCs. Those FSC lineages that are then lost will leave a residue of only, generally long-lived, ECs. Movement of FSCs between layers was also inferred more directly by finding the derivatives of single marked cells in more than one layer at a reasonably high frequency even within 3 days.
The heterogeneity of FSCs presents a complication in identifying FSCs by markers because many proteins that are highly expressed in Escort cells are graded in expression across the FSC domain. For example, more anterior FSCs express much higher levels of the Wnt reporter Frizzled3-RFP and the enhancer trap PZ1444 than posterior FSCs. Other markers, such as Castor, are roughly evenly expressed across FSC layers but are also expressed in FCs. We found that only Fax-GFP stood out as being enriched in FSCs compared with ECs or FCs.
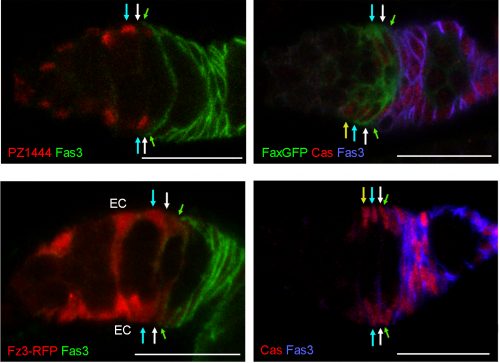
Wnt signaling
We had for many years been puzzled as to why FSCs were lost at an enhanced rate when Wnt pathway activity was either eliminated or increased in FSCs. For Hedgehog and JAK-STAT pathways the results were simpler; more activity caused better survival in competition with wild-type neighbors. With our new understanding of FSC organization and behavior we could look more carefully at the consequences of manipulating Wnt pathway activity, and what we found was straightforward and striking. When Wnt pathway activity is eliminated, FSCs of that lineage become heavily biased towards occupying the most posterior FSC layer and produce FCs, with virtually no EC production. On the other hand, excess Wnt signaling forces FSCs at first into the more anterior FSC layers and eventually to become ECs. These results showed that the strength of Wnt signaling influences the A/P location and fates of FSCs.
Similarity to mammalian epithelial stem cells
With the updated organization of FSCs uncovered, we were amazed to find how similar FSC organization is to mammalian intestinal stem cells. Both FSCs and intestinal stem cells give rise to a constant supply of polarized epithelial cells. Similarities include the number of cells per niche, the organization of stem cells in heterogeneous layers, and use of Wnt as a critical niche factor[9]. Mammalian gut stem cells can produce transit amplifying (TA) or quiescent secretory cells, including Paneth cells, which eventually are retained in the crypt while TA cells migrate away. The initial location of stem cell derivatives that lead to these different outcomes are not clearly defined. Hence the spatial cues for differences in signaling pathway activities (Notch and Wnt) involved in deciding outcomes are not clear. FSCs appear to present a slightly different and simpler paradigm that may also inspire enquiry into an analogous organizational principle for gut stem cells. Wnt signaling is graded across the A/P axis of the FSC community. The relative levels of Wnt signaling dictate whether FSCs adopt more posterior positions and yield predominantly FCs (low Wnt pathway activity) or adopt more anterior positions and become ECs (high Wnt pathway activity), in accord with the natural gradient of Wnt pathway activity in this region. The paradigm of different direct differentiation outcomes for a stem cell population depending on stem cell location might apply to mammalian stem cell communities as well.
Amy Reilein and Daniel Kalderon
Department of Biological Sciences, Columbia University, New York, NY
References
- Reilein, A., et al., Alternative direct stem cell derivatives defined by stem cell location and graded Wnt signalling. Nat Cell Biol, 2017. 19(5): p. 433-444.
- Margolis, J. and A. Spradling, Identification and behavior of epithelial stem cells in the Drosophila ovary. Development, 1995. 121(11): p. 3797-807.
- Zhang, Y. and D. Kalderon, Hedgehog acts as a somatic stem cell factor in the Drosophila ovary. Nature, 2001. 410(6828): p. 599-604.
- Nystul, T. and A. Spradling, An epithelial niche in the Drosophila ovary undergoes long-range stem cell replacement. Cell Stem Cell, 2007. 1(3): p. 277-85.
- Hartman, T.R., et al., Novel tools for genetic manipulation of follicle stem cells in the Drosophila ovary reveal an integrin-dependent transition from quiescence to proliferation. Genetics, 2015. 199(4): p. 935-57.
- Lopez-Garcia, C., et al., Intestinal stem cell replacement follows a pattern of neutral drift. Science, 2010. 330(6005): p. 822-5.
- Snippert, H.J., et al., Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell, 2010. 143(1): p. 134-44.
- Kirilly, D., S. Wang, and T. Xie, Self-maintained escort cells form a germline stem cell differentiation niche. Development, 2011. 138(23): p. 5087-97.
- Beumer, J. and H. Clevers, Regulation and plasticity of intestinal stem cells during homeostasis and regeneration. Development, 2016. 143(20): p. 3639-3649.


 (3 votes)
(3 votes)