Mouse embryology
Posted by bedu, on 30 October 2018
Posted by bedu, on 30 October 2018
Posted by Steffen Scholpp, on 30 October 2018
Closing Date: 15 March 2021
Several MRC-funded positions are available in the Scholpp lab in the Living Systems Institute (LSI) at the University of Exeter to elucidate various aspects of cytoneme-mediated Wnt trafficking in vertebrates.
We are looking for
a Postdoc (3years, starting in Spring 2019), two PhD students (3.5years, starting in Autumn 2019, UK/EU only), and a Tech (2years, starting in Spring 2019)
Application deadlines: November / December 2018!
More information about the lab, about the LSI, and about our recent research in The Node.
For more information contact s.scholpp@exeter.ac.uk.

Posted by __Deleted user__, on 28 October 2018
Closing Date: 15 March 2021
Several postdoctoral positions are available in the group of Taija Mäkinen at Uppsala University. The lab studies fundamental mechanisms of tissue morphogenesis in the vascular system. The aim is to understand how endothelial cells communicate with each other and the tissue environment to co-ordinate vascular morphogenesis and formation of functionally specialised blood and lymphatic vessel types. To do so, the group utilises advanced mouse genetic tools and state-of-the-art cell and molecular biology techniques (including single cell RNA sequencing, flow cytometry, confocal, light-sheet and super-resolution microscopy). For more details about the group’s research please see: http://www.makinenlab.com/
Selected recent publications from the laboratory:
Zhang et al, Nat Commun 2018; Frye et al, Nat Commun 2018; Zhang et al, Development 2018; Wang et al, Development 2017; Martina-Almedina et al, J Clin Invest 2016; Martinez-Corral et al, Circ Res 2015; Stanczuk et al, Cell Rep 2015; Tatin et al, Dev Cell 2013; Lutter et al, J Cell Biol 2012; Bazigou et al, J Clin Invest 2011.
Work description:
The selected candidate(s) will work on one of the following topics: 1) functional characterisation of tissue-specific lymphatic endothelial progenitor cells (as part of an ERC-funded project), 2) identification and functional characterisation of vascular-bed specific genes, or 3) elucidation of disease mechanisms in vascular malformations. In addition, one position will be in collaboration with the group of Ingvar Ferby at Uppsala University (http://www.imbim.uu.se/forskargrupper/cancer/ferby-ingvar/), exploring how vesicular trafficking and compartmentalisation of growth factor receptors instruct behaviour of epithelial and endothelial cells using e.g. live-cell imaging approaches.
Qualifications:
We are looking for highly motivated individuals with a PhD and research background in molecular or cell biology, developmental biology or biochemistry, and a proven track record of successful scientific work. Strong background in molecular/cell biology, mouse genetics, flow cytometry and/or imaging is required.
How to apply:
To apply, please send your CV together with the names of three references and a short description of yourself and the motivation to join the group to: taija.makinen@igp.uu.se
The position is open until 28 December 2018, or until suitable candidate(s) is found.
Posted by ecloke, on 26 October 2018
Closing Date: 15 March 2021

Are you interested in applying mathematics, statistics or deep learning/machine learning to biomedical problems? Apply now for a MRC WIMM Prize Studentship, to start in October 2019. The studentship is fully-funded for four years, including a stipend of £18,000 p.a. and all University and College fees paid.
Applicants with a background in Physics, Mathematics, Engineering, Statistics or Computer Science are encouraged to apply. To be eligible for a full award, applicants must have no restrictions on how long they can stay in the UK and must have been ordinarily resident in the UK for at least 3 years prior to the start of the studentship. Further details about residence requirements may be obtained here.
For further information on how to apply can be found here.
All applications must be received by 12 noon (UK time) on Friday, 11 January 2019
Interviews will take place the week commencing 28 January 2019.
Profiles
Iotchkova Group – Statistical Genetics
Koohy Group – Machine learning and integrative approaches in Immunology
Morrissey Group – Quantitative biology of cell fate and tissue dynamics
Sahakyan Group – Integrative Computational Biology and Machine learning

Posted by macasey, on 25 October 2018
The Zebrafish Interest Group at the University of Utah held its first Utah Fish Conference (UFC) on October 8, 2018. The conference was organized by pre- and post-doctoral trainees to celebrate the University’s Zebrafish Interest Group (ZIG), as well as to unite the Mountain West fish community. This 1-day event hosted over 80 attendees from 6 institutes. UFC was sponsored by the University’s ZIG, as well as Tecniplast USA, Aquatic Enterprises, IDT, Aquaneering, wFluidx, NCI, NEB, Zeiss, ThermoFisher by Life Technologies, and Developmental Dynamics.

The event featured two excellent keynotes, Trista E. North at Boston Children’s Hospital, and Bruce Draper at University of California-Davis. Dr. North jump-started the day’s event with a stimulating talk on the hematopoietic signaling connectome. She was followed by two hour-long sessions of trainee talks, which represented labs across the University and trainees from outside institutes. There was an active poster session comprising 30 posters from 16 labs, with presenters from all career stages, from undergraduate to faculty researchers.

An “Ask Me Anything” panel followed the poster session and featured both senior and junior faculty from the University, as well as Dr. North. A pre-doctoral trainee, Deeptha Vasudevan, moderated questions from the audience, and the panel covered topics from how to settle on a career path to advice on how to start your lab. The AMA was followed by an exciting evening talk from Dr. Bruce Draper about sex determination.

During dinner, the awards for best talk and best posters were announced. Robert Mackin (U of Idaho) won the Outstanding Young Investigator award for his excellent talk. Poster awards were given to 2 trainees in each category: undergraduate, graduate, or post-doctoral trainee. Awards went to: Jeffrey Dunn (BYU) and Samuel Caton (BYU) in the undergraduate category; Dana Klatt Shaw (U of Utah) and Srishti Kotiyal (U of Utah) in the graduate category; and Chelsea Herdman (U of Utah) and Angie Serrano (U of Utah) in the post-doctoral category. Awards were cash prizes sponsored by Developmental Dynamics. Following dinner, an after-party was hosted at The Porcupine Pub, sponsored by ThermoFisher by Life Technologies. The conversation was lively and offered more opportunities for trainees to intermix and mingle with faculty, Dr. North, and Dr. Draper.
The UFC was an invigorating moment for the University’s ZIG community, as well as for the Mountain West fish community. It was an excellent way to highlight the working happening within the region, and unite different institutes.
UFC2018 Organizing Committee:
Gabriel Bossé, PhD, @GabrielBosse1 (Randall Peterson Lab)
Macaulie Casey, @_macro16 (Kristen Kwan Lab)
Alexis Fulbright, @AlexisFulbright (Kim Evason Lab)
Chelsea Herdman, PhD, @ChelseaHerdman (H. Joseph Yost Lab)
Pui Ying Lam, PhD, (Randall Peterson Lab)
Sarah Lusk, @TheRealSlusk (Kristen Kwan Lab)
Luke Sanders, (H. Joseph Yost Lab)
Angie Serrano, PhD, @MAngieSerrano (H. Joseph Yost Lab)
Bhawika Sharma, @bhawi89 (H. Joseph Yost Lab)
Dana Klatt Shaw, @DanaShawsome (David Grunwald Lab)
Deeptha Vasudevan, @DeepthaVasu (Richard Dorsky Lab)
Kristen Kwan, PhD, @BlockInTheBack, kwan-lab.org (Faculty, Department of Human Genetics)
James Gagnon, PhD, @james_gagnon, gagnonlab.org (Faculty, Department of Biology)
Contributors:
Macaulie A. Casey (graduate student with Kristen M. Kwan, University of Utah)
Sarah L. Lusk (graduate student with Kristen M. Kwan, University of Utah)
Chelsea Herdman (postdoctoral fellow with H. Joseph Yost, University of Utah)
People to Follow:
University of Utah Zebrafish Interest Group, @Utah_ZIG
Utah Fish Conference, utahfishconference@gmail.com
Posted by LauraJohnston, on 25 October 2018
Join us for the Conference on Cell Competition in Development & Disease, in Tahoe, California!
February 24–28, 2019
Granlibakken Tahoe – Lake Tahoe, California, USA
Cell competition is a highly conserved process that promotes the context-dependent elimination of less fit cells and stimulates growth of more fit cells during growth and homeostasis. It has long been known that the basis of competition is the ability of growing cells to monitor their fitness and that of their neighbors, but only recently have signaling and effector mechanisms been identified. New technologies have uncovered the prevalence of cell competition in humans, with surprising outcomes and implications for human disease. This conference aims to bring together, for the first time, researchers from diverse fields who study competitive and cooperative interactions between cells.
Scientific Organizers:
Margaret A. Goodell, Baylor College of Medicine, USA
Laura A. Johnston, Columbia University, USA
Thomas P. Zwaka, Icahn School of Medicine at Mount Sinai, USA
Scholarship/Discounted Abstract Deadline: Oct 24, 2018; Abstract Deadline: Nov 28, 2018; Discounted Registration Deadline: Jan 8, 2019
Visit www.keystonesymposia.org/19B6 for more details.
Posted by boerckel, on 25 October 2018
Closing Date: 15 March 2021
Position Summary:
A postdoctoral position is available in the Developmental Mechanobiology and Regeneration lab of Joel Boerckel at the University of Pennsylvania in Philadelphia, PA, USA (http://www.med.upenn.edu/orl/boerckellab/). Our laboratory is housed in the Departments of Orthopaedic Surgery and Bioengineering and studies how mechanical cues direct morphogenesis, repair, and regeneration. A major focus of the lab is understanding how the transcriptional co-activators YAP and TAZ mediate progenitor cell mechanosensation, motility, and differentiation during development and regeneration.
We are looking for postdoctoral fellows with backgrounds in either cell and developmental biology or bioengineering to join our multidisciplinary team. NIH-funded projects are available to define the roles of YAP and TAZ in osteoprogenitor cell mobilization, mechanical regulation of endochondral ossification, and development-mimetic tissue engineering. Projects feature a combination of cell and bioreactor culture, biomaterials, and animal modeling using transgenic mice and rats.
What we offer:
What we’re looking for:
Start date immediately or upon mutual agreement.
Application materials (email to boerckel@pennmedicine.upenn.edu):
Posted by Marika Charalambous, on 25 October 2018
Closing Date: 15 March 2021
A Postdoctoral position is available in the Charalambous lab in the Department of Medical and Molecular Genetics at King’s College, London. Our team investigates genetic and epigenetic determinants of mammalian body composition. The candidate will be part of an exciting collaboration with the University of Bath, investigating the role of imprinted genes in adipose tissue development and turnover.
There is considerable phenotypic variation of body composition in the human population. This is clinically important because while some people maintain their health when exposed to a ‘Western’ lifestyle others develop obesity and associated metabolic disorders such as Type II Diabetes and cardiovascular disease. There is strong evidence that a large component of the variation in body composition is genetic, and furthermore – many of the genes may be acting in developmental pathways to modify skeletal muscle mass and adipose plasticity for a lifetime. By using in-vivo models to manipulate lean:adipose distribution we hope to identify the genetically determined developmental pathways that determine body composition, and understand how their compromise predisposes to metabolic disease.
The candidate will be part of a team based at Guy’s Campus, King’s College London, embedded within a centre of excellence for developmental biology and stem cell research. Moreover, the Department of Medical and Molecular Genetics has recently recruited a critical mass of researchers in the field of epigenetics, providing a strong source of crossover opportunities with this project.
Qualifications:
Applicants should have a recent Ph.D. degree or M.D./Ph.D. degree. Candidates with experience in stem cell and/or developmental biology, confocal microscopy, image acquisition and analysis will be preferred, as will those with bioinformatics skills. This position seeks a highly motivated individual with a strong interest in developmental biology.
Starting date:
The position is open starting from December 2018.
Requirements:
Please apply via the KCL portal below:
Contact:
Dr. Marika Charalambous, Department of Medical and Molecular Genetics, KCL, London, UK
marika.charalambous@kcl.ac.uk
Posted by the Node, on 25 October 2018
This Editorial by
The zebrafish is an increasingly popular model organism for human genetic disease research. CRISPR/Cas9-based approaches are currently used for multiple gene-editing purposes in zebrafish, but few studies have developed reliable ways to introduce precise mutations. Point mutation knock-in using CRISPR/Cas9 and single-stranded oligodeoxynucleotides (ssODNs) is currently the most promising technology for this purpose. Despite some progress in applying this technique to zebrafish, there is still a great need for improvements in terms of its efficiency, optimal design of sgRNA and ssODNs and broader applicability. The papers discussed in this Editorial provide excellent case studies on identifying problems inherent in the mutation knock-in technique, quantifying these issues and proposing strategies to overcome them. These reports also illustrate how the procedures for introducing specific mutations can be straightforward, such that ssODNs with only the target mutation are sufficient for generating the intended knock-in animals. Two of the studies also develop interesting point mutant knock-in models for cardiac diseases, validating the translational relevance of generating knock-in mutations and opening the door to many possibilities for their further study.
One of the great potentials of zebrafish (Danio rerio) is to generate accurate models of human genetic diseases to recapitulate their clinical features, to understand the molecular mechanisms that underpin them, and to model treatments and disease management approaches. Before the advent of precise genome editing, understanding a missense mutation within a protein-coding human gene in zebrafish necessitated designing an mRNA expression vector or a transgenic construct. These approaches did not actually replace the normal (wild-type) zebrafish gene with an altered human one. Additionally, expression of a human (trans)gene raises the question whether that gene is, in fact, functional in fish. Does the mutant gene function in fish? Are expression levels sufficient? Is expression in the right place and at the right time? These answers may be elusive.
Imagine instead the ability to map an amino acid in a protein from another species to a specific zebrafish protein residue and then mutate it. This is the essence of point mutation knock-in; namely, the replacement of wild-type nucleotides with mutant ones by inducing endogenous recombination with genome-editing reagents. An investigator can then confidently know that his or her gene of interest is modified precisely and expressed in the biologically relevant way – under the control of the endogenous promoter. With this refined approach, the mutation is the only variable under study. One potential disadvantage of current knock-in methodologies is that they cannot be used directly for introducing dominant and lethal mutations, which might be better studied using transgenic approaches that afford inducibility.
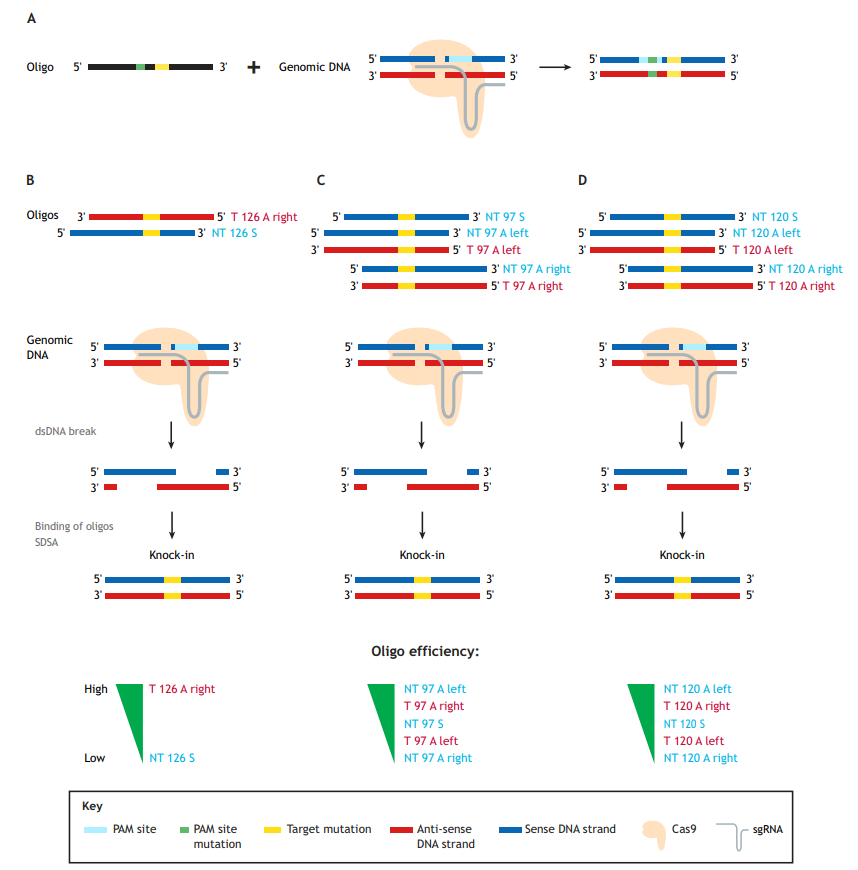
Generation of precise point mutations in zebrafish for basic research and disease modeling studies is an important contribution of the burgeoning CRISPR/Cas9 technology, which uses a single-guide RNA (sgRNA) molecule to guide the Cas9 endonuclease to the genomic site of choice. Single-stranded oligodeoxynucleotides (ssODNs), or oligos for short (as referred to herein), are currently the donor templates of choice for mutation knock-ins (Fig. 1A). These oligos can vary in length and be either ‘target’ (T) or ‘non-target’ (NT) in orientation, and have either symmetric or asymmetric homology arms. NT oligos correspond to the strand not bound by the sgRNA, which contains the protospacer-adjacent motif (PAM) sequence; hence, the oligos are sometimes referred to as ‘sense’. Conversely, T oligos are derived from the strand bound by the sgRNAs and are correspondingly often referred to as ‘anti-sense’.
Our own group previously reviewed zebrafish studies aimed at developing precise point mutation techniques using CRISPR/Cas9 (Prykhozhij et al., 2018a), including the first successful zebrafish study on germline transmission of a point mutation knock-in (Armstrong et al., 2016). In this Editorial, we will explore the implications of three papers published in this issue of Disease Models & Mechanisms that demonstrate point mutation knock-ins in zebrafish, and the advantages and challenges of working with such precise mutants (Boel et al., 2018; Tessadori et al., 2018; Farr et al., 2018). These papers pave the way toward CRISPR-based oligo knock-ins becoming widely acceptable as the preferred approach to generate point mutations in zebrafish.
The paper by Boel et al. focuses on the technical aspects of knock-in optimization (Boel et al., 2018). The first key observation in the Boel paper is that using oligos to guide homology-directed repair (HDR) is quite error prone. Although this has been previously shown in zebrafish (Burg et al., 2016; Hruscha et al., 2013; Hwang et al., 2013; Prykhozhij et al., 2018b), this report does so much more systematically. The authors identified typical ‘knock-in with indel’ events, insertions of partial and multiple oligos, inverse oligo insertions and other abnormal events. The prevalence of these abnormal events ranged from a few percent to >50% of the total knock-in events, thereby reducing the correct knock-in rates from 4-8% (total knock-ins) to 1-4% (correct). This result suggests that caution and routine use of next-generation sequencing (NGS) approaches are needed to assess the error rate of any new knock-in strategy. Given that each oligo in this study contained six nucleotide changes, the authors examined how much the distance between the Cas9-induced cut site and mutation influences the knock-in efficiency. Predictably, increasing this distance resulted in decreased knock-in efficiency, which was more pronounced for oligos with shorter homology arms, such as 60 nucleotides (nt), and for asymmetric (A) oligos, in which, for example, the left homology arm is 30 nt and the right is 90 nt long. This is consistent with previous studies (Paquet et al., 2016), and suggests that researchers use longer or symmetric (S) homology arms for engineering knock-ins in which the mutation site is at large distances from the Cas9-induced cut sites.
Boel and colleagues also investigated whether asymmetric homology arm design for 120 nt oligos can improve knock-in efficiency (Boel et al., 2018). No single type of oligo design performed best in their system, so they concluded that homology arm asymmetry is likely not a general strategy for improving knock-in rates. This somewhat contradicts previous studies in cell culture systems that found target asymmetric ‘T 126 A right’ oligos superior to non-target symmetric ‘NT 126 S’ oligos (Fig. 1B) (Richardson et al., 2016), and that ‘NT 97 A left’ or ‘T 97 A right’ were both superior to the symmetric ones (Fig. 1C) (Liang et al., 2017).
In zebrafish, our group recently confirmed the in vitro findings of Richardson et al. (2016) that target asymmetric oligos perform significantly better than the non-target symmetric oligos for two different knock-ins into the tp53 gene (Fig. 1B) (Prykhozhij et al., 2018b). Boel and colleagues also note, however, that for three out of the four sites in the zebrafish genome they targeted, the combined knock-in rates for 120 nt asymmetric (‘NT 120 A left’ and ‘T 120 A right’) oligos were higher than those of symmetric ones. These 120 nt asymmetric oligos also performed consistently better than the ones with opposite asymmetries (‘NT 120 A right’ and ‘T 120 A left’) (Fig. 1D), which is consistent with the known properties of homologous recombination-based DNA repair by resection and synthesis-dependent strand annealing (Paix et al., 2017). The authors make a reasonable recommendation to test these oligos in parallel with symmetric ones. Boel et al.’s main aim was technical knock-in optimization. The oligos they tested contained six nucleotide mismatches and were not intended to change the amino acid sequence of the protein product (Boel et al., 2018). The oligos used in our study contained non-silent disease-relevant mutations, which is likely a more common scenario for mutation knock-in studies. Targeted cells might have different tolerance for silent and non-silent mutations, which can result in different frequencies of successfully targeted clones. Future efforts will either resolve these efficiency discrepancies or perhaps render them irrelevant due to further improvement of genome-editing technologies.
Large-scale sequencing technology has greatly facilitated the identification of novel potentially disease-causing genomic variants. We have yet to understand the functional implications of these variants, but zebrafish can assist us in clarifying the complex genetic and molecular underpinning of these variants. The zebrafish has become one of the most sought after organisms in which to generate mutants that can serve as models of diseases caused by specific genomic variants. Most of these variants cannot be accurately modeled by complete gene inactivation or knockout and require knock-in approaches to introduce a specific mutation. Despite recent technological advances, few disease-associated zebrafish point mutants have been generated to date. The papers by Farr et al. and Tessadori et al. in this issue of Disease Models & Mechanisms describe point mutation knock-ins of several genetic variants implicated in inherited cardiac diseases (Farr et al., 2018; Tessadori et al., 2018). Tessadori et al. focus on mutations found in Cantú syndrome, a rare disease characterized by multiple cardiac abnormalities, bony changes and hair thickening. Farr et al. describe a zebrafish model of a PBX3 A136V mutation, which is enriched in a subset of congenital heart defects.
Both studies employed in vitro-transcribed sgRNA and either nCas9n mRNA (Jao et al., 2013) or Cas9 protein (Tessadori et al., 2018), together with oligos encoding the modifications. The distances between the Cas9 cut site and the nucleotide(s) to be mutated were 0-5 nt in the paper by Tessadori et al. and 10 nt in the paper by Farr et al. Both distances were in the previously reported optimal range (Paquet et al., 2016). Importantly, the mutations overlapped the sgRNA binding sites. Tessadori et al. used non-target strand mutant oligos with 25 nt homology arms and Farr et al. tested both non-target and target oligos, which were asymmetric (35 nt and 15 nt homology arms) if counted from the cut site. Interestingly, Farr and colleagues found that oligos with 25 nt homology arms worked well, whereas oligos with 20 nt and 40 nt homology arms failed. It is not clear whether homology arm asymmetry accounts for the performance differences, but it is likely that the shorter 10 nt homology arms are generally too short and 50 nt homology arms oligos might be too long for HDR. Once knock-in reagents were injected and the fish were subsequently bred, the resulting knock-in alleles needed to be detected and verified. Both papers employed Sanger sequencing and restriction enzyme digestion of the targeted site to evaluate knock-ins. Alternative approaches, such as allele-specific PCR, could be a more broadly applicable option (Prykhozhij et al., 2018b).
Both groups leveraged cut site proximity to the mutation sites and undertook some oligonucleotide size optimization to achieve efficient knock-in generation and germline transmission. The approaches described in these papers might prove effective for other mutation knock-ins in which the desired change is proximal to the Cas9 cut site. However, longer homologies and asymmetric homology arm designs might be needed for targeting mutation sites that are more distant from the predicted Cas9 cut site. Boel et al. (2018) suggest that much of the increase in efficiency observed due to lengthening oligos from 60 nt to 120 nt comes from aberrant repair events. It is therefore possible that 50-60 nt is a ‘sweet spot’ oligo length for cut-site proximal target mutation. The Farr and Tessadori groups should be lauded for identifying the minimal effective oligo size and simple knock-in procedures.
The main aim of these two studies was to model heart disease in zebrafish. Therefore, the mutations the authors introduced needed to produce a tractable, disease-relevant phenotype. Farr et al. aimed to test whether the zebrafish pbx4 A131V variant (which is homologous to the human PBX3 A136V mutation) could function as a modifier allele, resulting in a congenital heart defect. The pbx4 A131V allele did not produce a clearly discernable phenotype, but it was also not completely functional because its presence increased the severity of heart defects when combined with a null pbx4 allele (Farr et al., 2018). Tessadori and colleagues identified a convincing phenotype for their heterozygous and homozygous kcnj8V65M Cantú syndrome mutants. Their abcc9 G989E knock-in had a phenotype similar to that of the kcnj8V65M knock-in mutants, confirming a more generalized genotype-phenotype correlation (Tessadori et al., 2018). These newly developed zebrafish models could be used to improve our understanding of genetic heart disease and to test therapeutic approaches.
In sum, these three papers highlight various technical optimizations that can achieve robust and reproducible knock-in mutations, leading to zebrafish models with greater fidelity to the human diseases they are modeling. Lessons from these papers will be instructive to other investigators by providing important factors to consider when designing CRISPR/Cas9-based knock-ins in zebrafish. Although the generation of knock-in mutations continues to pose challenges, its successful implementation promises to be of tremendous value to the broader model organism community to study complex genetics that contribute to disease, in genes and in non-coding regions of the genome. By incorporating these mutants into high-throughput drug screening pipelines, the zebrafish holds great potential to provide rapid, cost-effective preclinical therapeutic data in a uniquely whole-organism vertebrate context that can streamline confirmatory murine studies and ultimately inform future clinical trials for patients with genetic disorders.
Armstrong, G. A. B., Liao, M., You, Z., Lissouba, A., Chen, B. E. and Drapeau, P.
(2016). Homology directed knockin of point mutations in the zebrafish tardbp and
fus genes in ALS using the CRISPR/Cas9 system. PLoS ONE 11, e0150188.
Boel, A., De Saffel, H., Steyaert, W., Callewaert, B., De Paepe, A., Coucke, P. J.
and Willaert, A. (2018). CRISPR/Cas9-mediated homology-directed repair by
ssODNs in zebrafish induces complex mutational patterns resulting from genomic integration of repair-template fragments. Dis. Model. Mech. 11,
dmm035352.
Burg, L., Zhang, K., Bonawitz, T., Grajevskaja, V., Bellipanni, G., Waring, R. and
Balciunas, D. (2016). Internal epitope tagging informed by relative lack of
sequence conservation. Sci. Rep. 6, 36986.
Farr, G. H., Imani, K., Pouv, D. and Maves, L. (2018). Functional testing of a human
PBX3 variant in zebrafish reveals a potential modifier role in congenital heart
defects. Dis. Model. Mech. 11, dmm035972.
Hruscha, A., Krawitz, P., Rechenberg, A., Heinrich, V., Hecht, J., Haass, C. and
Schmid, B. (2013). Efficient CRISPR/Cas9 genome editing with low off-target
effects in zebrafish. Development 140, 4982-4987.
Hwang, W. Y., Fu, Y., Reyon, D., Maeder, M. L., Kaini, P., Sander, J. D., Joung,
J. K., Peterson, R. T. and Yeh, J. R. J. (2013). Heritable and precise Zebrafish
genome editing using a CRISPR-Cas system. PLoS ONE 8, 1-9.
Jao, L.-E., Wente, S. R. and Chen, W. (2013). Efficient multiplex biallelic zebrafish
genome editing using a CRISPR nuclease system. Proc. Natl. Acad. Sci. USA
110, 13904-13909.
Liang, X., Potter, J., Kumar, S., Ravinder, N. and Chesnut, J. D. (2017).
Enhanced CRISPR/Cas9-mediated precise genome editing by improved design
and delivery of gRNA, Cas9 nuclease, and donor DNA. J. Biotechnol. 241,
136-146.
Paix, A., Folkmann, A., Goldman, D. H., Kulaga, H., Grzelak, M. J., Rasoloson,
D., Paidemarry, S., Green, R., Reed, R. R. and Seydoux, G. (2017). Precision
genome editing using synthesis-dependent repair of Cas9-induced DNA breaks.
Proc. Natl. Acad. Sci. USA 114, E10745-E10754.
Paquet, D., Kwart, D., Chen, A., Sproul, A., Jacob, S., Teo, S., Olsen, K. M.,
Gregg, A., Noggle, S. and Tessier-Lavigne, M. (2016). Efficient introduction of
specific homozygous and heterozygous mutations using CRISPR/Cas9. Nature
533, 125-129.
Prykhozhij, S. V., Caceres, L. and Berman, J. N. (2018a). New developments in
CRISPR/Cas-based functional genomics and their implications for research using
zebrafish. Curr. Gene Ther. 17, 286-300.
Prykhozhij, S. V., Fuller, C., Steele, S. L., Veinotte, C. J., Razaghi, B., Robitaille,
J. M., McMaster, C. R., Shlien, A., Malkin, D. and Berman, J. N. (2018b).
Optimized knock-in of point mutations in zebrafish using CRISPR/Cas9. Nucleic
Acids Res., 46, e102.
Richardson, C. D., Ray, G. J., DeWitt, M. A., Curie, G. L. and Corn, J. E. (2016).
Enhancing homology-directed genome editing by catalytically active and inactive
CRISPR-Cas9 using asymmetric donor DNA. Nat. Biotechnol. 34, 339-344.
Tessadori, F., Roessler, H. I., Savelberg, S. M. C., Chocron, S., Kamel, S. M.,
Duran, K. J., van Haelst, M. M., van Haaften, G. and Bakkers, J. (2018).
Effective CRISPR/Cas9-based nucleotide editing in zebrafish to model human
genetic cardiovascular disorders. Dis. Model. Mech. 11, dmm035469.
Posted by the Node Interviews, on 24 October 2018
The construction of complex three-dimensional tissue structures during embryogenesis requires precise control of cell and tissue mechanics. The Xenopus embryo provides a powerful tool for interrogating this relationship, as demonstrated by a recent Development paper reporting the use of tissue explants to test predictions of mechanical models. We caught up with first author and recent graduate Joseph Shawky and his supervisor Lance Davidson, Professor in the Department of Bioengineering in the University of Pittsburgh, to hear more about the work.

LD I arrived as a stranger in a strange land. My interests as a kid were a mix of collecting (perfect for biology) and astronomy (perfect for making). As many of my generation I wanted to be an astronaut and focused on physics and math through a BS in Physics at the University of Illinois and an MS in Space Science from York University in Toronto. I exited the academic stream for several years working as a scientific analyst in the nascent field of data science, but returned to earn a PhD in Biophysics from UC Berkeley. While at Berkeley I encountered radicals looking at the many roles of mechanics in biology, from the physics of virus capsid formation to the impact of waves on coastal kelp forests to the mechanics of sea urchin gastrulation. I had arrived in Berkeley as a theorist and left as an experimentalist. In my postdoc I cross-trained in amphibian embryology and cell biology. My mentors, Ray Keller and Doug DeSimone, were incredibly generous with training and tolerant of my digressions. They let me roam the embryo working on various processes ranging from neurulation to wound healing. In 2006 after an extended 8 year trip through the embryo, I took a faculty position in Bioengineering at the University of Pittsburgh. My group sits squarely between several cell and developmental biology labs and have we have terrific colleagues both at the University and at our neighbour institutions, Carnegie Mellon University.
We have two main questions in the lab and one area for technology development. The first question is “how do embryos work?” and by that I really mean the physical principle of work (force times distance), how forces are generated and how they shape tissues. My early experience as a theorist left me unsatisfied with the state of experimental embryology and we have consistently worked to develop tools and conceptual working models of morphogenesis. The second question is “why is development so robust?” This is a newer question to our group where we seek to understand how regulative programs of development integrate mechanical cues with conventional signalling and gene expression networks. This is an exciting new area for us and is leading in some surprising directions. The last topic, is much more focused on turning principles of development into tangible tissue engineering tools. The remarkable events of morphogenesis continually put tissue engineers to shame and we are trying to bridge that gap, both by training tissue engineers in concepts from morphogenesis, and in developing microfabrication tools to mimic embryonic tissue self-assembly.
JS I joined the Davidson lab as an eager graduate student fascinated by the world of cell and tissue mechanics. It was through the research opportunities I had during my undergraduate and master’s studies that I became interested in pursuing my PhD in the field of developmental biomechanics. I was first introduced to cell culture during my undergraduate studies at Rensselaer Polytechnic Institute (RPI) where I investigated potential therapies for breast cancer. From there, I continued to study breast cancer at Cornell University but in a different context. I studied how cancer cells reorganize their extracellular matrix during cell migration. Here, I was introduced to techniques to measure forces and stresses on the nanometer scale. I became very interested in the various approaches to study cell mechanics and the ability to link mechanical properties with biological phenomena. When I was deciding where to complete my graduate work, I was impressed by the various devices and techniques developed in house at the Davidson Lab to probe cell and tissue mechanics and was also excited to study tissue mechanics within developing embryonic tissues.
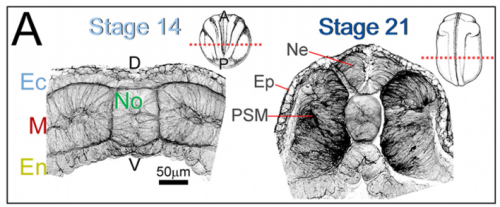
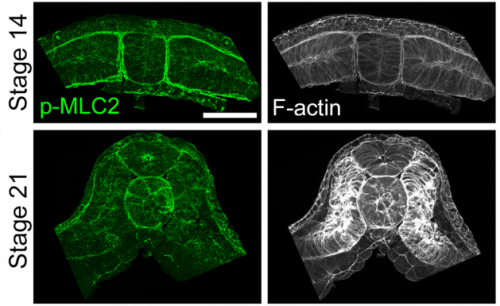
JS&LD The cellular solids model (CSM) is a theoretical framework that relates the stiffness of cellular materials to the microstructure of component cells. CSM was developed to characterize the mechanical properties of open- and closed-cell materials, both synthetic and biological in origin. CSM describes how factors such as cellular density, cell wall thickness and mechanical material properties affect the bulk stiffness of cellular materials. In our study, we tested the basic premise of the CSM to investigate how different features including cell size and cortex properties might affect the bulk mechanical properties of embryonic tissues. This model was useful because it makes predictions and allowed us to test the contribution of features at different size scales to critical bulk properties. However, at its finest scale the basic equations of the CSM do tell us how F-actin microstructure, including organization and degree of cross-linking, may be influencing mechanical material properties. Such mechanical models are key to understanding the direct role of mechanics in shaping tissues as well as the indirect role of mechanical cues in providing positional information for development.
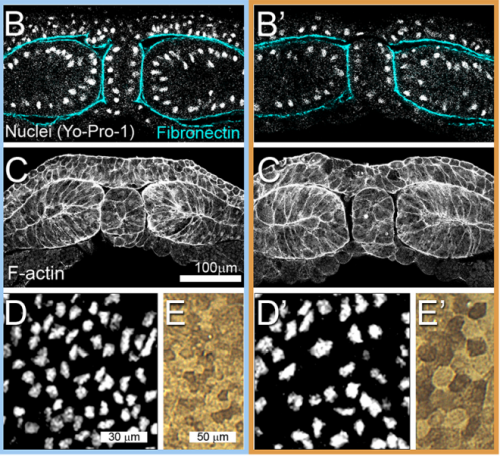
JS&LD In this study, our goal was to better understand the factors that contribute to bulk mechanics in Xenopus dorsal isolates and to shed light on possible mechanisms of stiffening during development. To formally make predications, we adapted the cellular solids model to embryonic tissues to predict how factors at different size scales may influence mechanics. We first confirmed the central assumption of the CSM, that tissue modulus does not depend on embedded structural elements, by engineering and mechanically testing a tissue devoid of large coherent 3D structures. We found that large-scale tissue architecture and cell size are not likely to influence bulk mechanical properties of early embryonic tissues. Our findings suggest that regulation of F-actin cortical thickness, density, and integrity plays a central role in regulating the physical mechanics of embryonic multicellular tissues.
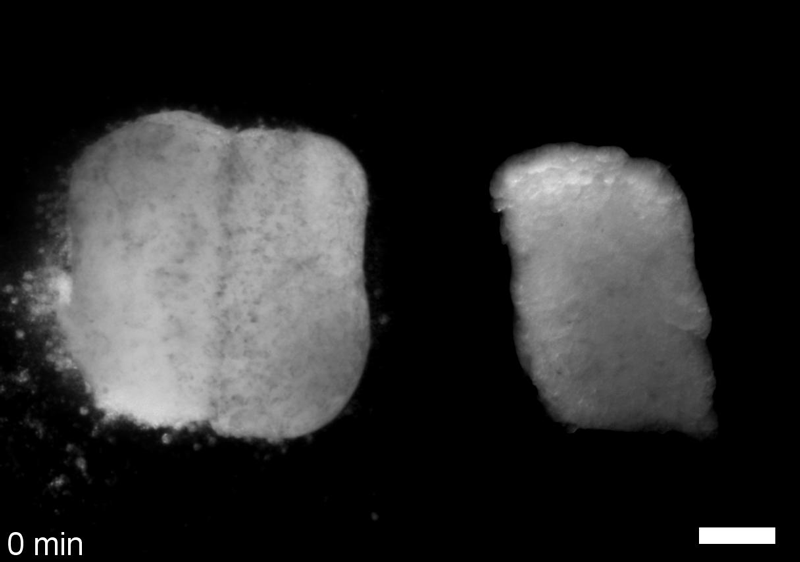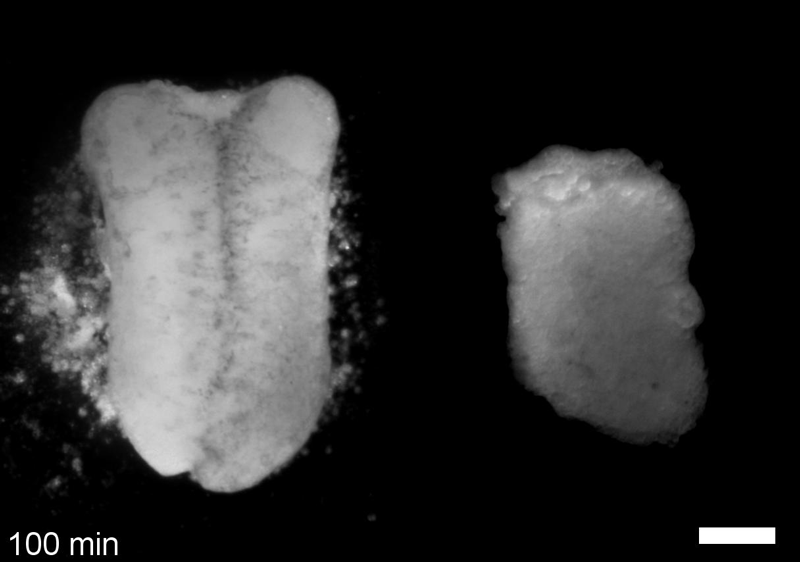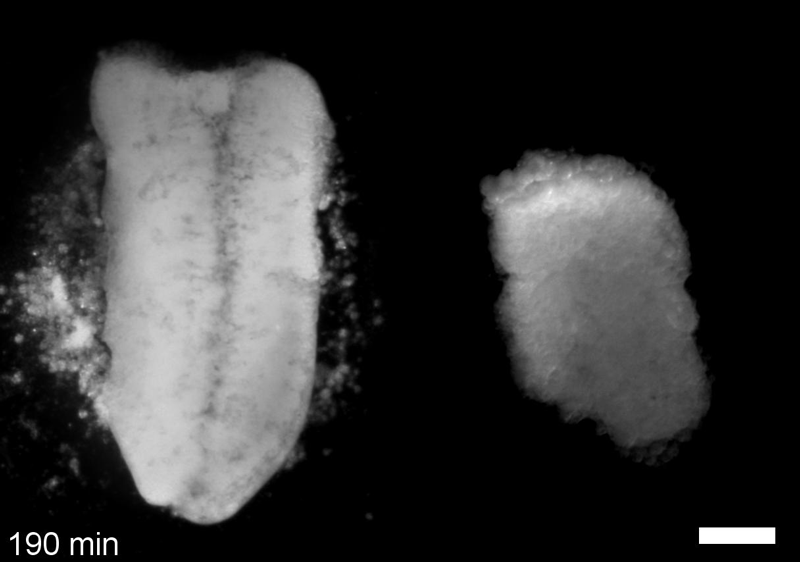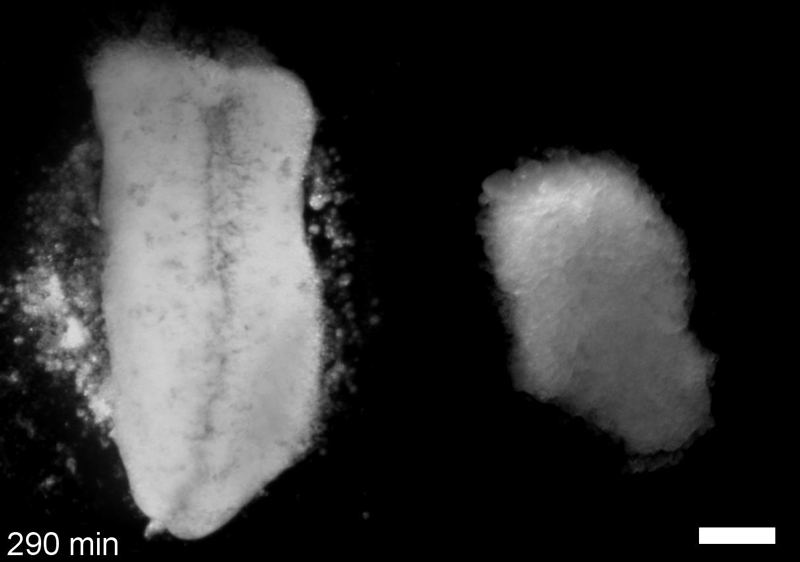
JS&LD Scrambled tissues were not able to reorganize or stiffen to the full extent of native tissues over time, suggesting that the organization of germ layers or germ layer tissue shape might be important for mechanical maturation. It remains unclear what aspects of architecture facilitate stiffening however could be organization of the ECM or in response to bulk morphogenesis movements. Fibronectin, the most abundant ECM protein during gastrulation and neurulation, does not directly contribute to mechanical properties as shown in a previous study from our lab however ECM may serve to align cells within the native stress field and lead to mechanical maturation.
(1) We have sparse experimental understanding of the mechanics of morphogenesis, gleaned from only a few model systems (fly, worm, frog, chick) with mechanically-accessible tissues (e.g. Drosophila epithelium, Xenopus explants), but know little about the lesions in human development that drive structural birth defects (e.g. spina bifida, congenital heart defects, cleft palate). How do these defects arise and are their ways they can be predicted and prevented?
(2) The mechanical microenvironment within embryos is largely hidden and has been the subject of long speculation but seldom the subject of rigorous experimental approaches. New technologies are removing the veil from these properties and allow us to start describing mechanical cues in embryos and testing how they may be involved in patterning.
(3) The field of tissue engineering arose from the needs of surgeons to reconstruct organs and tissues when regeneration and endogenous programs of healing fall short. More recently tissue engineers have begun to realize the remarkable robustness of developing tissues and organs. Harnessing evolved programs of self-assembly would open up an entirely new field where tissues and biological structures could be designed from the ground up in the same manner that electrical engineers design new circuits and civil engineers design new bridges. Co-opting the ‘technology’ of development would transform basic research, human health, and establish whole new industries.
JS One of the exciting moments in the lab was when we developed a process to generate viable and reproducible scrambled tissues. It took several design iterations and hours in the lab’s machine shop to develop a chamber and protocol that worked. These experiments were particularly exciting because it was an out of the box approach that allowed us to flip our research question on its head and tackle the question using a novel technique.
JS Early on it was challenging to identify techniques to alter cell size, particularly decreasing cell size, without disrupting development. We tried several approaches including removing cytoplasm from the single cell embryo. Once we got focused on manipulating cell cycle we were able to find reliable ways to inhibit and accelerate the cell cycle and generate tissues with increased and decreased cell density.
JS I’ve since graduated from the Davidson lab and completed a medical device policy fellowship at the U.S. Food and Drug Administration. Now, I am currently working as a Strategy and Analytics Consultant supporting clients within government health agencies.
LD The Xenopus embryo provides unparalleled access to the mechanics of morphogenesis and allows us to test ideas that folks working with other models would think preposterous. By focusing on the most elementary of tissue movements, convergent extension of the dorsal axis, we have been free from confounding changes in cell identity and 3D reorganizations that drive more complex morphogenesis movements. We are now applying tools and theoretical frameworks developed from this work to expose cryptic mechanical cues and their roles in shaping organs such as the heart, skin, and cardiovascular network.
LD I enjoy early morning dog walks through the forested city parks of Pittsburgh and lead a domestic life of cooking and gardening, but really I love spending free time in the lab since it is full of microscopes, mechanical gadgets, and frogs and there are so many open questions about embryonic development.
JS My favorite things to do in Pittsburgh are long bike rides on the beautiful riverside trails and enjoying the vibrant restaurant scene. Aside from biking and eating, I also enjoy grilling, hanging out with friends and a good podcast.
Multiscale analysis of architecture, cell size and the cell cortex reveals cortical F-actin density and composition are major contributors to mechanical properties during convergent extension. Joseph H. Shawky, Uma L. Balakrishnan, Carsten Stuckenholz, Lance A. Davidson
Development 2018 145: dev161281 doi: 10.1242/dev.161281
This is #51 in our interview series. Browse the archive here.