This Spotlight article was written by Stefan Schulte-Merker and Didier Y. R. Stainier, and was first published in Development.
Morpholino oligomers have been used widely and for many years in the zebrafish community to transiently knock down the function of target genes. It has often been difficult, however, to reliably discriminate between specific and non-specific effects, and thus generally accepted guidelines to control for morpholino side effects do not exist. In light of recent methodologies to generate mutant lines in virtually any zebrafish gene, we discuss these different approaches with a specific focus on how the first description of a loss-of-function phenotype in zebrafish should be accomplished.
Initially, the genetic analysis of zebrafish development and physiology was dominated by mutants identified in small- and large-scale forward genetic screens (Chakrabarti et al., 1983; Driever et al., 1996; Haffter et al., 1996).Whereas forward genetics was instrumental in establishing zebrafish as an additional vertebrate model system, progress was hampered by the fact that there was no reliable technology to carry out reverse genetics in this model.TILLING (a reverse genetics approach based on high-throughput sequencing of ENU-mutagenized fish)was introduced only in 2002 (Wienholds et al., 2002) and requires considerable up-front investment in logistics and infrastructure. The introduction of morpholinos (MOs) in frogs (Heasman et al., 2000) and zebrafish (Nasevicius and Ekker, 2000) as an antisense reagent to transiently knock down gene function was therefore greeted with considerable excitement, as it appeared to fill a real void in the toolbox. Since its inception, countless studies using this technology have been published, including some using MOs to knock down maternally deposited transcripts to circumvent the generation of maternal-zygotic mutants, and others using caged MOs, which allow for inducible release of these antisense reagents (Shestopalov et al., 2012).
The MO antisense technology is based on nucleic acid bases that are linked tomorpholine rings and a non-charged phosphorodiamidate backbone. The rationale for this design was that MOs would not bind electrostatically to protein, hence causing less toxicity, while at the same time being resistant to nucleases (Summerton, 2007). MOs are injected into early zebrafish embryos using standard techniques. Commonly, they are ∼25-mers designed to be an exact antisense match against the region surrounding the first translated ATG (to block translation) or against a splice donor or acceptor site (to interfere with precursor mRNA splicing). It quickly became apparent that some MOs could work extremely well, and there are many MO phenotypes that efficiently mimic mutant phenotypes without any noticeable side effects. However, it has also become clear that MOs can lead to artifacts and that for many MOs the phenotypes caused by specific binding to the intended target RNA are difficult to separate from those caused by the non-specific binding to unintended targets (Eisen and Smith, 2008). In fact, a simple calculation suggests that binding to targets other than the intended precursor or mature mRNA is likely. A zebrafish embryo contains ∼500 ng of RNA, 2-5% of which is translatable (25 ng) (A. Giraldez, personal communication; see also Davidson, 1986). Assuming that at any given time there are more than 104 different mRNA species present in a cell (Davidson, 1986), and that those transcripts are equally represented among the 25 ng of mRNA, only 2.5 pg of a specific mRNA species is available for targeting. Injections typically deliver ∼1 ng of MO, often more. Assuming further that the target mRNA has an average length of 1.25 kb, whereas the MO is a 25-mer, this equates to a 2×104-fold molar excess of MO versus target mRNA. It is therefore most likely that this vast excess of MO will bind other RNA or other macromolecules. This situation would not be such a serious problem if there were reliable ways to distinguish specific from nonspecific effects. However, this is not the case, and one can at best only show that MOs affect the target sequence; non-specific effects cannot easily be identified, even when using mRNA rescues (see Del Giacco et al., 2010; Tao et al., 2011). The literature now contains several examples in which developmental delay, defects in organ asymmetry and pericardial edema (among many other ‘phenotypes’) are attributed to knocking down a specific gene, but in which subsequent generation of a mutation in that gene revealed a very different phenotype, and often no phenotype at all. Recent examples include mutations in sox18, nr2f1a and prox1a/b, all genes that had been reported to show morphant phenotypes within the lymphatic vasculature, whereas the mutant alleles do not (van Impel et al., 2014).
In several cases it has been possible to circumvent some of the non-specific phenotypes by suppressing p53 activity (Robu et al., 2007), which can reduce the ectopic cell death caused by nonspecific MO effects; however, this approach has its own caveats as it effectively generates a phenotype not on a wild-type, but on a p53-deficient, background. Applying such drastic corrective measures to allow a phenotypic analysis raises a number of questions that cannot be easily addressed.
Two surprisingly efficient alternatives for reverse genetics have been recently implemented in zebrafish (Chang et al. 2013; Huang et al., 2011; Sander et al., 2011; Hwang et al., 2013; Zu et al., 2013) and other organisms (Beumer et al., 2008; Tesson et al., 2011; Yang et al., 2013). TALE nucleases and the Crispr/Cas9 system are very efficient at generating mutations. As both techniques have been
reviewed extensively (Auer and Del Bene, 2014), we will restrict the discussion here to comparing the principles of the MO and TALEN/ Crispr approaches.
First, it should be noted that – like MOs – the implementation of these new technologies can be carried out in virtually any lab. Whereas TILLING really only makes sense for those willing to analyze large numbers of samples and genes, TALENs and Crispr do not require a substantial investment and, once established, can be used to generate targeting constructs within 1 (Crispr) to 2 (TALEN) weeks.
Second, as TALENs and Crisprs affect genomic DNA, rather than RNA transcripts, their molecular effect can be determined at the single embryo level (which is more difficult with MOs) to obtain a clear phenotype/genotype correlation. Of course, such an approach requires caution, as these nuclease-injected embryos are most likely to be mosaic for the resulting genomic lesions. Furthermore, TALEN and Crispr constructs can sometimes be efficient enough to generate loss-of-function situations in the actual injected embryos (Dahlem et al., 2012), and so, in a minority of cases, can be used almost like an MO; injection, scoring for phenotypes and confirming that the nuclease works efficiently can be performed within a few days.
Third, the published evidence, although currently limited, suggests that the side effects of these nucleases are often negligible (Hruscha et al., 2013), even though both TALE and Crispr-Cas nucleases can bind and cleave off-target loci (Reyon et al., 2012; Fu et al., 2014). When additional mutations are introduced, they can usually be segregated away from the mutation of interest by one or two outcrosses (as with mutations identified in ENU mutagenesis screens). This specificity is of course a tremendous advantage, and very different from MOs: an MO that binds non-specifically will most likely do so in every injected embryo. Lastly, it is relatively easy to generate multiple mutant alleles in one gene (e.g. by using TALEN pairs that affect different regions of the targeted gene), thus further reducing the chance of being misled by off-target mutations.
Hence, it seems fair to say that within the last year or so, the landscape of reverse genetics in zebrafish has changed, and it has changed for the better. Anyone can now, within a few weeks, generate reagents that can be used for reverse genetic experiments that appear to be of superior reliability and that are less burdened with side effects compared with MOs. Does that mean that we should do away with MOs altogether? Not necessarily: as we pointed out in the first paragraph, there are many MOs that are very useful and that appear to work specifically. We know they work specifically because we can compare them with a mutant phenotype. We would argue that this is the criterion that should be used in most cases: if one can show that an MO phenotype is an exact replicate of a mutant phenotype, then use of this MO is certainly acceptable and can save valuable time; for example, for injection into transgenic lines or for generating ‘double mutants’. However, the description of a phenotype that is provided for the first time and that is based solely on MOs without the ability to compare with a genetic mutant, should in the future be viewed very critically. In most cases, there are better alternatives in the form of nuclease based targeted approaches and there is no good reason not to use them.
References
Auer, T. O. and Del Bene, F. (2014). CRISPR/Cas9 and TALEN-mediated knock-in approaches in zebrafish. Methods pii: S1046-2023(14)00129-7 (in press).
Beumer, K. J., Trautman, J. K., Bozas, A., Liu, J.-L., Rutter, J., Gall, J. G. and Carroll, D. (2008). Efficient gene targeting in Drosophila by direct embryo injection with zinc-finger nucleases. Proc. Natl. Acad. Sci. USA 105, 19821-19826.
Chakrabarti, S., Streisinger, G., Singer, F. and Walker, C. (1983). Frequency of gamma-ray induced specific locus and recessive lethal mutations in mature germ cells of the zebrafish, Brachydanio rerio. Genetics 103, 109-123.
Chang, N., Sun, C., Gao, L., Zhu, D., Xu, X., Zhu, X., Xiong, J.-W. and Xi, J. J. (2013). Genome editing with RNA-guided Cas9 nuclease in zebrafish embryos. Cell Res. 23, 465-472.
Dahlem, T. J., Hoshijima, K., Jurynec, M. J., Gunther, D., Starker, C. G., Locke, A. S., Weis, A. M., Voytas, D. F. and Grunwald, D. J. (2012). Simple methods for generating and detecting locus-specific mutations induced with TALENs in the zebrafish genome. PLoS Genet. 8, e1002861. Davidson, E. (1986). Gene Activity in Early Development. Waltham: Academic Press.
Del Giacco, L., Pistocchi, A. and Ghilardi, A. (2010). prox1b Activity is essential in zebrafish lymphangiogenesis. PLoS ONE 5, e13170.
Driever, W., Solnica-Krezel, L., Schier, A. F., Neuhauss, S. C., Malicki, J., Stemple, D. L., Stainier, D. Y., Zwartkruis, F., Abdelilah, S., Rangini, Z. et al. (1996). A genetic screen for mutations affecting embryogenesis in zebrafish. Development 123, 37-46.
Eisen, J. S. and Smith, J. C. (2008). Controlling morpholino experiments: don’t stop making antisense. Development 135, 1735-1743.
Fu, Y., Sander, J. D., Reyon, D., Cascio, V. M. and Joung, J. K. (2014). Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat. Biotechnol. 32, 279-284.
Haffter, P., Granato, M., Brand, M., Mullins, M. C., Hammerschmidt, M., Kane, D. A., Odenthal, J., van Eeden, F. J., Jiang, Y. J., Heisenberg, C. P. et al. (1996).
The identification of genes with unique and essential functions in the development of the zebrafish, Danio rerio. Development 123, 1-36.
Heasman, J., Kofron, M. and Wylie, C. (2000). Beta-catenin signaling activity dissected in the early Xenopus embryo: a novel antisense approach. Dev. Biol. 222, 124-134.
Hruscha, A., Krawitz, P., Rechenberg, A., Heinrich, V., Hecht, J., Haass, C. and Schmid, B. (2013). Efficient CRISPR/Cas9 genome editing with low off-target effects in zebrafish. Development 140, 4982-4987.
Huang, P., Xiao, A., Zhou, M., Zhu, Z., Lin, S. and Zhang, B. (2011). Heritable gene targeting in zebrafish using customized TALENs. Nat. Biotechnol. 29, 699-700.
Hwang, W. Y., Fu, Y., Reyon, D., Maeder, M. L., Tsai, S. Q., Sander, J. D., Peterson, R. T., Yeh, J.-R. J. and Joung, J. K. (2013). Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 31, 227-229.
Nasevicius, A. and Ekker, S. C. (2000). Effective targeted gene ‘knockdown’ in zebrafish. Nat. Genet. 26, 216-220.
Reyon, D., Tsai, S. Q., Khayter, C., Foden, J. A., Sander, J. D. and Joung, J. K. (2012). FLASH assembly of TALENs for high-throughput genome editing. Nat. Biotechnol. 30, 460-465.
Robu, M. E., Larson, J. D., Nasevicius, A., Beiraghi, S., Brenner, C., Farber, S. A. and Ekker, S. C. (2007). p53 activation by knockdown technologies. PLoS Genet. 3, e78.
Sander, J. D., Cade, L., Khayter, C., Reyon, D., Peterson, R. T., Joung, J. K. and Yeh, J.-R. J. (2011). Targeted gene disruption in somatic zebrafish cells using engineered TALENs. Nat. Biotechnol. 29, 697-698.
Shestopalov, I. A., Pitt, C. L. and Chen, J. K. (2012). Spatiotemporal resolution of the Ntla transcriptome in axial mesoderm development. Nat. Chem. Biol. 8, 270-276.
Summerton, J. E. (2007). Morpholino, siRNA, and S-DNA compared: impact of structure and mechanism of action on off-target effects and sequence specificity. Curr. Top. Med. Chem. 7, 651-660.
Tao, S., Witte, M., Bryson-Richardson, R. J., Currie, P. D., Hogan, B. M. and Schulte-Merker, S. (2011). Zebrafish prox1b mutants develop a lymphatic vasculature, and prox1b does not specifically mark lymphatic endothelial cells. PLoS ONE 6, e28934.
Tesson, L., Usal, C., Ménoret, S., Leung, E., Niles, B. J., Remy, S., Santiago, Y., Vincent, A. I., Meng, X., Zhang, L. et al. (2011). Knockout rats generated by embryo microinjection of TALENs. Nat. Biotechnol. 29, 695-696.
van Impel, A., Zhao, Z., Hermkens, D. M. A., Roukens, M. G., Fischer, J. C., Peterson-Maduro, J., Duckers, H., Ober, E. A., Ingham, P. W. and Schulte- Merker, S. (2014). Divergence of zebrafish and mouse lymphatic cell fate specification pathways. Development 141, 1228-1238.
Wienholds, E., Schulte-Merker, S., Walderich, B. and Plasterk, R. H. A. (2002). Target-selected inactivation of the zebrafish rag1 gene. Science 297, 99-102.
Yang, H., Wang, H., Shivalila, C. S., Cheng, A. W., Shi, L. and Jaenisch, R. (2013). One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell 154, 1370-1379.
Zu, Y., Tong, X.,Wang, Z., Liu, D., Pan, R., Li, Z., Hu, Y., Luo, Z., Huang, P., Wu, Q. et al. (2013). TALEN-mediated precise genome modification by homologous recombination in zebrafish. Nat. Methods 10, 329-331
 (2 votes)
(2 votes)
 Loading...
Loading...
 The emergence of haematopoietic stem cells (HSCs) during early mammalian development is crucial for the formation of all blood cell lineages. Previous studies indicate that Runx1 is required for the endothelial-haematopoietic transition that gives rise to definitive HSCs; however, this transition occurs in multiple steps and the precise stage at which Runx1 is required has been unclear. In this issue (p.3319), Alexander Medvinsky and colleagues define the exact point during murine HSC emergence at which Runx1 is required. Using a conditional reversible knockout strategy, the authors show that a deficiency of Runx1 does not affect commitment to the haematopoietic lineage as marked by the expression of CD41, as Runx1 knockout embryos still contain a population of CD41+ cells that can form HSCs when Runx1 expression is restored. However, the absence of Runx1 blocks progression to the next stage of HSC emergence, as marked by the expression of CD45. These results demonstrate a precise, stage-specific role for Runx1 in the molecular regulation of HSC emergence during embryo development.
The emergence of haematopoietic stem cells (HSCs) during early mammalian development is crucial for the formation of all blood cell lineages. Previous studies indicate that Runx1 is required for the endothelial-haematopoietic transition that gives rise to definitive HSCs; however, this transition occurs in multiple steps and the precise stage at which Runx1 is required has been unclear. In this issue (p.3319), Alexander Medvinsky and colleagues define the exact point during murine HSC emergence at which Runx1 is required. Using a conditional reversible knockout strategy, the authors show that a deficiency of Runx1 does not affect commitment to the haematopoietic lineage as marked by the expression of CD41, as Runx1 knockout embryos still contain a population of CD41+ cells that can form HSCs when Runx1 expression is restored. However, the absence of Runx1 blocks progression to the next stage of HSC emergence, as marked by the expression of CD45. These results demonstrate a precise, stage-specific role for Runx1 in the molecular regulation of HSC emergence during embryo development. The formation of the mammalian neocortex relies on the migration of projection neurons into specific layers, which is in turn regulated by cyclin-dependent kinase 5 (Cdk5). Cdk5 has previously been shown to interact with the G protein-coupled receptor serotonin 6 receptor (5-HT6R); however, whether and how this interaction might be important for neocortical migration remains unclear. Now, on p.3370, Alexandre Dayer and colleagues use a range of knockdown and rescue in utero electroporation experiments in mouse to show that 5-HT6R regulates the migration of the upper layer cortical projection neurons via its interaction with Cdk5, rather than with serotonin or other agonists. Knockdown of 5-HT6R in postmitotic upper layer neurons impairs their migration and leads to their presence in deeper layers of the neocortex, while re-expression of the full-length receptor or a Cdk5 kinase rescues this defect. The interaction between Cdk5 and 5-HT6R specifically affects the transition between multipolar and bipolar morphology in immature migrating neurons and provides in vivo evidence for the role of a G protein-coupled receptor in neocortical neuron migration.
The formation of the mammalian neocortex relies on the migration of projection neurons into specific layers, which is in turn regulated by cyclin-dependent kinase 5 (Cdk5). Cdk5 has previously been shown to interact with the G protein-coupled receptor serotonin 6 receptor (5-HT6R); however, whether and how this interaction might be important for neocortical migration remains unclear. Now, on p.3370, Alexandre Dayer and colleagues use a range of knockdown and rescue in utero electroporation experiments in mouse to show that 5-HT6R regulates the migration of the upper layer cortical projection neurons via its interaction with Cdk5, rather than with serotonin or other agonists. Knockdown of 5-HT6R in postmitotic upper layer neurons impairs their migration and leads to their presence in deeper layers of the neocortex, while re-expression of the full-length receptor or a Cdk5 kinase rescues this defect. The interaction between Cdk5 and 5-HT6R specifically affects the transition between multipolar and bipolar morphology in immature migrating neurons and provides in vivo evidence for the role of a G protein-coupled receptor in neocortical neuron migration. The sonic hedgehog (Shh) signalling pathway is a crucial mediator of cell proliferation, morphogenesis and fate, influencing the development and homeostasis of multiple organ systems during all stages of life. A central premise of Shh signalling is that the receptor patched 1 (Ptch1) mediates the Shh response, and that this response is greatest when Ptch1 is absent, allowing the release of pathway activator smoothened (Smo) and the eventual activation of Gli proteins. In this issue (p.3331), Henk Roelink and colleagues challenge this notion by showing that cells devoid of Ptch1 remain Shh responsive in both transcriptional and migrational assays. Furthermore, the authors demonstrate a role for patched 2 (Ptch2) in mediating the Shh response in the absence of Ptch1, since a response to Shh is seen in Ptch1–/– but not in Ptch1–/–;Ptch2–/– cells. These data are indicative of a complex relationship between Ptch1 and Ptch2 in their regulation of Smo activity in response to Shh.
The sonic hedgehog (Shh) signalling pathway is a crucial mediator of cell proliferation, morphogenesis and fate, influencing the development and homeostasis of multiple organ systems during all stages of life. A central premise of Shh signalling is that the receptor patched 1 (Ptch1) mediates the Shh response, and that this response is greatest when Ptch1 is absent, allowing the release of pathway activator smoothened (Smo) and the eventual activation of Gli proteins. In this issue (p.3331), Henk Roelink and colleagues challenge this notion by showing that cells devoid of Ptch1 remain Shh responsive in both transcriptional and migrational assays. Furthermore, the authors demonstrate a role for patched 2 (Ptch2) in mediating the Shh response in the absence of Ptch1, since a response to Shh is seen in Ptch1–/– but not in Ptch1–/–;Ptch2–/– cells. These data are indicative of a complex relationship between Ptch1 and Ptch2 in their regulation of Smo activity in response to Shh.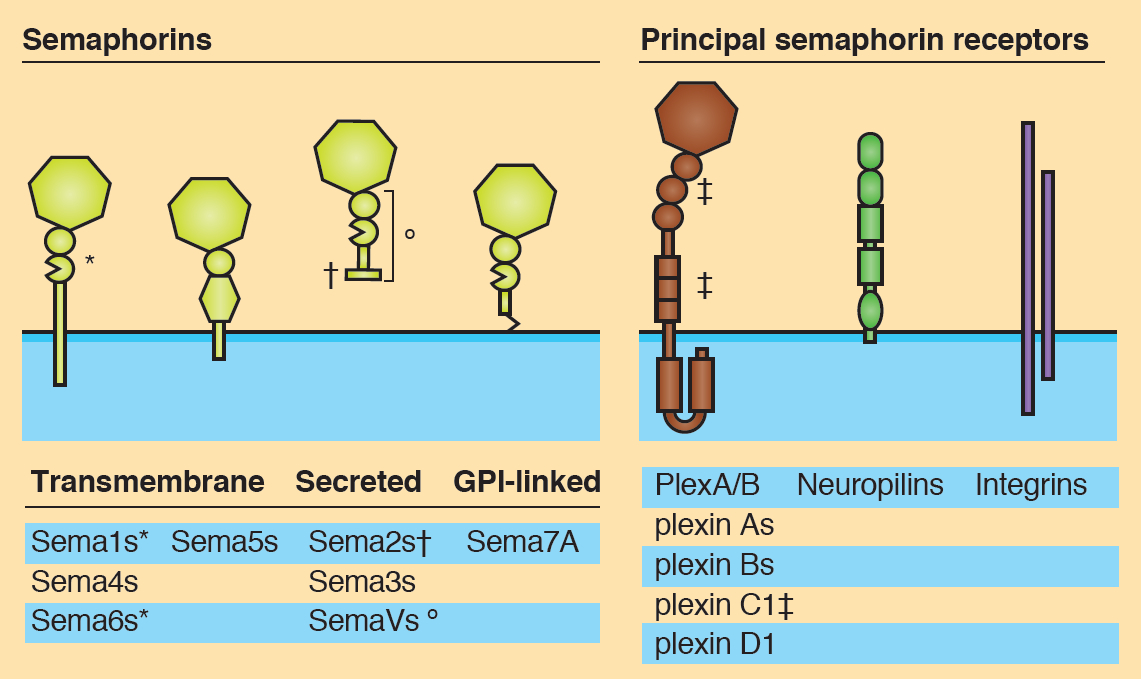 Semaphorins are secreted and membrane-associated proteins that regulate many cellular and developmental processes. In this article and accompanying poster, Jongbloets and Pasterkamp review the molecular biology of semaphorin signalling in different contexts. See the Review on p.3292
Semaphorins are secreted and membrane-associated proteins that regulate many cellular and developmental processes. In this article and accompanying poster, Jongbloets and Pasterkamp review the molecular biology of semaphorin signalling in different contexts. See the Review on p.3292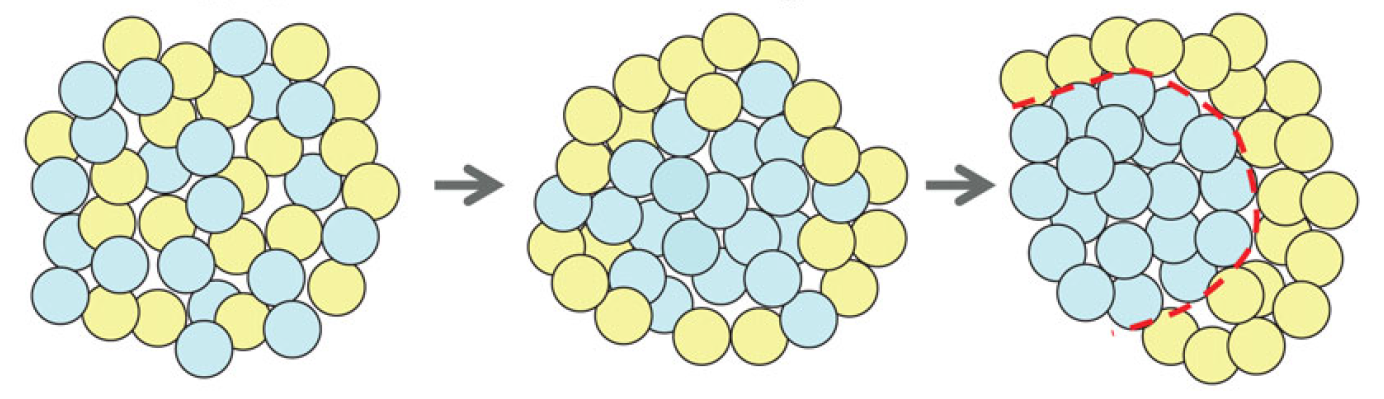 The separation of the embryo into physically distinct regions is one of the most important processes in development. François Fagotto discusses various boundary formation models and summarizes recent studies that have examined this process at the cellular level. See the Review on p.3303
The separation of the embryo into physically distinct regions is one of the most important processes in development. François Fagotto discusses various boundary formation models and summarizes recent studies that have examined this process at the cellular level. See the Review on p.3303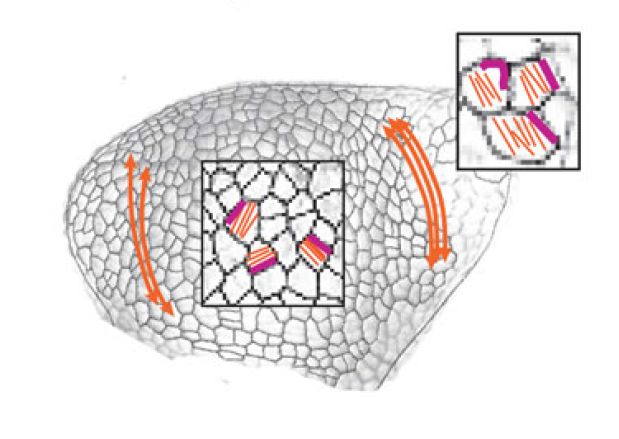 Jeffrey Axelrod and Dominique Bergmann report from The Company of Biologists workshop ‘Coordinating Cell Polarity’, which brought together researchers working on plant and animal systems to discuss the emerging themes in the field. See the Meeting Review on p.3298
Jeffrey Axelrod and Dominique Bergmann report from The Company of Biologists workshop ‘Coordinating Cell Polarity’, which brought together researchers working on plant and animal systems to discuss the emerging themes in the field. See the Meeting Review on p.3298

 (1 votes)
(1 votes) (No Ratings Yet)
(No Ratings Yet)



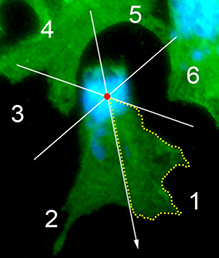 The neural crest (NC) is a transient and migratory population of cells that gives rise to a variety of cell types. During development, NC cells delaminate from the neural tube in a process that is closely coordinated with the process of neural tube closure, and studies have shown that signalling via the planar cell polarity (PCP) pathway is essential for both of these processes in Xenopus and zebrafish. However, it is unclear if PCP signalling is required for NC migration in mammals. Here, Andrew Copp and colleagues address this issue (p.
The neural crest (NC) is a transient and migratory population of cells that gives rise to a variety of cell types. During development, NC cells delaminate from the neural tube in a process that is closely coordinated with the process of neural tube closure, and studies have shown that signalling via the planar cell polarity (PCP) pathway is essential for both of these processes in Xenopus and zebrafish. However, it is unclear if PCP signalling is required for NC migration in mammals. Here, Andrew Copp and colleagues address this issue (p. 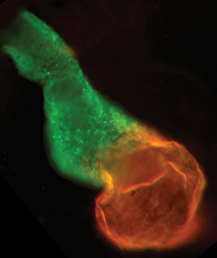 The production of cardiomyocytes is required both for embryonic heart formation and for cardiac regeneration following injury. The transcription factor Hand2 has been implicated in cardiomyocyte formation, but now, Deborah Yelon and colleagues demonstrate that Hand2 can in fact drive cardiomyocyte production in zebrafish (p.
The production of cardiomyocytes is required both for embryonic heart formation and for cardiac regeneration following injury. The transcription factor Hand2 has been implicated in cardiomyocyte formation, but now, Deborah Yelon and colleagues demonstrate that Hand2 can in fact drive cardiomyocyte production in zebrafish (p.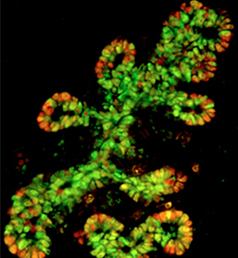 NR5A2 is an orphan nuclear hormone receptor that has diverse developmental and physiological functions. It is expressed in the inner cell mass of early mouse embryos and in the developing endoderm, but its role in organogenesis is unclear. Here, Ray MacDonald and co-workers reveal a crucial role for NR5A2 during pancreatic development in mice (p.
NR5A2 is an orphan nuclear hormone receptor that has diverse developmental and physiological functions. It is expressed in the inner cell mass of early mouse embryos and in the developing endoderm, but its role in organogenesis is unclear. Here, Ray MacDonald and co-workers reveal a crucial role for NR5A2 during pancreatic development in mice (p.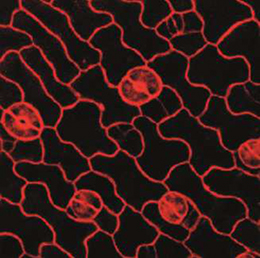 The development of stomata – the epidermal pores on plant leaves that regulate gas exchange – is tightly regulated by various environmental factors. Light, for example, promotes stomatal development; very few stomata are found on the epidermis of dark-grown seedlings. Here, on p.
The development of stomata – the epidermal pores on plant leaves that regulate gas exchange – is tightly regulated by various environmental factors. Light, for example, promotes stomatal development; very few stomata are found on the epidermis of dark-grown seedlings. Here, on p. Recent research suggests that circadian clock mechanisms control more than just daily timekeeping. Here, Steven Brown discusses how such mechanisms can influence stem cell biology and hence tissue development, homeostasis and regeneration. See the Review on p.
Recent research suggests that circadian clock mechanisms control more than just daily timekeeping. Here, Steven Brown discusses how such mechanisms can influence stem cell biology and hence tissue development, homeostasis and regeneration. See the Review on p.