The people behind the papers: Fernando Ferreira & Min Zhao
Posted by the Node Interviews, on 14 December 2016
Today’s paper comes from the final issue of Development for 2016, and reveals a link between bioelectricity and reactive oxygen species during tail regeneration in Xenopus. We caught up with first author Fernando Ferreira and his advisor Min Zhao, Professor in Dermatology at UC Davis.

So Min, can you give me the brief history of your lab, and what questions you are interested in?
MZ The main goal of my lab is to electrically heal wounds and regenerate tissues. Demonstrated over one and half centuries ago, the minute electric signals naturally produced at wounds are very poorly understood and appreciated. Epithelial cells and many other types of cells follow the guidance of the electric signals and migrate and grow directionally. We demonstrated that in epithelial sheets, the guidance effect of physiological electric signals overrides other co-existing guidance cues.
I had medical training in trauma with Zhengguo Wang, one of the founding fathers of trauma surgery in China. I then had research training with Geoffrey Burnstock at University College London, and Colin McCaig and John Forrester at University of Aberdeen. I started my lab at the University of Aberdeen with a Welcome Trust University Award and held professor/personal chair and honorary consultant positions at University of Aberdeen and Aberdeen Royal Infirmary (Scotland), before moving to University of California at Davis to take up a professorship in the Department of Dermatology and Department of Ophthalmology.
Supported by grants from NIH, NSF, California Institute of Regenerative Medicine and other federal and state agencies, my lab is interested in answering the following questions:
- How do cells sense and respond to physiological electric fields?
- How are the wound electric currents/fields produced and regulated?
- Can we exploit the “electrical signalling” to enhance wound healing and induce regeneration?
And Fernando, how did you end up in Min’s lab?
FF During my Master’s degree, my adviser and Min met in an international conference. In the discussion they mentioned me; on arrival, my adviser told me that Min’s lab would be willing to host me and that could be an opportunity that I probably should not let escape. By this time, I already knew and admired much of Min’s work and thought it could be an excellent opportunity to test some old and new ideas falling within the framework of his lab. Thus, naturally, I readily set the goal to work with him.
Meanwhile, however, I applied for other closer positions, but I was oddly satisfied or left with a bitter-sweet feeling after receiving no or negative responses; I guess that this proved that I really wanted to join Min’s lab. Things aren’t straightforward though, and after a considerable despair for not getting a 1 year long Fulbright grant to visit Min’s lab, I took a risky move and entered a PhD programme in the Portuguese university (Minho) without funding. I was working in non-scientific part-time jobs to cover the tuition fees and, simultaneously, applied for a studentship grant sponsored by the Portuguese Science and Technology Foundation (FCT). With the Fulbright denial lingering in my mind, I had low expectations. Fortunately, I got the grant and after a way too long “quarantine” process (~8 months), I finally joined the lab in the USA…
It is pressing and fair to recognise that after joining the University of Minho, I received precious encouragement and help from the PhD programme professors and especially my supervisor Andreia Gomes, who accepted me without second thought, always supported me and gifted me with wise advice. All the happiness and sorrow of this journey, allied with the sheer size of the Atlantic Ocean plus the departure delay, made the farewell from family particularly hard; it is a natural cliche to be grateful for family care and support, and undoubtedly I am.
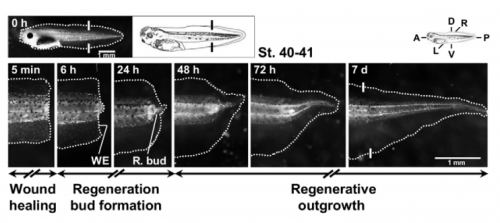
Do you think bioelectricity outside of the nervous system is adequately appreciated in developmental and regenerative biology?
MZ Bioelectricity is not as adequately appreciated in developmental and regenerative biology as I’d wish, which I believe perhaps is due to the following. Research in bioelectricity (not the traditional electrophysiology) was restarted by Lionel Jaffe and his students Richard Borgens, Richard Nuccitelli and Ken Robinson with some wonderful results from the 1960s-1990s. This happened in an era that coincided with the revolutionary discovery of the double helix and the great advances in biology that ensued. Genetic, molecular and biochemical mechanisms are in the lime light of biology, including developmental and regenerative biology. Great advances in technologies and tools in genetics and molecular biology provided developmental and regenerative biologists with powerful tools to understand some of the most fundamental mechanisms. Research technology in bioelectricity, however, has since stayed virtually unchanged. It is also worthwhile to mention that some “charlatan claims” in bioelectricity have tarnished and discredited this field.
“Great advances in technologies and tools in genetics and molecular biology provided developmental and regenerative biologists with powerful tools to understand some of the most fundamental mechanisms. Research technology in bioelectricity, however, has since stayed virtually unchanged”
Bioelectricity is therefore off the radar of most developmental and regenerative biologists. Very few laboratories have continued the efforts in bioelectricity, for example Michael Levin at Tufts University, USA and Colin McCaig at University of Aberdeen, Scotland. Some of their research has provided very impressive results in combination with genetics and molecular biology.
Was anything known about the connection between reactive oxygen species (ROS) and bioelectricity in regeneration before your paper?
MZ & FF Strictly in regeneration, we couldn’t find studies demonstrating a direct connection between redox and bioelectric states. When we started this study we already knew – from decades old evidence – that electric currents were important for a successful regeneration, especially in amphibians; however, no evidence existed about ROS (excluding wound healing in small-scale injuries). Then, a wave of papers came, showing that ROS were also required for regeneration in widespread models, such as Xenopus tadpoles and adult zebrafish. The link between ROS and bioelectricity (membrane potential, transepithelial potential (TEP) and electric currents/fields), remained, however, elusive. In the regeneration field, our study thus appeared in a timely fashion to link the fairly old but re-emerging field of bioelectricity with the emergent field of redox biology.
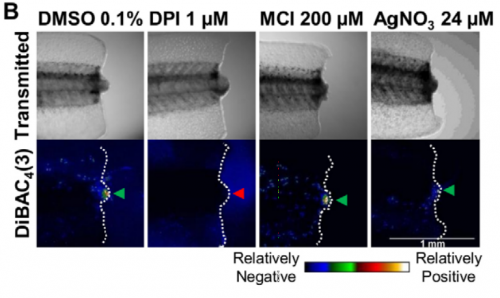
Could you give us the key results of your paper in a paragraph?
MZ & FF The general take home message is that redox and bioelectric activities interact during regeneration. More specifically, there is a two-way regulation of bioelectric activities by NADPH oxidases: the driven electron flow depolarizes the membrane potential, whereas the produced H2O2 increases the magnitude of TEP (positive inside) and switches the direction of electric current (to inward) in the regeneration bud. The depletion of ROS during the regenerative period mimics the abnormally low TEP and non-reversed electric currents measured during the refractory (non-regenerative) period. The external application of H2O2 for a short period normalizes the bioelectric activities and, by doing so, rescues and induces regeneration. External H2O2 was also inductive enough to form ectopic tails in injuries severing the spinal cord during the regenerative period. Finally and molecularly speaking, H2O2 regulates voltage-gated Na+ channels in order to modulate regeneration.
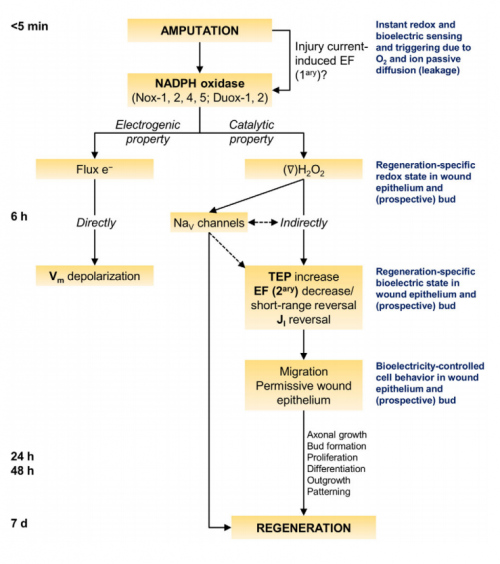
Your paper ends with a model including the proposition that immediately post-amputation, an electrical signal activates the redox signal. How did you come to this hypothesis?
MZ & FF To better understand this hypothesis we need to go back a bit and point out the underlying assumptions. One of the highlights of this study is that ROS are immediately required for regeneration. Those ROS are produced from NADPH oxidases, holoenzymes with complex assembly and regulation; therefore, an ultra-fast signal must activate the enzyme to generate sufficient ROS for the task ahead.
One of the highlights of this study is that ROS are immediately required for regeneration…an ultra-fast signal must activate NADPH oxidases to generate sufficient ROS for the task ahead.
An electric short-circuit is an instantaneous response to amputation, which results in the so-called injury current and subsequent electric field. This immediate and automatic electric field is, according to the hypothesis, what activates the NADPH oxidases. In fact, there is some evidence showing that applied electric fields induce production of ROS in cells in vitro. We are currently designing experiments to test this hypothesis during regeneration. If true, a redox-bioelectric feedback module would exist in regeneration. This is, the injury-induced electric signals activate the upstream redox signals that regulate downstream electric signals. With caution, we think that the now hypothetical feedback module could be used as theoretical evidence, because it could allow a more tight or efficient regulation of regeneration owing to evolution.
Before speculating or designing more experiments to understand how the electric fields would activate the NADPH oxidases, it is more pressing to test whether the hypothesis is true.
How do you think your mechanism might relate to the ‘canonical’ intercellular signalling pathways that are also involved in regeneration?
MZ & FF The integration between ROS and bioelectric activities occurs very early in the regeneration process. Many of the ‘canonical’ signalling pathways, such as Wnt, BMP and Notch, appear to be activated later on. By itself, this may indicate that those pathways are regulated by ROS and/or bioelectricity, i.e., act downstream. In fact, several studies in the regeneration context have shown that ROS or bioelectricity, independently, regulate some signalling pathways (and also cell behaviours), such as Wnt, FGF and Delta.
Given the high penetrance in regeneration, we think that pathways like the ones already mentioned and others might be affected by redox and bioelectric activities; we also think that follow up studies will unveil this, aiming for a higher level of mechanistic integration in regeneration.
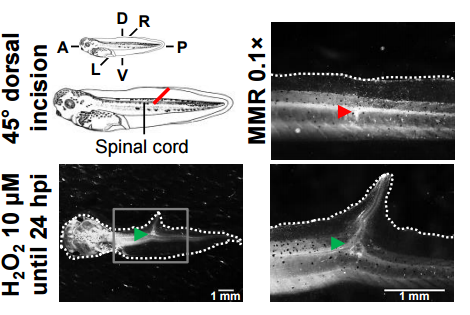
Why do you think the H2O2 treatment induced the formation of ectopic tails?
MZ & FF The induction of ectopic tails was a thrilling finding, but it is important to note that the purpose of the assay was to check whether H2O2 induced ectopic tails and not why; the why deserves further research. That said, studies found that fin wounds in both Xenopus tadpoles and adult zebrafish generate ROS, likely H2O2. Unpublished results from us show that blocking the production of ROS impairs healing in Xenopus fin wounds. Therefore, we think that a threshold of H2O2 could define or tune the morphogenetic outcome, meaning that if the threshold is passed we may get an ectopic tail instead of just healing.
“The induction of ectopic tails was a thrilling finding”
Mechanistically speaking, another study found that Wnt signalling induced ectopic tails in the same model as ours. H2O2 could thus regulate Wnt for the same purpose, a pathway that could be mediated by bioelectric activities. Not mutually exclusive, another, maybe more speculative, possibility is an analogy with the accessory limb and blastema formation in axolotls and Xenopus, respectively. To induce them, it is required extra neuronal tissue, usually a deviated nerve. The incision we made in the tail severed the spinal cord, therefore, H2O2 treatment could, maybe via bioelectricity, affect the neuronal tissue so it becomes inductive.
When doing the research, was there a particularly exciting result or eureka moment that has stayed with you?
FF As many have at one point or another, I had the privilege to experience both life-guiding eureka moments and exciting results. A first eureka-like moment occurred back in my last bachelor year. Anxiously to find what path to follow, I learned about ROS and the caudal regeneration in lizards came to my mind. I had a minor thought experiment: I grabbed a common lizard making it autotomize the tail; with the inner tissues now exposed to the atmosphere, I imagined the oxygen entering the amputation plane down its chemical gradient and then pictured its transformation into ROS; as the levels rose, ROS alarmed local cells that something went wrong and triggered regeneration without any delay. In my innocence at the time, I let this single moment guide me through science ever since; there is no regret!
A second eureka-like moment occurred just before my arrival in the USA. When analysing a review paper, I read that the NADPH oxidases are electrogenic; this triggered a late night chain reaction that led me to the core of this manuscript. As NADPH oxidases work, they transfer electrons through the plasma membrane, so I thought that this was the origin of the membrane depolarization previously shown in regeneration. Then, I thought that since ROS are produced, these would, in turn, affect other facets of the bioelectric phenomena, namely electric currents.
“When analysing a review paper, I read that the NADPH oxidases are electrogenic; this triggered a late night chain reaction that led me to the core of this manuscript”
During the research, the exciting moments were when I was performing the critical experiments testing the redox-bioelectric crosstalk during regeneration. I blocked the production of ROS and imaged the membrane potential and measured the electric currents; during experiments, before getting the positive evidence, I was lightly sweating and my hands and belly were shivering. I know that these “symptoms” are analogous of a romantic encounter, guess that made them even more exciting!
Other unforgettable exciting moment was when, in the very first attempt, I saw a well-defined ectopic tail induced by H2O2. I was so impressed by it that I was childish enough to call other lab members to see an undisclosed “hopeful monster” in the microscope.
And what about the flipside: any particular moments of frustration and despair?
FF Other than now and then when, by procedural vicissitudes – for example, compromised batches of tadpoles (fungal infection, deficient animals, etc.), or all electrodes ending up breaking when touched, or readings were too noisy or strange because the earth wire was somehow disconnected – or for no obvious reason, experiments didn’t work out, I didn’t have any moments of frustration or despair worth noting. If you are surprised, so am I; not having major setbacks by the end of this manuscript surprised me and sometimes even “scared” me, since frustrations are a common theme in the research process and I really don’t want to think that the hypothesis and experimental design put forth were bullet proof. So, for modesty’s sake let’s just call it ‘beginners luck’; probably, the use of well-established techniques and methods helped. Just to highlight the surprise, we are about to submit a new study, where I had my share of despair moments, which I guess covers their absence in this paper, or at least I joke in that way! In fact, I was fortunate enough to get some serendipitous findings which we might end up following.
And finally, Min: where do you think this work will take you next?
MZ We followed an interdisciplinary approach hoping to merge apparently disparate research fields during regeneration. We think that this approach is important and potentially rewarding, and so several lines of research can be pursued with that in mind. We will have at least two exciting possible directions. One is to detail the molecular mechanisms of the redox-bioelectric interplay and to integrate them with ‘canonical’ signalling pathways in the Xenopus and other regeneration models. To help, we are currently establishing a redox and bioelectric sensor facility in the lab. The other is to take advantage of some of the cutting-edge technologies in wound healing and tissue regeneration in mammals. It appears that we are able to manipulate local electric fields and/or redox activities. A combined approach may provide promising therapies for chronic and non-healing wounds.
“I would be tempted to propose a term, “electrobiology”, hoping to suggest that electricity in biology has significant roles”
Evidence is accumulating suggesting bioelectricity as a different layer of mechanism, usually very upstream, together with the fundamental genetics and molecular/cellular processes that orchestrate during development and regeneration. I would be tempted to propose a term, “electrobiology”, hoping to suggest that electricity in biology has significant roles, in contrast to the more than the phenomenological nature of the word “bioelectricity”.
, , , Early bioelectric activities mediate redox-modulated regeneration. Development 143: 4582-4594.
Browse the People Behind the Papers archive here.


 (4 votes)
(4 votes)