An “ordinary” first day at the BSCB/BSDB Joint Spring Meeting 2013
Posted by Andrei Luchici, on 9 April 2013
Before I even start to tell you anything about this year’s BSCB/BSDB Joint Spring Meeting, I would like to apologise to you if you expect this post to be a detailed description of the science presented there. As tempting as that was, I forced myself no to do it because I wanted to give you a feeling of the general atmosphere of the conference and about the people that were there. Having said this, please get comfortable in your seats and put your seat belts on because we are about to prove Einstein wrong and travel back in time!
It was 8.30am and after spending a late night at the postgraduate social event, I found myself in the Arts centre of Warwick University. From the moment I first entered the building I could not restrain myself from noticing that the air was filled with the smell of freshly ground coffee. For some unknown reason this smell gave me a sudden urge to go and buy a cup of the energising liquor. As I was standing there, I looked around and I realised, to my surprise, that the place was already swarming with people finding their way to the first session of the conference. After getting some coffee, I dragged my half asleep feet and joined another queue… this time it was the queue to the lecture theatre where the Epithelia and Mechanosensing session was about to begin.
After a brief introduction to the conference beautifully delivered by Dr Jim Smith, it was time for the hard-core science to begin. The first round of talks was opened by Dr Barry Thompson who’s talk helped me (an engineer working in biology) understand how well regulated is the epithelial polarity. Using Drosophila as a model organism and a combination of genetics, molecular biology and computer modelling techniques, he discovered that apicobasal polarity requires a combination of positive feedback among apical determinants and mutual antagonism between apical and basal determinants. Wow! It must have been Barry’s presentation skills that made this last sentence have perfect sense at that time because as I write this I realise that things are much more complicated!
Following from this really successful interdisciplinary effort, Dr Pierre-François Lenne changed the tone of the session by presenting how the interaction between actomyosin network and adhesion complexes creates mechanical forces during tissue morphogenesis. Again using Drosophila as a model organism, Pierre employed confocal microscopy to directly measure cortical forces during embryo elongation and to investigate how adhesion complexes, especially E-cadherin, are capable to generate and respond to mechanical cues.
The last talk of this morning session tried to address how could synthetic biology and biomimetics help answer questions that are relevant for in-vivo systems. Jenny Gallop has managed to successfully recreate filopodia-like structures in vitro using only artificial membranes and frog egg extracts to recapitulate the actin signalling pathway required to produce these structures. After having to take in so much new information, it was now the time to go and recharge our batteries for the next sessions. As people started to form another seemingly interminable queue for coffee, I could see and hear that everywhere around me people were still discussing about the science presented in the talks we have just heard.
During this break you could see how the swarming I saw in the morning came to a rest. Everyone was still puzzled about how cells can generate, transmit and understand mechanical forces, so we were hanging around in groups discussing about mechanotrasduction and its roles inside a living organism. As it always happens when you engage in a passionate conversation, during that morning too, time passed so fast that we barely realised that the break was over and it was now time to head back towards the lecture theatres for the second part of the morning session.
Dr Shigenobu Yonemura opened the second part of this session. His talk was a perfect fit for what I kept asking during the break: how are different proteins sensing force? In the short time he had available, he managed to give me an answer to my question using alpha-catenin and adherens junctions as a practical example.
Changing scales, it was Daniel Grimes’ turn to further illustrate the roles of forces in development. His work managed to show how essentially a 1D structure, cilia, are capable of creating 3D left-right asymmetries during mice embryo development. Following the same trend, Katja Roper works on tubulogenesis of the Drosophila salivary glands revealed how an anisotropy in plasma membrane distribution of a protein, Crumbs, determines the subcellular localisation of a supracellular actomyosin cable in the cells at the pacode border.
There could not have been a better way to end this session than Dr Guillaume Charras’ talk on how physical sciences, especially nanotechnology, are capable of offering new tools and techniques to analyse biological systems. His brief survey of microfluidics and atomic-force microscopy combined with practical applications of each technique succeeded to point out how engineering and biology could work together to advance our understanding of the differences between animate and inanimate matter.
During lunch, the swarming I saw as I was waiting in the queue to buy myself a cup of coffee in the morning reappeared, only now people were buzzing around posters. This provided a good opportunity for everyone to mix and meet other people. After a while the swarming calmed down as it was now time to head to the second session of the day on Motors and Morphogenesis.
As I expected, this session was mind-blowing; all speakers presented cutting edge research at the interface of cell biology, developmental biology and engineering. Out of a very impressive pool of speakers, there was one that clearly set the tone of each part of the session. In the first part, I was very impressed by Dr Darren Gilmour’s talk on how zebra fish lateral line primordium (a migrating epithelium) is capable to generate its own local gradient of chemokine to power its collective migration.
Exactly when I thought that I finally got an idea of how cells integrate biochemical pathways and mechanical cues, Dr Jennifer A. Zallen brought back one question on mechanotransduction that I tried to answer for myself, but failed every single time: how local changes in cell architecture can generate long range effects in a tissue? Using Drosophila embryos as a model organism it was possible for her to show that asymmetries in contraction and adhesion and a mechanical feedback loop are all the ingredients needed to reorganise the cells during body axis elongation. I was impressed by the simplicity of this mechanism that cells use to transmit forces at a distance compared to its complex role during development.
As this was not exciting enough, the afternoon session offered me another pleasant surprise as Prof Kate Nobes and Mr John Robert Davis presented two different roles for contact inhibition of locomotion. First Prof Nobes showed how invasive cancer cells change their biochemistry and fail to contact inhibit in vitro. In a completely different system, Drosophila hemocytes (macrophages), John clearly demonstrated that contact inhibition of locomotion is the major driver of the even hemocyte dispersal during embryo development in vivo. These seemingly different talks made me think how magnificent biology is… once cells find an efficient mechanism for performing a function they stick to it and even transfer it from one cell type to another or from one species to another with virtually no major modifications. This gives a completely different meaning to cross-scales communication and for an engineer this is simply mind-blowing!!! I know this starts to become somehow philosophical, but I couldn’t help myself to wonder about it while I was sitting in the back of that half lit lecture theatre and listened to so many examples of this communication across scales.
Unfortunately it all finished too fast, at least that’s how I felt it, because the next thing I remember it is how I was finding my way to the cafeteria to have dinner with a lively group of people which I already knew or have just met there. We were all exchanging our opinions about the science of that day and then suddenly began to discuss a SciFi topic… how we could engineer artificial cells to perform any task we want them to? This continued for a while until everyone decided that it was about time to go and have the first pint of the night and then to head for the memorial lectures.
After this we were all a little tired, and most of us just wanted to call it a day, but we had no idea what surprise was waiting for us as we waited for the Waddington Medal talk: Sir John Gurdon has kindly agreed to come to the meeting and he was sitting there mysteriously waiting to see who has won this years medal. If that was not enough, organisers had another ace down their sleeve… Jim Smith won the Waddington Medal for his fruitful career as a developmental biologist and gave a very inspiring and entertaining talk about his science and his life! Through his stories he managed to convey how much science has changed over the past three decades… from the days when any result was a good result (it did not have to be only a positive one to be considered valuable science) and when it sometimes took as long as six days to get published in a highly respected journal, to the present when it could take up to a few years to get one publication(and that cannot be a negative result!).
This last talk crowned the day, but I think it also did much more… Jim managed to wrap up a very informative first day of the conference and inspired the next generation of cell and developmental biologists to pursue their passion despite any hardship that comes in their way!


 (3 votes)
(3 votes)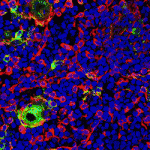 The thymus is the primary organ responsible for generating T cells. Although thymus development has been studied in mice, little is known about how the human thymus develops. Here (
The thymus is the primary organ responsible for generating T cells. Although thymus development has been studied in mice, little is known about how the human thymus develops. Here (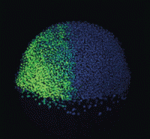 During embryogenesis, the anterior-posterior (AP) and dorsal-ventral (DV) axes are specified by the activity of key signalling pathways. FGF, Wnt and retinoic acid together pattern the AP axis: high activity defines more posterior tissues, which are specified later in development than anterior tissues. The BMP pathway specifies ventral fate; low BMP activity defines dorsal. Whether and how these pathways intersect to coordinate patterning of the two axes is poorly understood. On
During embryogenesis, the anterior-posterior (AP) and dorsal-ventral (DV) axes are specified by the activity of key signalling pathways. FGF, Wnt and retinoic acid together pattern the AP axis: high activity defines more posterior tissues, which are specified later in development than anterior tissues. The BMP pathway specifies ventral fate; low BMP activity defines dorsal. Whether and how these pathways intersect to coordinate patterning of the two axes is poorly understood. On 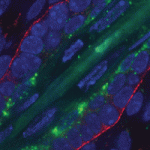 MicroRNAs are important for the regulation of gene expression in a vast array of processes. In the skin, miR-203 has been shown to be crucial for the proper differentiation of the interfollicular progenitor cells, although the specific mechanism of this has remained elusive. In this issue (
MicroRNAs are important for the regulation of gene expression in a vast array of processes. In the skin, miR-203 has been shown to be crucial for the proper differentiation of the interfollicular progenitor cells, although the specific mechanism of this has remained elusive. In this issue ( The correct establishment of adaxial-abaxial patterning is crucial for leaf expansion and growth. The AUXIN RESPONSE FACTOR (ARF) family of proteins are key determinants of organ symmetry and abaxial patterning in Arabidopsis thaliana and are subject to complex regulatory control at both the transcriptional and translational level. Here (
The correct establishment of adaxial-abaxial patterning is crucial for leaf expansion and growth. The AUXIN RESPONSE FACTOR (ARF) family of proteins are key determinants of organ symmetry and abaxial patterning in Arabidopsis thaliana and are subject to complex regulatory control at both the transcriptional and translational level. Here ( In the C. elegans embryo, anterior-posterior polarity is defined at the one-cell stage, via asymmetric and reciprocal localisation of cortex-associated PAR protein complexes: PAR-3, PAR-6 and aPKC localise to the anterior, whereas PAR-1, PAR-2 and LGL-1 are enriched at the posterior. Polarity maintenance involves mutual antagonism between the anterior and posterior complexes and may also involve CDC-42-dependent regulation of myosin activity. Kenneth Kemphues and co-workers (
In the C. elegans embryo, anterior-posterior polarity is defined at the one-cell stage, via asymmetric and reciprocal localisation of cortex-associated PAR protein complexes: PAR-3, PAR-6 and aPKC localise to the anterior, whereas PAR-1, PAR-2 and LGL-1 are enriched at the posterior. Polarity maintenance involves mutual antagonism between the anterior and posterior complexes and may also involve CDC-42-dependent regulation of myosin activity. Kenneth Kemphues and co-workers (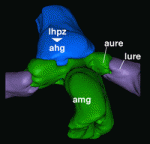 Many animal tissues maintain populations of slowly proliferating stem cells that contribute to tissue homeostasis and repair. In Drosophila, for example, stem cells reside throughout the midgut and within the hindgut and renal tubules. But how and when do these cells arise? Volker Hartenstein and colleagues now show that Drosophila gut progenitors migrate across tissue boundaries and adopt the fate of the organ in which they come to reside (
Many animal tissues maintain populations of slowly proliferating stem cells that contribute to tissue homeostasis and repair. In Drosophila, for example, stem cells reside throughout the midgut and within the hindgut and renal tubules. But how and when do these cells arise? Volker Hartenstein and colleagues now show that Drosophila gut progenitors migrate across tissue boundaries and adopt the fate of the organ in which they come to reside ( Orientation of the cell division axis is essential for both symmetric cell divisions and for the asymmetric distribution of fate determinants during, for example, stem cell divisions. Lu and Johnston review both the well-established spindle orientation pathways and recently identified regulators to provide a updated view of how positioning of the mitotic spindle occurs. See the
Orientation of the cell division axis is essential for both symmetric cell divisions and for the asymmetric distribution of fate determinants during, for example, stem cell divisions. Lu and Johnston review both the well-established spindle orientation pathways and recently identified regulators to provide a updated view of how positioning of the mitotic spindle occurs. See the 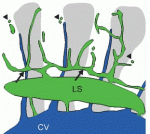 New insights into lymphatic vascular development have recently been achieved thanks to the use of alternative model systems, new molecular tools, novel imaging technologies and a growing interest in the role of lymphatic vessels in human disorders. Here. Hogan and colleagues review the most recent advances in lymphatic vascular development, with a major focus on mouse and zebrafish model systems. See the
New insights into lymphatic vascular development have recently been achieved thanks to the use of alternative model systems, new molecular tools, novel imaging technologies and a growing interest in the role of lymphatic vessels in human disorders. Here. Hogan and colleagues review the most recent advances in lymphatic vascular development, with a major focus on mouse and zebrafish model systems. See the  (No Ratings Yet)
(No Ratings Yet) Voting for the stem cell image contest
Voting for the stem cell image contest – Over the last couple of years, Kim Cooper (a post-doc in the Tabin lab) has been providing us with updates (see her intro post
– Over the last couple of years, Kim Cooper (a post-doc in the Tabin lab) has been providing us with updates (see her intro post 
 An unproven stem cell therapy has taken centre stage in Italy after patients successfully lobbied the Italian government to allow its use in public hospitals. The highly controversial and untested procedure was created by the privately owned Stamina Foundation, but blocked by the Italian Medicine’s Agency, AIFA. Last week’s decision by the Ministry of Health to override AIFA’s block has horrified Italy’s leading stem cell scientists. In a letter to the Ministry, they describe the decision as providing “a dangerous short circuit between patients’ hopes and lucrative commercial practices” of organisations operating outside the “scientific and moral foundations” of medicine.
An unproven stem cell therapy has taken centre stage in Italy after patients successfully lobbied the Italian government to allow its use in public hospitals. The highly controversial and untested procedure was created by the privately owned Stamina Foundation, but blocked by the Italian Medicine’s Agency, AIFA. Last week’s decision by the Ministry of Health to override AIFA’s block has horrified Italy’s leading stem cell scientists. In a letter to the Ministry, they describe the decision as providing “a dangerous short circuit between patients’ hopes and lucrative commercial practices” of organisations operating outside the “scientific and moral foundations” of medicine.  Our
Our 
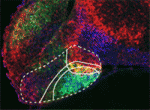 The hypothalamus is a key integrative centre in the vertebrate brain that regulates many essential functions, including homeostasis and stress responses. Several transcription factors that are essential for hypothalamic development have been identified but the production of diverse neuron types in this complex brain region is poorly understood. Here (
The hypothalamus is a key integrative centre in the vertebrate brain that regulates many essential functions, including homeostasis and stress responses. Several transcription factors that are essential for hypothalamic development have been identified but the production of diverse neuron types in this complex brain region is poorly understood. Here (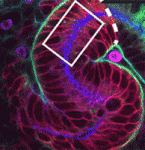 The formation and elongation of polarised epithelial tubules is essential for the structure and function of several metazoan organ systems but the molecular mechanisms that regulate tubulogenesis are largely unknown. Here (
The formation and elongation of polarised epithelial tubules is essential for the structure and function of several metazoan organ systems but the molecular mechanisms that regulate tubulogenesis are largely unknown. Here ( Planar cell polarity depends on the asymmetric localisation of core planar polarity proteins at apicolateral junctions. This asymmetric distribution probably develops through amplification of an initial asymmetry and seems to require the regulation of core protein levels. Now, Helen Strutt, Elizabeth Searle and co-workers (
Planar cell polarity depends on the asymmetric localisation of core planar polarity proteins at apicolateral junctions. This asymmetric distribution probably develops through amplification of an initial asymmetry and seems to require the regulation of core protein levels. Now, Helen Strutt, Elizabeth Searle and co-workers (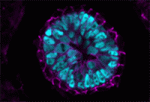 During mammalian embryogenesis, substantial cell proliferation occurs before the establishment of the body plan during gastrulation. Thus, before gastrulation, individual embryonic cells must be pluripotent. In vitro experiments with embryonic stem cells (ESCs) have indicated that the transcription factors Oct4, Sox2 and Nanog are components of a gene regulatory network (GRN) that stimulates self-renewal of pluripotent cells and have identified Tcf7l1 (Tcf3) as an inhibitor of GRN activity. But what is Tcf7l1’s function during embryonic development? To find out, Bradley Merrill and colleagues have been examining embryogenesis in Tcf7l1-/- mouse embryos (see
During mammalian embryogenesis, substantial cell proliferation occurs before the establishment of the body plan during gastrulation. Thus, before gastrulation, individual embryonic cells must be pluripotent. In vitro experiments with embryonic stem cells (ESCs) have indicated that the transcription factors Oct4, Sox2 and Nanog are components of a gene regulatory network (GRN) that stimulates self-renewal of pluripotent cells and have identified Tcf7l1 (Tcf3) as an inhibitor of GRN activity. But what is Tcf7l1’s function during embryonic development? To find out, Bradley Merrill and colleagues have been examining embryogenesis in Tcf7l1-/- mouse embryos (see 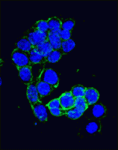 Canonical Wnt signalling and E-cadherin-mediated cell adhesion are both involved in mouse embryonic stem (mES) cell maintenance. β-catenin (Ctnnb1) is central to both these processes – it mediates the transactivation of Wnt target genes and also connects E-cadherin to the actin cytoskeleton via α-catenin. But which β-catenin function is absolutely required for mES cell self-renewal and pluripotency? On
Canonical Wnt signalling and E-cadherin-mediated cell adhesion are both involved in mouse embryonic stem (mES) cell maintenance. β-catenin (Ctnnb1) is central to both these processes – it mediates the transactivation of Wnt target genes and also connects E-cadherin to the actin cytoskeleton via α-catenin. But which β-catenin function is absolutely required for mES cell self-renewal and pluripotency? On 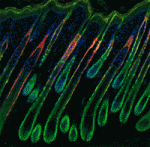 Adult tissue-specific stem cells both self-renew and generate functional progeny. Mammalian hair follicles, which are characterised by cyclical phases of growth (anagen), regression (catagen) and rest (telogen), are an ideal system in which to investigate the homeostasis of an adult stem cell population. On
Adult tissue-specific stem cells both self-renew and generate functional progeny. Mammalian hair follicles, which are characterised by cyclical phases of growth (anagen), regression (catagen) and rest (telogen), are an ideal system in which to investigate the homeostasis of an adult stem cell population. On 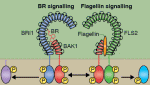 Zhi-Yong Wang and colleagues provide an overview of the highly integrated BR signalling network and explain how this steroid hormone functions as a master regulator of plant growth, development and metabolism.
Zhi-Yong Wang and colleagues provide an overview of the highly integrated BR signalling network and explain how this steroid hormone functions as a master regulator of plant growth, development and metabolism.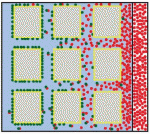 Alex Schier et al analyze various morphogen transport models using the morphogens Nodal, fibroblast growth factor and Decapentaplegic as case studies. They propose that most of the available data support the idea that morphogen gradients form by diffusion that is hindered by tortuosity and binding to extracellular molecules.
Alex Schier et al analyze various morphogen transport models using the morphogens Nodal, fibroblast growth factor and Decapentaplegic as case studies. They propose that most of the available data support the idea that morphogen gradients form by diffusion that is hindered by tortuosity and binding to extracellular molecules.