“My dear, here we must run as fast as we can, just to stay in place.
And if you wish to go anywhere you must run twice as fast as that.”
L. Carroll ‘Alice Through the Looking Glass’
We write this while we finish the last experiments for the final Show n’ Tell on Saturday full of reluctance to finish this course. Samples fly all over the lab; solutions are changed and something blue boils somewhere in the room.
Looking back, six weeks ago we had an approximate idea of what the MBL Embryology Course was about: people doing science 24/7, lectures and a lot of organisms with names you can barely pronounce. What we did not know is that the experience was all of that and more.
The first day was weird. People hanging their posters on the walls, rushing to get ready to tell other people about what is basically their life in the lab. After a while, everybody was talking with each other about science and how much they want to try new things. Our excitement grew as we realized that we would not only get to experience a plethora of organisms, but we would also learn from peers who are expert at widely diverged backgrounds. Little did we know about what we were really in for, though!
After that, the routine was as follows:
You wake up, rush to stuff yourself with breakfast, get a coffee and run to the Speck Auditorium to hear about the nuts and bolts of a new organism. From sea urchins to worms (flat or not), through articulated arthropods, squishy jellyfish and ctenophores, hemichordates, urochordates, glowing zebrafish or little mice, we learned about how they develop. One of the first things you realize is the kind of amazing things people are doing on this crazy endeavor of understanding how life works. All organisms share common features and are different from each other, highlighting how important all of them are. The second thing you realize is how passionate people are about their work, and you cannot help but remember why you became a scientist on the first place.
After the lectures we had what’s been called The Sweat Box for many years. This consists on a freestyle Q&A session about the contents of the lecture that usually would diverge on questions about anything related to the subject. During this time, we had the opportunity to discover how much people know about their (and other) organisms, and how they have contributed to their field and biology in general. As the course went on, our confidence grew to ask seemingly the strangest and deepest of questions. These turned out to be questions of broad interest or potential projects, which may highlight the key points of developmental research for the next decade.
After lunch, we had laboratory until the (usually very, very late) evening. The laboratories consisted on a brief introduction to the organism and the available tools. Then, it was up to you which type of question you wanted to ask: Big or small, simple or complex, you were given complete freedom and the TAs and PIs would usually challenge us to try new and classic experiments. You didn’t need to have any hypothesis or rigorous experimental design, as there should be no limitation for your curiosity. You just followed your intuition to give it a try for anything jumping up from your mind. It was a great chance to examine your crazy ideas, which your advisor might not allow you to test at your home laboratory, applying new available technologies to try experiments that were not previously feasible. Looking back, this curiosity-driven science is what we think that makes being a scientist an awesome job and we encourage everybody to do it, because that’s where magic happens. What would happen if you fuse two sea urchin embryos? Would a worm develop normally if you spin it down in the centrifuge? What is the smallest piece of an animal that would regenerate? Is that experiment mentioned earlier during the lecture really impossible? These questions are also important, and we had so much fun trying to answer them. Most of our experiments did not work, but science is about that, too: What basic principle have you learned by doing that experiment?
(It is important to mention that it is great seeing how the kind of experiments we the students propose still can surprise somebody that has been working in the field for many years. This is one of the great things about science: the capacity of being amazed)
Most importantly for our research in the ‘real world’, we had the opportunity to try new techniques and explore the use of tools we had never otherwise been able to use. For each established organism*, there are usually are some common experimental skills which are not utilized by researchers studying other organisms. We had the chance to learn many interdisciplinary, useful experimental skills and tool-making methods that can be applied to different organisms to broaden the possibility and feasibility of experimental designs in each module. Moreover, the training on different types of microscopes and imaging techniques enabled us to utilize appropriate methods, generating high-resolution images for different purposes and organisms. We could even separate overlapping spectra to distinguish (up to eight!!) different fluorescent markers on one sample for co-localization studies.
Every two weeks we got to present our data in the so-called Show n’ Tell. The recipe for it is a powerpoint presentation, a timer and people waiting to be amazed by the kind of data you collected in two weeks. During the time you were given, it was up to you presenting (or even dancing!) one, two or n experiments. It was very common seeing jaws dropping or people clapping, and everybody was smiling as they saw what a great time people had trying new experiments or collecting beautiful images. Our evening was spent laughing and embracing each other’s scientific creativity, an essential factor for the development of us as scientists.
Starting from first cell division, we had the opportunity to visualize the various types and patterns of cleavage processes among different organisms. We got to witness the beauty of biodiversity and how precisely it is regulated. When observing how a new life begins and grows, it is difficult to describe how deeply we were touched by the exquisiteness of life.
But not everything was about science! We had Sundays off, that we usually spent (not necessarily in this order) doing laundry, sleeping or exploring the beautiful towns of Woods Hole and Falmouth. We also had the opportunity of going Whale Watching, participating in the Independence Day costume parade and swimming in the bioluminescent ocean. During these six weeks of being awake at all hours, failing and succeeding together and enjoying our time off together, we became closer and closer to each other. With our virtues and defects, we have become friends.
Because science should be about that: whether your organism is big or small, squishy or hard, pigmented or transparent, we all are in the same boat. We all are trying to understand how life works, and we all should collaborate towards a better understanding of (in this case) embryology and development.

We’d like to finish this little story thanking our Course Directors, Richard and Alejandro, who made all of this possible. We’d also like to thank all the Faculty and TAs that spent endless hours preparing all the experiments so we could have fun in the lab. We couldn’t have done any of this without our amazing Course Assistants Wes, Chris and Brittany, who took care of everything and are a fundamental part of the course. We also appreciate the endless support from outside of course: the specialists from Zeiss and Leica, teaching us how to generate amazing images, and other sponsor companies providing the latest-model equipment for us to perform fancy experiments. And last, but not least, we’d like to thank you reader, for being curious enough to read this little piece. Never stop being amazed and get out your comfort zone, and magic will happen.
Joaquín, Aleisha and Tsai-Ming
MBL Embryology Course, Class of 2016
Joaquín Navajas Acedo is a Grad Student from the Grad School of the Stowers Institute for Medical Research in Kansas City, Missouri (USA). He is currently working on the establishment of cell polarity using zebrafish at the Piotrowski Lab (http://research.stowers.org/piotrowskilab)
Aleisha Symon is a PhD student from Monash University in Melbourne (Australia). She is currently researching genes that are involved in the development of the mammalian testis at the Hudson Institute of Medical Research in the Harley lab (http://hudson.org.au/profile-prof-vincent-harley/)
Tsai-Ming Lu is a PhD student from Marine Genomics Unit at Okinawa Institute of Science and Technology (OIST) Graduate University, Okinawa, Japan. He is working on the genome project of dicyemid mesozoan, Dicyema japonicum, a parasite inhabiting in the renal sac of octopus, to understand the mechanisms of regressive evolution and adaptive radiation (https://groups.oist.jp/mgu)
PS: Some of us live-tweeted during the 6 weeks we spent at the MBL using the hashtag #embryo2016. If you want to get a good idea about what the course was like, please visit: https://twitter.com/search?q=%23embryo2016&src=typd
*The usage of the expression ‘model organism’ and ‘non-model organism’ was passionately discussed during one of the sessions in the Sweat Box. Since each organism can be a model, we decided that the term ‘canonical’ or ‘established’ would be more appropriate or not even adding a label. There is a recent article that discusses the matter (http://www.cell.com/current-biology/fulltext/S0960-9822(16)30604-2)
 (10 votes)
(10 votes)
 Loading...
Loading...



 (No Ratings Yet)
(No Ratings Yet)
 (3 votes)
(3 votes)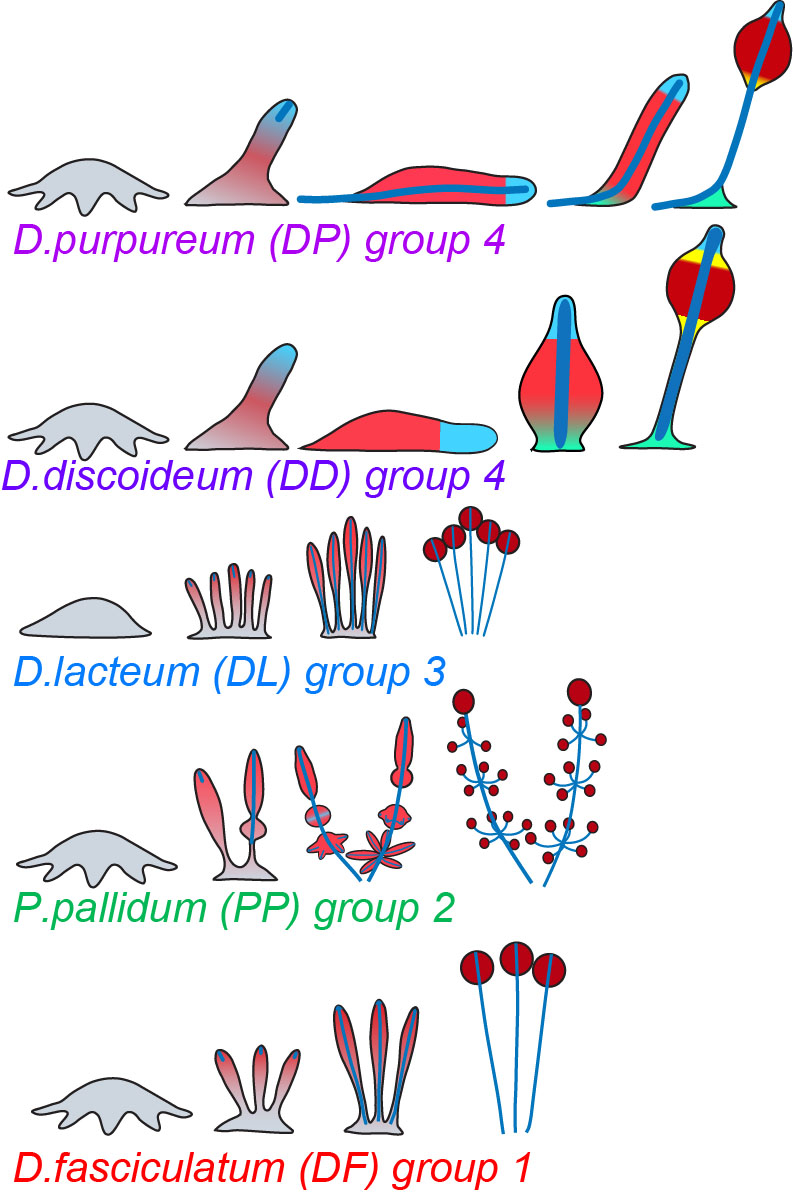




 Coincident with the blossoming of the sakura was the 14th annual CDB Symposium hosted by the RIKEN Center for Developmental Biology in Kobe, Japan. This year’s meeting, ‘Size in Development: Growth, Shape and Allometry’ focused on the molecular and cellular mechanisms underlying differences in size and shape and how they have evolved. Here, Iswar Hariharan highlights the advances presented at this meeting and the open questions in the field. See the Meeting Review on p.
Coincident with the blossoming of the sakura was the 14th annual CDB Symposium hosted by the RIKEN Center for Developmental Biology in Kobe, Japan. This year’s meeting, ‘Size in Development: Growth, Shape and Allometry’ focused on the molecular and cellular mechanisms underlying differences in size and shape and how they have evolved. Here, Iswar Hariharan highlights the advances presented at this meeting and the open questions in the field. See the Meeting Review on p.  Growing evidence suggests that current methods of direct lineage conversion may rely on the transition through a developmental intermediate. Here, Sam Morris explores pioneer transcription factor-driven direct lineage reprogramming between mature cell states, proposing that this depends on reversion to a developmentally immature state. See the Hypothesis article on p.
Growing evidence suggests that current methods of direct lineage conversion may rely on the transition through a developmental intermediate. Here, Sam Morris explores pioneer transcription factor-driven direct lineage reprogramming between mature cell states, proposing that this depends on reversion to a developmentally immature state. See the Hypothesis article on p.  Blood vessels in the skeletal system control multiple aspects of bone formation and provide niches for hematopoietic stem cells that reside within the bone marrow. Here,
Blood vessels in the skeletal system control multiple aspects of bone formation and provide niches for hematopoietic stem cells that reside within the bone marrow. Here, 
 (4 votes)
(4 votes)