The people behind the papers – Agnes Cheong and Jesse Mager
Posted by the Node Interviews, on 21 November 2020
This interview, the 82nd in our series, was published in Development earlier this year.
Nuclear-encoded mitochondrial ribosomal proteins (MRPs) are crucial for mitochondrial function and hence cellular energy production. A new paper in Development investigates the function of MRPs in early mouse development, thanks to a battery of knockout mutants. We caught up with first author (and recent graduate) Agnes Cheong and her supervisor Jesse Mager, Associate Professor at the Department of Veterinary and Animal Sciences at the University of Massachusetts, Amherst, to find out more.

Jesse, can you give us your scientific biography and the questions your lab is trying to answer?
JM I did my PhD work with Terry Magnuson at UNC (University of North Carolina at Chapel Hill), studying the Polycomb Group Gene EED and connecting PRC2 with genomic imprinting, X-inactivation and gastrulation defects. I then did postdoctoral work with Rudolf Jaenisch and Marisa Bartolomei, again investigating epigenetic regulation during early development, which I have continued in my own group since 2008 at UMass. We continue to identify novel parent of origin events during gastrulation, as well as characterizing a large number of early lethal phenotypes arising from the Knockout Mouse Project that advance our understanding of the diverse array of genes required for implantation and gastrulation.
Agnes, how did you come to work in Jesse’s lab and what drives your research today?
AC My first research experience involved the study of mammalian implantation and I was really fascinated by the remarkable transformations the embryos undergo during early development. I wanted to continue exploring my interest in embryogenesis, which was the reason why I decided to stay at Jesse’s lab after my rotation. We know so much more than we did decades ago, yet when we take a magnifying glass to zoom into different developmental processes, many blank spaces remain. The desire to comprehend this convoluted process is my motivation to pursue research in development.
When we take a magnifying glass to zoom into different developmental processes, many blank spaces remain
How are each of you coping in the current COVID-19 pandemic?
AC A workday at home is certainly very different from that at the lab. Since I started working from home, I lost the privilege of casually reaching my 10,000 steps daily. Besides my baking hobby, I am otherwise eager to be able to get back to the lab and do some bench work. Fortunately, the Mager lab still has regular virtual lab meetings so that we can present our research ideas and catch up with our lab family, even though we are not able to be physically present together.
JM Since the shutdown mid-March, the lab is now basically just me and the mice…so a bit lonely. But we’ve had lots of Zoom meetings and are trying to keep the lab members busy writing and reading. It definitely took a few weeks to find productive work patterns (and bonus regular lunches with my kids!). Daily evening strolls with my family have been a silver lining – we are fortunate to be able to get outside quite a bit.
Let’s get to your current paper then, which uses mutants produced by the Knockout Mouse Project. What were the aims of that project and your lab’s involvement with it?
AC & JM IMPC/KOMP is a global initiative, and its main goal is to generate a comprehensive and public resource comprising mouse embryonic stem cells and mice containing a null mutation in every gene within the mouse genome. Although the bulk of the effort has focused on adult phenotypes, ∼25% of nulls are lethal and 12% die prior to E9.5. Our involvement with KOMP is to characterize a large number of these early lethal phenotypes since there is no planned pipeline to analyse early embryos. Thus far, we have phenotyped more than 110 gene knockouts, and will grow to ∼250 over the next few years. Check out all our results at http://blogs.umass.edu/jmager/.
Can you give us the key results of the paper in a paragraph?
AC & JM Our paper demonstrates the importance of a functional oxidative phosphorylation (OXPHOS) system during early development. Although there are over 70 mitochondrial ribosomal protein (Mrp) genes in mammals, the absence of a single Mrp gene results in pre-gastrulation developmental arrest. Despite the presence of pluripotent epiblast cells in the Mrp mutants, these embryos fail to initiate EMT and progress through gastrulation. This disruption is the result of a malfunctioning mitochondrial translation system which contributes to a lack of ATP production, and hence results in cell cycle disruption and stalling at the G2/M checkpoint. Our results are aligned with other developmental studies of mitochondrial functioning genes showing that the OXPHOS is indispensable during early embryogenesis.
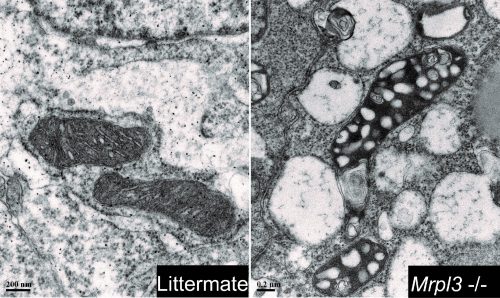
Before developmental arrest, early embryonic development runs normally in Mrp mutants – does this mean the MRPs are doing nothing before then?
AC & JM Not necessarily: since the majority of Mrp genes are also present in the oocyte, maternal Mrp protein could be supporting development through implantation and/or an alternative energy system, such as glycolysis, may be a major factor driving developmental progression prior to gastrulation. A more-detailed study of protein turnover and maternal deletion experiments will provide insights on the precise time point at which Mrp proteins are essential during embryogenesis.
When doing the research, did you have any particular result or eureka moment that has stuck with you?
AC My memorable eureka moment was when I analysed phospho-cdc25c foci and found a significant increase in the mutants, confirming my suspicion that the malfunctioning OXPHOS system leads to cell cycle disruption.
And what about the flipside: any moments of frustration or despair?
The endless sea of genotyping when working with 20+ different knockout lines and needing to genotype every mouse and embryo.
So what next for you after this paper?
AC I recently completed my PhD defence. My plan is to diversify my scientific experience and continue my research career in a biotech or industrial setting.
Where will this work take the Mager lab?
JM Our findings that so many nuclear-encoded mitochondrial functioning genes have such similar phenotypes (gastrulation arrest) are prompting the design of experiments to try to assess the metabolic needs of distinct pre-gastrulation lineages and cell populations. This is a new and exciting area or inquiry for our group. We will also pursue conditional knockout approaches to examine the maternal protein contribution.
Finally, let’s move outside the lab – what do you like to do in your spare time in Amherst?
AC Amherst is a small college town surrounded by many local breweries. When the weather is nice and warm, I enjoy walking to town and reward myself with a hoppy and cold beer. On cold rainy days, I like warming up my apartment with baked goods and sharing them with my friends.
JM I’ll be honest – I’m an exercise enthusiast (to put it mildly)…so if I’m not at work or with my family I’ll generally be found getting those endorphins.


 (No Ratings Yet)
(No Ratings Yet)