In Development this week (Volume 137, Issue 16)
Posted by Seema Grewal, on 27 July 2010
Here are the highlights from this week’s issue of Development:
Lymphatic networks follow arterial lead
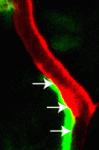 The vertebrate lymphatic system consists of lymphatic vessels, which collect fluid from the tissues and return it to the blood, and lymph nodes, which are involved in immune defence. Lymphatic vessels, like blood vessels, form a complex vascular network, but what guides the development of this network? According to Stefan Schulte-Merker and co-workers, arteries provide crucial guidance cues to the lymphatic endothelial cells (LECs) that form the lymphatic network in the zebrafish trunk (see p. 2653). Using transgenic zebrafish lines that allow the direct visualization of arteries, veins and lymphatic vessels in single embryos, the researchers show first that intersegmental lymphatic vessels (ISLVs) in the zebrafish trunk align with arterial intersegmental vessels (aISVs) but not with venous intersegmental vessels. Then, using time-lapse confocal imaging, they show that LECs migrate exclusively along aISVs and that LEC migration is blocked in zebrafish mutants that lack aISVs. Together, these data reveal a crucial role for arteries in LEC guidance; future research will unravel the mechanism underlying this guidance.
The vertebrate lymphatic system consists of lymphatic vessels, which collect fluid from the tissues and return it to the blood, and lymph nodes, which are involved in immune defence. Lymphatic vessels, like blood vessels, form a complex vascular network, but what guides the development of this network? According to Stefan Schulte-Merker and co-workers, arteries provide crucial guidance cues to the lymphatic endothelial cells (LECs) that form the lymphatic network in the zebrafish trunk (see p. 2653). Using transgenic zebrafish lines that allow the direct visualization of arteries, veins and lymphatic vessels in single embryos, the researchers show first that intersegmental lymphatic vessels (ISLVs) in the zebrafish trunk align with arterial intersegmental vessels (aISVs) but not with venous intersegmental vessels. Then, using time-lapse confocal imaging, they show that LECs migrate exclusively along aISVs and that LEC migration is blocked in zebrafish mutants that lack aISVs. Together, these data reveal a crucial role for arteries in LEC guidance; future research will unravel the mechanism underlying this guidance.
Flowering plant fertilization model over turned
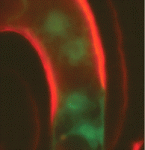 Fertilization in flowering plants involves two sperm cells and two female gametes – the egg cell and the central cell, progenitors of the embryo and endosperm, respectively. A previous study suggested that Arabidopsis plants carrying loss-of-function mutations in cyclin dependent kinase A1 (CDKA:1) make a single sperm cell that preferentially fertilizes the egg cell to produce an embryo that triggers central cell division. Now, Frédéric Berger and colleagues overturn these widely accepted results by showing that a significant proportion of cdka;1 pollen actually delivers two sperm cells (see p. 2683). Delivery of a single cdka;1 sperm cell to a wild-type ovule can fertilize either female gamete, they report. However, when two cdka;1 sperm cells are delivered, one fertilizes the egg cell and the other activates central cell division. Fusion of the gamete nuclei fails in the central cell, however, which prevents paternal genome incorporation and causes seed abortion. Thus, this new analysis of the cdka;1 phenotype reveals an essential role for the paternal genome during early seed development.
Fertilization in flowering plants involves two sperm cells and two female gametes – the egg cell and the central cell, progenitors of the embryo and endosperm, respectively. A previous study suggested that Arabidopsis plants carrying loss-of-function mutations in cyclin dependent kinase A1 (CDKA:1) make a single sperm cell that preferentially fertilizes the egg cell to produce an embryo that triggers central cell division. Now, Frédéric Berger and colleagues overturn these widely accepted results by showing that a significant proportion of cdka;1 pollen actually delivers two sperm cells (see p. 2683). Delivery of a single cdka;1 sperm cell to a wild-type ovule can fertilize either female gamete, they report. However, when two cdka;1 sperm cells are delivered, one fertilizes the egg cell and the other activates central cell division. Fusion of the gamete nuclei fails in the central cell, however, which prevents paternal genome incorporation and causes seed abortion. Thus, this new analysis of the cdka;1 phenotype reveals an essential role for the paternal genome during early seed development.
Med(12)iating Wnt signalling in mouse development
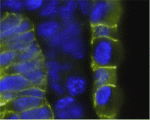 Mediator, a conserved multiprotein complex that connects DNA-bound transcription factors to the RNA polymerase II machinery, is part of the intricate mechanism that regulates eukaryotic transcription. The Med12 mediator subunit is required for gene-specific functions during zebrafish development, but are its developmental functions conserved in mammals? On p. 2723, Heinrich Schrewe and colleagues address this question by examining embryos generated from mouse embryonic stem (ES) cells in which Med12 has been targeted. Embryos generated from Med12 hypomorphic ES cells fail to develop beyond embryonic day 10, the researchers report, and have severe defects in neural tube closure, axis elongation, somitogenesis and heart formation. The Wnt/planar cell polarity pathway and canonical Wnt/β-catenin signalling are both disrupted in the Med12 hypomorphic embryos, they note. Furthermore, embryos generated from Med12 null ES cells fail to establish the anterior visceral endoderm, activate brachyury expression or complete gastrulation. Together, these results indicate that Med12 is necessary for gene-specific functions and for correct Wnt/β-catenin and Wnt/PCP signalling during early mouse development.
Mediator, a conserved multiprotein complex that connects DNA-bound transcription factors to the RNA polymerase II machinery, is part of the intricate mechanism that regulates eukaryotic transcription. The Med12 mediator subunit is required for gene-specific functions during zebrafish development, but are its developmental functions conserved in mammals? On p. 2723, Heinrich Schrewe and colleagues address this question by examining embryos generated from mouse embryonic stem (ES) cells in which Med12 has been targeted. Embryos generated from Med12 hypomorphic ES cells fail to develop beyond embryonic day 10, the researchers report, and have severe defects in neural tube closure, axis elongation, somitogenesis and heart formation. The Wnt/planar cell polarity pathway and canonical Wnt/β-catenin signalling are both disrupted in the Med12 hypomorphic embryos, they note. Furthermore, embryos generated from Med12 null ES cells fail to establish the anterior visceral endoderm, activate brachyury expression or complete gastrulation. Together, these results indicate that Med12 is necessary for gene-specific functions and for correct Wnt/β-catenin and Wnt/PCP signalling during early mouse development.
Mechanics of morphogenesis revealed
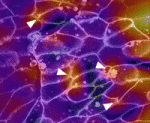 The morphogenetic movements that shape embryos depend on the forces generated by embryo’s cells and on the resistance of its tissues to these forces. Microtubules and F-actin are largely responsible for both these cellular properties but the contribution of these structural elements to morphogenesis is unclear. Now, Lance Davidson and colleagues unexpectedly report that nocodazole-induced depolymerization of microtubules stiffens the converging and extending dorsal tissues in Xenopus embryos (see p. 2785). The researchers attribute this result to the release of Xlfc – a guanine exchange factor that binds to microtubules and that regulates actomyosin contractility by activating Rho family GTPases. Consistent with this idea, drugs that reduce actomyosin contractility rescue nocodazole-induced embryonic stiffening and partly rescue the morphogenetic defects of stiffened embryos. Other experiments that combine drug treatments and Xlfc activation and knockdown indicate that microtubules have no direct role in maintaining bulk tissue stiffness in Xenopus embryos. The researchers conclude, therefore, that microtubules indirectly regulate the mechanical properties of embryonic tissues through RhoGTPase pathways.
The morphogenetic movements that shape embryos depend on the forces generated by embryo’s cells and on the resistance of its tissues to these forces. Microtubules and F-actin are largely responsible for both these cellular properties but the contribution of these structural elements to morphogenesis is unclear. Now, Lance Davidson and colleagues unexpectedly report that nocodazole-induced depolymerization of microtubules stiffens the converging and extending dorsal tissues in Xenopus embryos (see p. 2785). The researchers attribute this result to the release of Xlfc – a guanine exchange factor that binds to microtubules and that regulates actomyosin contractility by activating Rho family GTPases. Consistent with this idea, drugs that reduce actomyosin contractility rescue nocodazole-induced embryonic stiffening and partly rescue the morphogenetic defects of stiffened embryos. Other experiments that combine drug treatments and Xlfc activation and knockdown indicate that microtubules have no direct role in maintaining bulk tissue stiffness in Xenopus embryos. The researchers conclude, therefore, that microtubules indirectly regulate the mechanical properties of embryonic tissues through RhoGTPase pathways.
Bicoid gradient: precision through hunchback
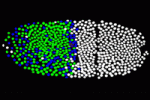 Morphogenetic gradients determine cell identity during development through the concentration dependent activation of target genes, but how the precision of the response to morphogens is determined is unclear. On p. 2798, Nathalie Dostatni and co-workers provide new insights into this developmental puzzle by examining the transcriptional response to Bicoid in Drosophila embryos. The Bicoid gradient is established in Drosophila embryos after eight nuclear divisions (cycle 9) and target protein expression is specified by cycle 14 with a precision that corresponds to a 10% Bicoid concentration difference. To understand how this precision is achieved, the researchers analyze nascent transcripts of the Bicoid target gene hunchback. They report that hunchback is already transcribed from both alleles in most anterior nuclei in cycle 11 interphasic embryos. This synchronous expression is specified within a 10% difference of Bicoid, a precision that is compatible with the fast mobility of Bicoid in the nucleus measured using fluorescent correlation spectroscopy. Finally and importantly, genetic experiments reveal that maternal Hunchback contributes to the early synchrony of the Bicoid response.
Morphogenetic gradients determine cell identity during development through the concentration dependent activation of target genes, but how the precision of the response to morphogens is determined is unclear. On p. 2798, Nathalie Dostatni and co-workers provide new insights into this developmental puzzle by examining the transcriptional response to Bicoid in Drosophila embryos. The Bicoid gradient is established in Drosophila embryos after eight nuclear divisions (cycle 9) and target protein expression is specified by cycle 14 with a precision that corresponds to a 10% Bicoid concentration difference. To understand how this precision is achieved, the researchers analyze nascent transcripts of the Bicoid target gene hunchback. They report that hunchback is already transcribed from both alleles in most anterior nuclei in cycle 11 interphasic embryos. This synchronous expression is specified within a 10% difference of Bicoid, a precision that is compatible with the fast mobility of Bicoid in the nucleus measured using fluorescent correlation spectroscopy. Finally and importantly, genetic experiments reveal that maternal Hunchback contributes to the early synchrony of the Bicoid response.


 (No Ratings Yet)
(No Ratings Yet)