Alan Turing’s patterning system can explain the arrangement of shark scales
Posted by rorylcooper, on 7 November 2018

Understanding how complex biological patterns arise is a long standing and fascinating area of scientific research. The patterning, or spatial arrangement, of vertebrate skin appendages (such as feathers, hair and scales) has enabled diverse adaptations, allowing animals to both survive and thrive in varied and challenging environments. Such adaptations include temperature control of mammalian hair1 and drag reduction of shark scales2. In our recent paper published in Science Advances, we examine how such patterning is regulated throughout development of the shark3.
Alan Turing was a remarkably influential scientist, renowned for his pivotal role as a code breaker during the Second World War. He is also considered the forefather of the modern computer. In 1952, two years before his tragic death following prosecution under the homophobic law of ‘Gross Indecency’, Turing wrote a mathematical model describing how interactions between diffusing chemicals known as morphogens can produce patterns4,5. These morphogens include a short-range, self-promoting activator which also promotes a long-range inhibitor. Turing demonstrated that when his model is appropriately tuned, stable patterns can arise.
Since its publication, there has been a surge of theoretical and experimental research discussing Turing’s model. In fact, his seminal paper has now been cited almost 12000 times4, and experimental work has suggested that this system controls the patterning of both mouse hair and bird feathers6,7. These animals – which belong to a group of vertebrates known as tetrapods – are both classic model species for studying developmental biology. However, until recently, the role of Turing’s system in the skin appendage patterning of vertebrates that arose prior to tetrapods has been poorly understood.

Sharks belong to a group of vertebrates known as cartilaginous fishes, which branched from most other jawed vertebrates before tetrapods diversified. They possess scale-like skin appendages known as denticles, which have been observed in the fossil record as long as 450 million years ago8. Denticles are made from dentine and enamel-like materials, similar to our own teeth. In our recent study, we demonstrate that Turing’s patterning system can explain the arrangement of denticles in an emerging model shark3 – the small-spotted catshark (Scyliorhinus canicula).
We first used computer modelling based on Turing’s equations to demonstrate that denticle patterning is consistent with his system. Next, we examined genes that act as morphogens during the Turing patterning of feathers, and revealed their expression is conserved throughout shark denticle patterning. This includes sonic hedgehog (shh) and fibroblast growth factors (FGFs) as activators, and bone morphogenetic proteins (BMPs) as inhibitors7. By inhibiting FGF signalling, we showed there is likely functional evolutionary conservation of these genes, meaning they appear to play the same roles in both feather and denticle development.
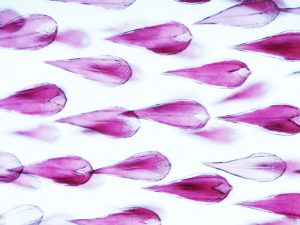
Having found this evidence for Turing-like patterning of shark denticles, we went on to show that altering the parameters of the model can produce patterns with different densities of coverage. These patterns were reflective of arrangements observed in species of sharks and rays that are alive today, demonstrating that simple alterations to Turing’s model can explain the diversity of patterns observed in nature. Therefore, we provide a potential mechanism for how important adaptations, such as hydrodynamic drag reduction and defensive armour, have arisen in sharks. It is likely that Turing’s model is of widespread importance throughout the skin organ patterning of diverse vertebrate groups, all the way from sharks to mammals6,7.
The pattern of shark denticles is one important factor in achieving drag reduction. Another is the shape of individual denticles, which vary both within and between different shark species. Our next research goal is to examine the developmental mechanisms underlying the diversity of denticle shape. Together, this research will provide us with an insight into how important functional traits have arisen in the shark.

Shark-inspired materials have already been created, with the aim of improving the efficiency of travel9. Potential applications include aeroplanes, boats and cars, which are all subject to drag, that is, resistance to the forward motion of an object. Understanding how both denticle patterning and shape contribute towards reducing drag may help to improve the effectiveness of these materials, leading to reduced energy consumption in a wide range of industries. This is an ever-important challenge in a time of depleting resources and climate crisis.
References
- Ruxton, G. D. & Wilkinson, D. M. Avoidance of overheating and selection for both hair loss and bipedality in hominins. Proc. Natl. Acad. Sci. 108, 20965–20969 (2011).
- Dean, B. & Bhushan, B. Shark-skin surfaces for fluid-drag reduction in turbulent flow: a review. Philos. Trans. A. Math. Phys. Eng. Sci. 368, 4775–806 (2010).
- Cooper, R. L. et al. An ancient Turing-like patterning mechanism regulates skin denticle development in sharks. Sci. Adv. 4, 11, eaau5484 (2018).
- Turing, A. M. The chemical basis of morphogenesis. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 237, 37–72 (1952).
- Kondo, S. & Miura, T. Reaction-Diffusion Model as a Framework of Understanding Biological Pattern Formation. Science 329, 1616–1620 (2010).
- Sick, S., Reinker, S., Timmer, J. & Schlake, T. WNT and DKK Determine Hair Follicle Spacing Through a Reaction-Diffusion Mechanism. Science 314, 1447–1450 (2006).
- Jung, H. et al. Local Inhibitory Action of BMPs and Their Relationships with Activators in Feather Formation: Implications for Periodic Patterning. Dev. Biol. 196, 11–23 (1998).
- Sansom, I. J., Smith, M. M. & Smith, P. Scales of thelodont and shark-like fishes from the Ordovician of Colorado. Nature 379, 628–630 (1996).
- Domel, A. G. et al. Shark skin-inspired designs that improve aerodynamic performance. J. R. Soc. Interface 15, (2018).


 (5 votes)
(5 votes)