A regeneration retrospective: hands-on hard graft
Posted by Alex Eve, on 7 September 2023
This post is part of the regeneration retrospective series.
Yesterday, we went through a couple of examples of amphibian tail regeneration. Limb regeneration is another example of epimorphic regeneration, which is expertly implemented by some amphibians. Unlike the tail, the limb is patterned in three dimensions: anterior-posterior, ventral-distal and media-proximal to produce the proper number and position of the digits, making the patterning process more complex (McQueen and Towers, 2020). But the potential to – one day – regenerate lost human limbs makes limb regeneration a tantalising subject for researchers and science-fiction writers alike (Cox et al., 2019).
On Regeneration after the Amputation of Abnormal Structures: II. Supernumerary Induced Limbs
D. R. Newth
https://doi.org/10.1242/dev.6.3.384
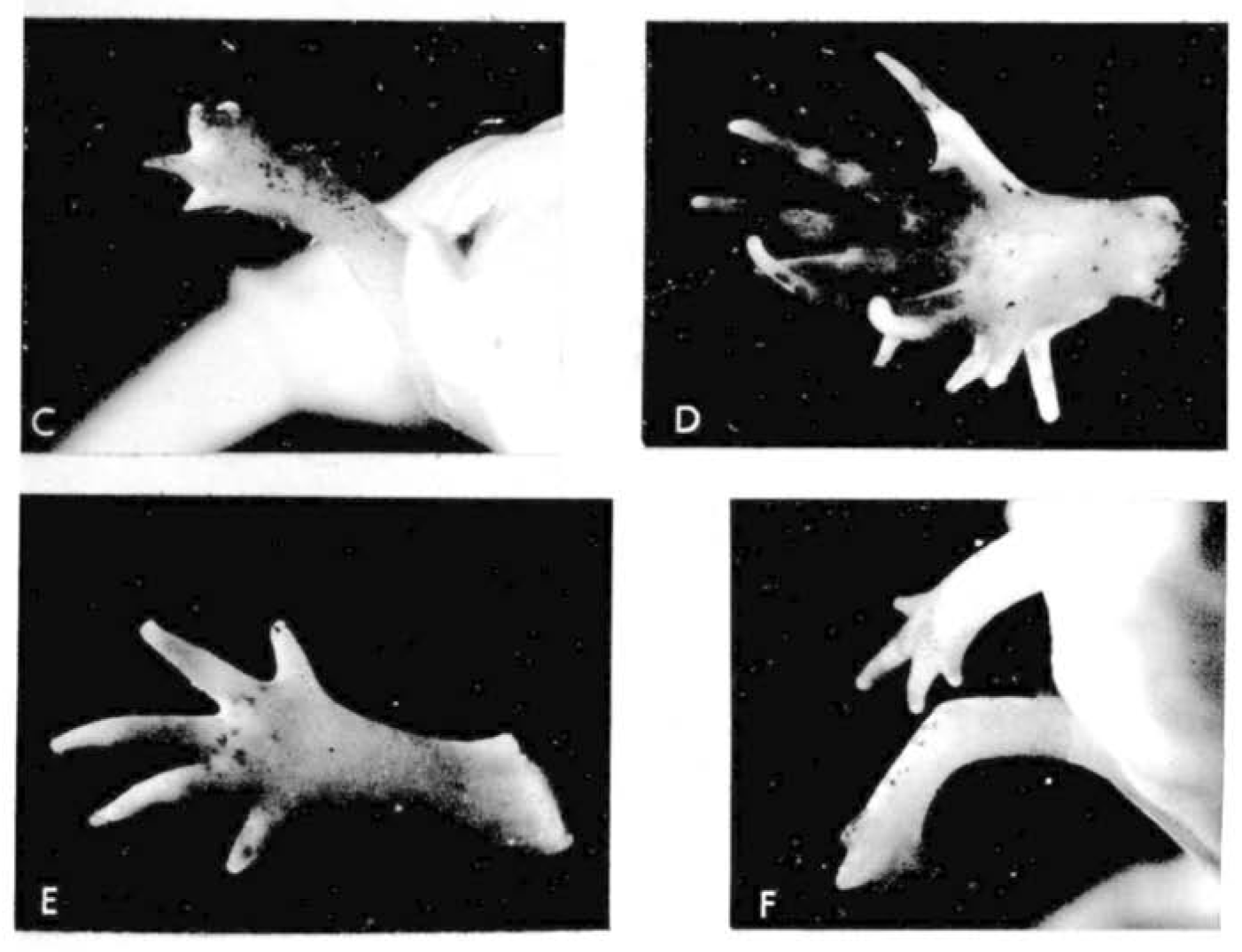
Let us return to the work of our old friend Dr D. R. Newth. In the previous post, we learnt how regeneration could produce a tail more perfect than the original; there’s a similar theme in today’s flashback. A few months after the 1958 publication on tails, Newth posted the second half of his work in JEEM/Development on Triturus helveticus newt limbs (Newth, 1958). There seemed to be an abundance at the time of biologists researching the regenerative abilities of ectopic (supernumerary) limbs by grafting bits of stump tissue, as well as other bits. Such transplants usually produced abnormal limbs with too few or too many digits, which were occasionally resolved during regeneration but there was a lot of conflicting evidence bouncing between journals at the time. One peculiar observation from the past literature is that grafting otic vesicles into the flank induced ectopic limb growth. Newth took a similar approach, engrafting nasal placodes into the body wall to produce an extra foreleg – if anyone has any idea about the mechanism behind placode-dependent limb outgrowth, please do let me know in the comments! This induction approach was quite inefficient (only 19/163 newts grew ectopic limbs) and, like previous reports, these limbs were abnormal with defects ranging from digit-less stumps to polydactyly (Fig. 1). Amputating these defective limbs, however, caused the regeneration of more ‘normal’ looking limbs, with a second amputation of the regenerated limb further improving the phenotype. For example, an ectopic limb that developed with 12 digits, regenerated eight digits following the first amputation and four digits (the normal number) following the amputation of the regenerated limb (Fig. 1). Newth owed these successful regeneration experiments to the amount of limb material, arguing that grafting an inducing centre to form a whole limb was a better approach than grafting stump tissue and provided more material for precise amputation. Again, the position of amputation was deemed significant, but the fascinating mechanism behind this phenomenon remained a mystery.
The limb development and regeneration field has really flourished in the past 65 years and is impossible to adequately summarise here. Evidence, largely from chick and mice, has revealed the many genetic, epigenetic and molecular regulators that control the position of limb budding, outgrowth and digit patterning (McQueen and Towers, 2019). The field has moved in all directions, investigating the roles of the microenvironment, immune system and progenitor populations, as well as their differentiation and lineages, using genome editing, high-resolution imaging and next-generation sequencing technologies. Grafting remains an extremely useful experimental approach (e.g. Vierira et al., 2023) with salamanders, and the axolotl in particular, the main model systems for limb regeneration experiments (Joven et al., 2019). Like in the 1950s, cross-species comparisons are crucial for understanding how tissue regeneration evolved, the factors that control it, and for identifying approaches to induce regeneration in non-regenerative species.
Newt Hoxa13 has an essential and predominant role in digit formation during development and regeneration
Takashi Takeuchi, Haruka Matsubara, Fumina Minamitani, Yukio Satoh, Sayo Tozawa, Tomoki Moriyama, Kohei Maruyama, Ken-ichi T. Suzuki, Shuji Shigenobu, Takeshi Inoue, Koji Tamura, Kiyokazu Agata, Toshinori Hayashi
https://doi.org/10.1242/dev.200282
Takeuchi and colleagues published their paper investigating the role of Hox genes during new limb development regeneration last year (Takeuchi et al., 2022; Fig. 2). Their work follows studies in mammals, which have shown that 5′ Hox genes (e.g. Hox13) are crucial for limb development, but whether this requirement is conserved in other tetrapods has not been clear. Takeuchi and team show that Hox13 genes Hoxa13, Hoxc13 and Hoxd13, are expressed both in developing limbs and in blastemas of the newt Pleurodeles waltl. Using CRISPR/Cas9 to knockout Hoxa13, they reveal that Hoxa13 is required for digit formation with the mutants having fewer digits than wild type. Hoxc13 and Hoxd13, however, seem to be dispensable but overexpression of Hoxd13 in Hoxa13 knockout newts partially rescues limb development; Hoxd13 expression in limb buds is strongly dependent on Hoxa13a. Lastly, the authors show that, following the amputation of the forelimbs, Hox13 genes, and Hoxa13 in particular, are also required for digit formation during limb regeneration.

Both of the articles discussed today concern digit formation in regenerating newt limbs. D.R. Newth showed that regeneration can rescue proper digit development in abnormal limbs and, given that Hox genes have specific positional expression patterns, the work from Takeuchi and colleagues suggests that Hox13 genes might be one of the factors involved in this process. Furthermore, Takeuchi and colleagues demonstrate another parallel, but not identical, link between development and regeneration. Tomorrow, in the final post of this mini-series, we look towards the nervous system.
References
Ben D. Cox, Maximina H. Yun, Kenneth D. Poss; Can laboratory model systems instruct human limb regeneration? Development 15 October 2019; 146 (20): dev181016. doi: https://doi.org/10.1242/dev.181016
Alberto Joven, Ahmed Elewa, András Simon; Model systems for regeneration: salamanders. Development 15 July 2019; 146 (14): dev167700. doi: https://doi.org/10.1242/dev.167700
Caitlin McQueen, Matthew Towers; Establishing the pattern of the vertebrate limb. Development 1 September 2020; 147 (17): dev177956. doi: https://doi.org/10.1242/dev.177956
D. R. Newth; On Regeneration after the Amputation of Abnormal Structures: II. Supernumerary Induced Limbs. Development 1 September 1958; 6 (3): 384–392. doi: https://doi.org/10.1242/dev.6.3.384
Takashi Takeuchi, Haruka Matsubara, Fumina Minamitani, Yukio Satoh, Sayo Tozawa, Tomoki Moriyama, Kohei Maruyama, Ken-ichi T. Suzuki, Shuji Shigenobu, Takeshi Inoue, Koji Tamura, Kiyokazu Agata, Toshinori Hayashi; Newt Hoxa13 has an essential and predominant role in digit formation during development and regeneration. Development 1 March 2022; 149 (5): dev200282. doi: https://doi.org/10.1242/dev.200282
W.A. Vieira, M. Raymond, K. Kelley, M. A. Cherubino, H. Sahin, C. D. McCusker; Integration failure of regenerated limb tissue is associated with incongruencies in positional information in the Mexican axolotl. Front. Cell Dev. Biol. 2023; 11:1152510. doi: https://doi.org/10.3389/fcell.2023.1152510


 (No Ratings Yet)
(No Ratings Yet)