A simple step to reverse ageing
Posted by Christele Gonneau, on 3 May 2014
How great would it be if we knew how to reverse ageing and turn old organs into young ones? Actually, this might not be as crazy as it sounds. As a matter of fact, a team of scientists managed to regenerate the thymus in old mice and observe what closely resembles the juvenile thymus!
The thymus is a key organ of the immune system as it is where T cells, major actors of one’s immunity, develop and mature. In normal healthy people, the thymus degenerates with age (a process called thymic involution) and this results in a decline of the immune system function. Since our immune system is what protects us against diseases, it is evident that being able to restore the function of the thymus in elder people would be very beneficial.
Interestingly, in this study recently published in Development, Bredenkamp and colleagues achieved thymus regeneration in old mice. They observed a juvenile-like thymus after using genetic engineering to force the expression of the protein foxn1 in the aged thymus.
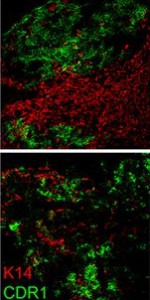
In this picture, you can observe the cortical thymic epithelial cell marker CDR1 in green and the medullary thymic epithelial cell marker keratin 14 (K14) in red. The cortex and the medulla are distinct regions of the thymus and they deteriorate during thymic involution. This results in reduced distinction between cortex and medulla as observed in the bottom picture taken in 24 months old mice. However, when foxn1 is over-expressed in the same 24 months old mice, you can observe that the clear distinction between cortex (green) and medulla (red) is restored, indicative of thymus regeneration.
In addition to the restoration of thymic architecture, the authors show that the regenerated organ has an increased T cell output and a gene expression profile similar to the juvenile thymus.
Most amazingly, apart from being able to “reverse ageing” in the thymus, they show that this regenerative process relies on the over-expression of a single factor (foxn1), making it a lot simpler than one might have anticipated! Thus, this study brings a new provocative concept that will most likely have a broad impact for regenerative biology.
Picture credit:
Bredenkamp, N., Nowell, C., & Blackburn, C. (2014). Regeneration of the aged thymus by a single transcription factor Development, 141 (8), 1627-1637 DOI: 10.1242/dev.103614


 (1 votes)
(1 votes)




 (No Ratings Yet)
(No Ratings Yet)
 Mesoangioblasts (MABs) are progenitor cells of embryonic derivation with mesodermal potential. They have been successfully used to restore skeletal muscle loss in dystrophic mice, but despite the clinical potential of these cells, their origin and role during development has not been defined. Now, on p.
Mesoangioblasts (MABs) are progenitor cells of embryonic derivation with mesodermal potential. They have been successfully used to restore skeletal muscle loss in dystrophic mice, but despite the clinical potential of these cells, their origin and role during development has not been defined. Now, on p.  The assembly of the peripheral nervous system occurs in a precise order: motor efferent axons (MEs) emerge first, followed by somatosensory afferent axons (SAs), and then by sympathetic efferent axons (SEs). While this order is clearly defined, it is not clear whether the pioneering axons provide instructive cues for the trailing axons to follow, and thus whether the network represents a true hierarchy. In this issue (p.
The assembly of the peripheral nervous system occurs in a precise order: motor efferent axons (MEs) emerge first, followed by somatosensory afferent axons (SAs), and then by sympathetic efferent axons (SEs). While this order is clearly defined, it is not clear whether the pioneering axons provide instructive cues for the trailing axons to follow, and thus whether the network represents a true hierarchy. In this issue (p.  How does a developing tissue know how much to grow and when to stop? On p.
How does a developing tissue know how much to grow and when to stop? On p.  Wound repair is a fundamental process that is required for tissue homeostasis and regeneration following damage. Most studies of wound healing have focussed on changes in the leading edge of wounded cells, but here William Razzell, Will Wood and Paul Martin show that morphogenetic cell shape changes that occur multiple cell rows back from the wound are important for efficient wound re-epithelialisation (p.
Wound repair is a fundamental process that is required for tissue homeostasis and regeneration following damage. Most studies of wound healing have focussed on changes in the leading edge of wounded cells, but here William Razzell, Will Wood and Paul Martin show that morphogenetic cell shape changes that occur multiple cell rows back from the wound are important for efficient wound re-epithelialisation (p.  Synaptogenesis is a complex process that involves the coordinated assembly of pre- and postsynaptic compartments. Various extracellular pathways and cues have been shown to regulate synapse formation but here, on p.
Synaptogenesis is a complex process that involves the coordinated assembly of pre- and postsynaptic compartments. Various extracellular pathways and cues have been shown to regulate synapse formation but here, on p.  Tissue morphogenesis is driven by coordinated cellular deformations and recent studies have shown that these changes in cell shape are powered by intracellular contractile networks comprising actin filaments, actin cross-linkers and myosin motors. In their Development at a Glance poster article, Munjal and Lecuit provide an overview of the mechanics, principles and regulation of actomyosin-driven cellular tension driving tissue morphogenesis. See the article on p.
Tissue morphogenesis is driven by coordinated cellular deformations and recent studies have shown that these changes in cell shape are powered by intracellular contractile networks comprising actin filaments, actin cross-linkers and myosin motors. In their Development at a Glance poster article, Munjal and Lecuit provide an overview of the mechanics, principles and regulation of actomyosin-driven cellular tension driving tissue morphogenesis. See the article on p.  Genes that are subject to genomic imprinting in mammals are preferentially expressed from a single parental allele. These imprinted genes can directly regulate fetal growth, and recent work has also demonstrated intricate roles for imprinted genes in the brain and in induced pluripotent stem cells and adult stem cells. As Bartolomei and colleagues review here, these findings highlight the complex nature and developmental importance of imprinted genes. See the Review on p.
Genes that are subject to genomic imprinting in mammals are preferentially expressed from a single parental allele. These imprinted genes can directly regulate fetal growth, and recent work has also demonstrated intricate roles for imprinted genes in the brain and in induced pluripotent stem cells and adult stem cells. As Bartolomei and colleagues review here, these findings highlight the complex nature and developmental importance of imprinted genes. See the Review on p. 
 (11 votes)
(11 votes)