Behind the Paper: Support beyond Countries
Posted by Tohru Yano, on 23 September 2012
 When Dr. Eva Amsen, Community Manager for the Node and Online Editor for Development, recommended me to write this post in connection with our recent publication about mechanisms of pectoral fin development in zebrafish, she was anxious to know the continuation of a story in Nature News last year after the earthquake (commented by my supervisor, Professor Koji Tamura, Tohoku University in Japan). We had almost forgotten our inexpressible anguish until Eva’s indication. Though it is true that we experienced a terrible disaster, what did we really suffer from? What kind of message should we give to readers? Although we decided to write this post about the road to publication of our article, I had hesitated to write it for a long time because I did not know from what I should begin to write.
When Dr. Eva Amsen, Community Manager for the Node and Online Editor for Development, recommended me to write this post in connection with our recent publication about mechanisms of pectoral fin development in zebrafish, she was anxious to know the continuation of a story in Nature News last year after the earthquake (commented by my supervisor, Professor Koji Tamura, Tohoku University in Japan). We had almost forgotten our inexpressible anguish until Eva’s indication. Though it is true that we experienced a terrible disaster, what did we really suffer from? What kind of message should we give to readers? Although we decided to write this post about the road to publication of our article, I had hesitated to write it for a long time because I did not know from what I should begin to write.
 Since our laboratory is located on a hill far from the sea, we were not directly affected by the tsunami, but the shock of the earthquake depressed us. Our fish room suffered from loss of power (for three days) and suspension of water supply (for three weeks). In other fish rooms in Tohoku University, many fish lines were lost because of low temperatures or damage caused by the earthquake. Fortunately, our fish room is located on the ground floor with air tightness maintained, I therefore decided to reduce feeding frequency to once every four days (to prevent water pollution) and reduce water circulation (to prevent excessive evaporation). When our water tank for fish became depleted of water after two weeks, we filtered water from a well at a place far from our fish room (the only place to obtain water in the campus) and we carried heavy plastic containers filled with water to the fish room. We were able to keep all of the fish lines alive (I had maintained the fish lines with tender care for six years from when I was an undergraduate student without the assistance of any fish technicians). Our laboratory also had other animals (frogs and reptiles) and most of them could be maintained, though some of them lost their ability to reproduce.
Since our laboratory is located on a hill far from the sea, we were not directly affected by the tsunami, but the shock of the earthquake depressed us. Our fish room suffered from loss of power (for three days) and suspension of water supply (for three weeks). In other fish rooms in Tohoku University, many fish lines were lost because of low temperatures or damage caused by the earthquake. Fortunately, our fish room is located on the ground floor with air tightness maintained, I therefore decided to reduce feeding frequency to once every four days (to prevent water pollution) and reduce water circulation (to prevent excessive evaporation). When our water tank for fish became depleted of water after two weeks, we filtered water from a well at a place far from our fish room (the only place to obtain water in the campus) and we carried heavy plastic containers filled with water to the fish room. We were able to keep all of the fish lines alive (I had maintained the fish lines with tender care for six years from when I was an undergraduate student without the assistance of any fish technicians). Our laboratory also had other animals (frogs and reptiles) and most of them could be maintained, though some of them lost their ability to reproduce.
 In our laboratory rooms (on the fifth floor of a six-story building), everything on the benches and desks fell down and many things were destroyed when the earthquake struck. We returned the fluorescence microscopes and DNA sequence analyzer back to their original positions, but they all fell onto the floor again when a big aftershock occurred. I remember that we were so depressed. The Japanese Government and Tohoku University provided money for repair. Four months later, we were able to restart experiments (though we could not concentrate on work immediately). Because of this chaotic period lasting for four months, submission of our manuscript to Development was delayed, and the PhD thesis defense was affected. Nevertheless, we received support from scientific communities all over the world. I was supposed to attend the JSDB-GFE Joint Meeting of Developmental Biology in Dresden (March 23-26, 2011), but I could not attend it. The German Society of Developmental Biologists supported our registration fee that we had cancelled. ZIRC (Zebrafish International Resource Center, USA) provided fish resources for us in order to allow us to restart experiments smoothly. In Japan, the CDB (Center for Developmental Biology) provided a shuttle bus to transport people and supplies. We received support from many people and organizations, and most of the costs for restoration were provided by public funds including donations from throughout the world. I appreciate this blessed environment, and consequently I could publish my article. However, there are still many people who are in need of support.
In our laboratory rooms (on the fifth floor of a six-story building), everything on the benches and desks fell down and many things were destroyed when the earthquake struck. We returned the fluorescence microscopes and DNA sequence analyzer back to their original positions, but they all fell onto the floor again when a big aftershock occurred. I remember that we were so depressed. The Japanese Government and Tohoku University provided money for repair. Four months later, we were able to restart experiments (though we could not concentrate on work immediately). Because of this chaotic period lasting for four months, submission of our manuscript to Development was delayed, and the PhD thesis defense was affected. Nevertheless, we received support from scientific communities all over the world. I was supposed to attend the JSDB-GFE Joint Meeting of Developmental Biology in Dresden (March 23-26, 2011), but I could not attend it. The German Society of Developmental Biologists supported our registration fee that we had cancelled. ZIRC (Zebrafish International Resource Center, USA) provided fish resources for us in order to allow us to restart experiments smoothly. In Japan, the CDB (Center for Developmental Biology) provided a shuttle bus to transport people and supplies. We received support from many people and organizations, and most of the costs for restoration were provided by public funds including donations from throughout the world. I appreciate this blessed environment, and consequently I could publish my article. However, there are still many people who are in need of support.
Earthquakes occur suddenly with almost no warning. After the earthquake, Koji decided to rearrange throughout the laboratory space and make anti-earthquake reinforcements. Both the number of casualties and the amount of money needed for restoration can be reduced by taking appropriate precautions against future possible disasters. It is also important to share experiences or information among scientists in order to be prepared for future disasters (the Node website is an advanced effort to communicate with developmental biologists/stem cell researchers throughout the world). We cannot thank you enough for your kindness!
We cannot thank you enough for your kindness!
Yano, T., Abe, G., Yokoyama, H., Kawakami, K. & Tamura, K. (2012). Mechanism of pectoral fin outgrowth in zebrafish development, Development, 139 (16) 2925. DOI: 10.1242/dev.075572


 (14 votes)
(14 votes) The fourth annual
The fourth annual  The evening closed with a reception and a visit to the exhibition area, where various companies and organisations have set up a stand for the next few days. There were many vendors and publishers, but also a few institutes: You can find out more about the future Francis Crick Centre in London, or about working at St Jude’s Children’s Hospital in Memphis. The Node is there, too, at the Company of Biologists booth just outside the exhibit hall (next to the job board), so do drop by if you’re attending. I’m there occasionally between talks and I love meeting Node readers.
The evening closed with a reception and a visit to the exhibition area, where various companies and organisations have set up a stand for the next few days. There were many vendors and publishers, but also a few institutes: You can find out more about the future Francis Crick Centre in London, or about working at St Jude’s Children’s Hospital in Memphis. The Node is there, too, at the Company of Biologists booth just outside the exhibit hall (next to the job board), so do drop by if you’re attending. I’m there occasionally between talks and I love meeting Node readers. (1 votes)
(1 votes) (No Ratings Yet)
(No Ratings Yet)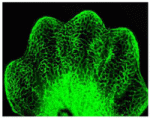 During organogenesis, vascular remodelling ensures that developing organs receive sufficient oxygen and nutrients. Factors that induce sprouting angiogenesis, which is essential for vascular remodelling, have been identified, but the mechanisms that terminate sprouting angiogenesis, thereby stabilising the vasculature, remain unclear. Here (p.
During organogenesis, vascular remodelling ensures that developing organs receive sufficient oxygen and nutrients. Factors that induce sprouting angiogenesis, which is essential for vascular remodelling, have been identified, but the mechanisms that terminate sprouting angiogenesis, thereby stabilising the vasculature, remain unclear. Here (p. 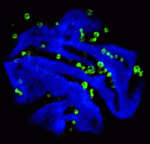 Endopolyploidy (increased cell ploidy) occurs during normal development in many eukaryotes. In higher plants, endopolyploidy is usually the result of endoreduplication – endonuclear DNA replication that produces chromosomes with multivalent chromatids. According to the ‘karyoplasmic ratio’ theory, a cell’s cytoplasmic volume is proportional to its nuclear DNA content. On p.
Endopolyploidy (increased cell ploidy) occurs during normal development in many eukaryotes. In higher plants, endopolyploidy is usually the result of endoreduplication – endonuclear DNA replication that produces chromosomes with multivalent chromatids. According to the ‘karyoplasmic ratio’ theory, a cell’s cytoplasmic volume is proportional to its nuclear DNA content. On p. 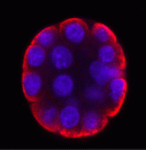 The inside-outside model of cell-fate specification in pre-implantation mammalian embryos proposes that blastomeres on the inside of the 16-cell stage embryo adopt an inner cell mass (ICM) fate, whereas those on the outside adopt a trophectoderm (TE) fate. Cell-cell contact should therefore be a key factor in this cell-fate specification event. On p.
The inside-outside model of cell-fate specification in pre-implantation mammalian embryos proposes that blastomeres on the inside of the 16-cell stage embryo adopt an inner cell mass (ICM) fate, whereas those on the outside adopt a trophectoderm (TE) fate. Cell-cell contact should therefore be a key factor in this cell-fate specification event. On p. 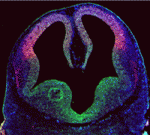 During brain development, neural progenitor cells (NPCs) give rise to various types of neurons and finally differentiate into astrocytes via switches in their differentiation competency. These switches involve changes in gene expression profiles that are thought to be governed partly by epigenetic control mechanisms, such as histone modification. Ryoichiro Kageyama and co-workers now report that the histone H3 Lys9 (H3K9) methyltransferase ESET (also known as Setdb1 or KMT1E) plays an essential role during mouse brain development (see p.
During brain development, neural progenitor cells (NPCs) give rise to various types of neurons and finally differentiate into astrocytes via switches in their differentiation competency. These switches involve changes in gene expression profiles that are thought to be governed partly by epigenetic control mechanisms, such as histone modification. Ryoichiro Kageyama and co-workers now report that the histone H3 Lys9 (H3K9) methyltransferase ESET (also known as Setdb1 or KMT1E) plays an essential role during mouse brain development (see p. 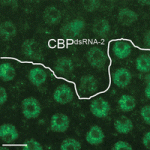 The genomic location and S-phase timing of origins of DNA replication change during multicellular development. Chromatin modifications influence differences in origin location and timing among different cells, but how is DNA replication coordinated with development programmes? Brian Calvi and colleagues have been examining developmental gene amplification in Drosophila ovarian follicle cells (p.
The genomic location and S-phase timing of origins of DNA replication change during multicellular development. Chromatin modifications influence differences in origin location and timing among different cells, but how is DNA replication coordinated with development programmes? Brian Calvi and colleagues have been examining developmental gene amplification in Drosophila ovarian follicle cells (p. 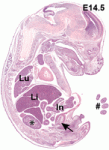 β-Catenin is a central component of adherens junctions, which are required for cell sorting and migration during development, and of canonical Wnt signalling, which controls numerous developmental processes. Mice that lack β-catenin die before gastrulation but mice that express 50% of wild-type levels develop normally. Now, on p.
β-Catenin is a central component of adherens junctions, which are required for cell sorting and migration during development, and of canonical Wnt signalling, which controls numerous developmental processes. Mice that lack β-catenin die before gastrulation but mice that express 50% of wild-type levels develop normally. Now, on p. 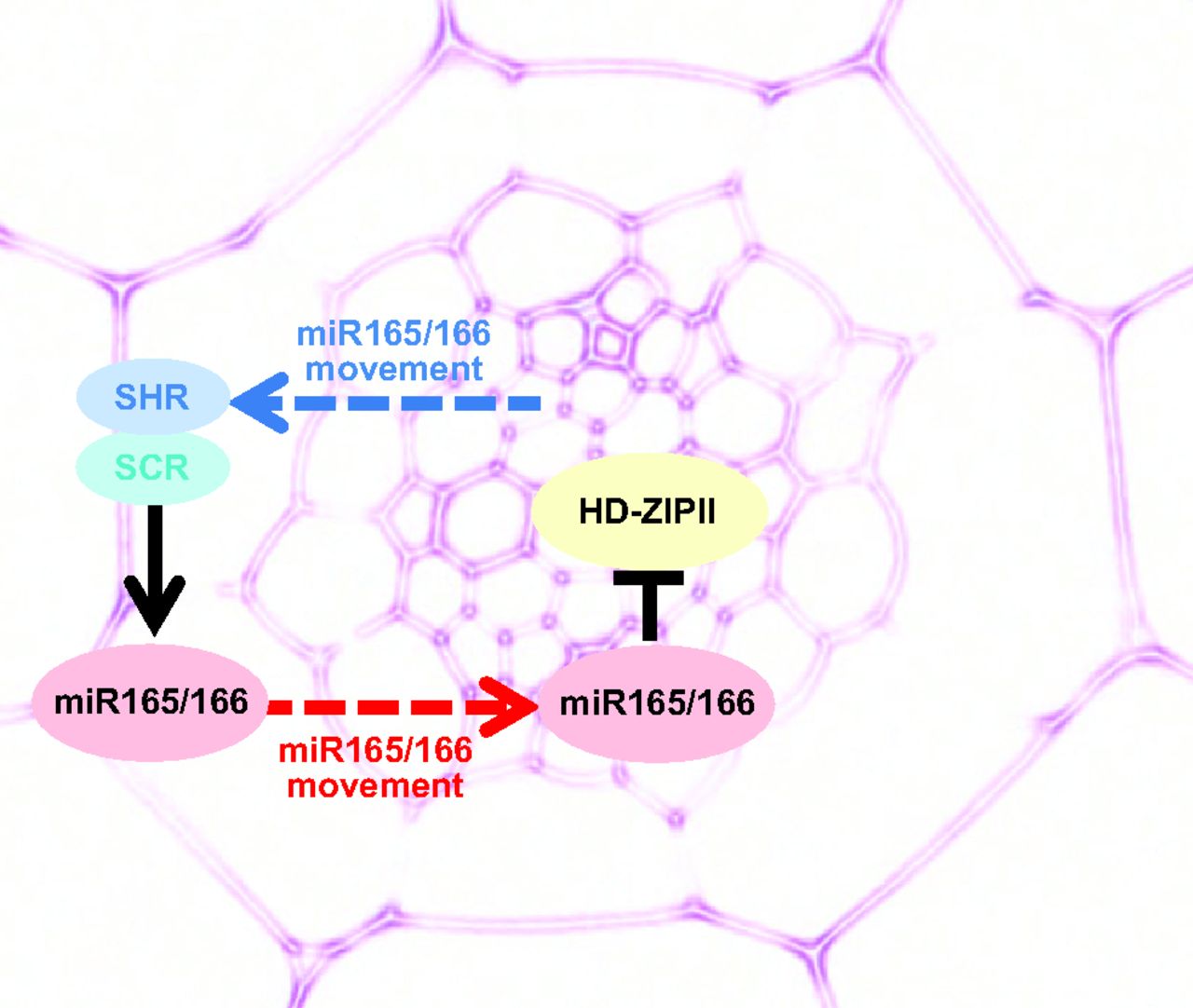 The third EMBO Conference on Plant Molecular Biology, which focused on ‘Plant development and environmental interactions’, was held in May 2012 in Matera, Italy. In this issue, Beeckman and Friml review some of the topics and themes that emerged from the meeting. See the Meeting Review on p.
The third EMBO Conference on Plant Molecular Biology, which focused on ‘Plant development and environmental interactions’, was held in May 2012 in Matera, Italy. In this issue, Beeckman and Friml review some of the topics and themes that emerged from the meeting. See the Meeting Review on p.  As part of our “
As part of our “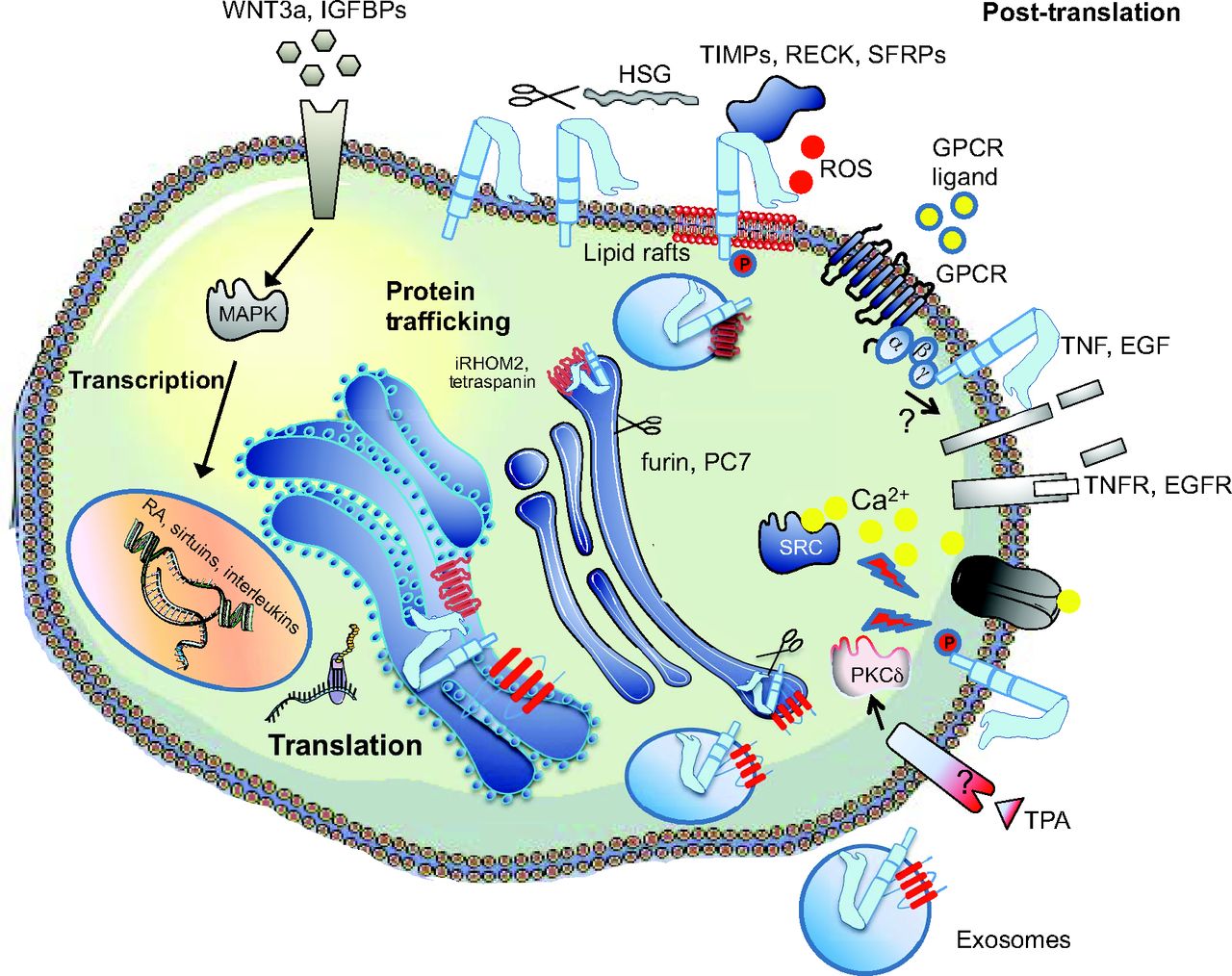 Weber and Saftig summarize the fascinating roles of A Disintegin And Metalloproteinases (ADAMs) in embryonic and adult tissue development in both vertebrates and invertebrates. See the Review on p.
Weber and Saftig summarize the fascinating roles of A Disintegin And Metalloproteinases (ADAMs) in embryonic and adult tissue development in both vertebrates and invertebrates. See the Review on p.