The pivotal role of the stem cell environment
Posted by mpicbg, on 7 February 2014
Why iodine deficiency during pregnancy may have disastrous consequences
Higher mammals, such as humans, have markedly larger brains than other mammals. Scientists from the Max Planck Institute of Molecular Cell Biology and Genetics (MPI-CBG) in Dresden recently discovered a new mechanism governing brain stem cell proliferation. It serves to boost the production of neurons during development, thus causing the enlargement of the cerebral cortex – the part of the brain that enables us humans to speak, think and dream. The surprising discovery made by the Dresden-based researchers: two components in the stem cell environment – the extracellular matrix and thyroid hormones – work together with a protein molecule found on the stem cell surface, a so-called integrin. This likely explains why iodine deficiency in pregnant women has disastrous consequences for the unborn child, affecting its brain development adversely – without iodine, no thyroid hormones are produced. “Our study highlights this relationship and provides a potential explanation for the condition neurologists refer to as cretinism”, says Wieland Huttner, Director at the Max Planck Institute in Dresden. This neurological disorder severely impairs the mental abilities of a person.
In the course of evolution, certain mammals, notably humans, have developed larger brains than others, and therefore more advanced cognitive abilities. Mice, for example, have brains that are around a thousand times smaller than the human one. In their study, which was conducted in cooperation with the Fritz Lipmann Institute in Jena, the researchers in Dresden wanted to identify factors that determine brain development, and understand how larger brains have evolved.
A cosy bed for brain stem cells
Brain neurons are generated from stem cells called basal progenitors that are able to proliferate in humans, but not in mice. In humans, basal progenitors are surrounded by a special environment, a so-called extracellular matrix (ECM), which is produced by the progenitors themselves. Like a cosy bed, it accommodates the proliferating cells. Mice lack such ECM, which means that they generate fewer neurons and have a smaller brain.
The scientists therefore conducted tests to see whether in mice, basal progenitors start to proliferate if a comparable cell environment is simulated. The result: “We simulated an extracellular matrix for the brain stem cells using a stimulating antibody. This antibody activates an integrin on the cell surface of basal progenitors and thus stimulates their proliferation”, explains Denise Stenzel, who headed the experiments.
Because a requirement of thyroid hormones for proper brain development was previously known, the researchers blocked the production of these hormones in pregnant rats to see if their absence would inhibit basal progenitor proliferation in the embryos. Indeed, fewer progenitors and, consequently, neurons were produced, likely explaining the abnormal brain development in the absence of thyroid hormones. When the action of these hormones on the integrin was blocked, the ECM-simulating antibody alone was no longer able to induce basal progenitor proliferation.
A combination of ECM and thyroid hormones thus appears necessary for basal progenitors to proliferate and produce enough neurons for brain development. Human brain stem cells produce the suitable environment naturally. “That is probably how, in the course of evolution, we humans developed larger brains”, says Wieland Huttner, summing up the study. The research produced another important finding: “We were able to explain the role of iodine in embryonic brain development at the cellular level”, says Denise Stenzel. Iodine is essential for the production of thyroid hormones, and an iodine deficiency in pregnant women is known to have adverse effects on the brain development of the unborn child.
Original publication:
Stenzel, Denise; Wilsch-Bräuninger, Michaela; Wong, Fong Kuan; Heuer, Heike; Huttner, Wieland B.:
Integrin αvβ3 and thyroid hormones promote expansion of progenitors in embryonic neocortex.
Development (2014)
doi: 10.1242/dev.101907
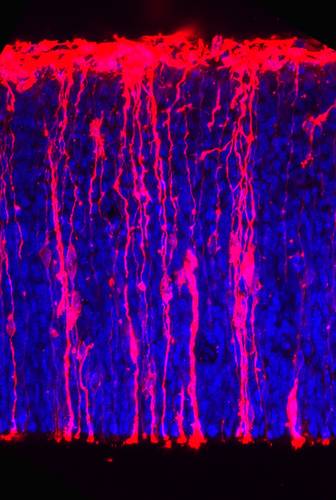
Stem cells in the cortex of a mouse embryo (cell nuclei: blue).
This article was first published on the 4th of February 2014 in the news section of the Max Planck Institute of Molecular Cell Biology and Genetics website


 (3 votes)
(3 votes) (No Ratings Yet)
(No Ratings Yet)






 This post is part of a series on a day in the life of developmental biology labs working on different model organisms. You can read the introduction to the series
This post is part of a series on a day in the life of developmental biology labs working on different model organisms. You can read the introduction to the series 


 I think that one of the scariest parts of being President of the Society for Developmental Biology is coming up with topics for these editorials in the Newsletter.
I think that one of the scariest parts of being President of the Society for Developmental Biology is coming up with topics for these editorials in the Newsletter. (25 votes)
(25 votes)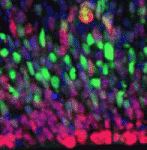 Within the developing neocortex, multiple progenitor cell types contribute to neuronal production, and the properties and relative abundance of these populations help to define the extent of neocortical growth. As well as apically localised radial glial cells (RGCs) that comprise the stem cell compartment, various populations of basal progenitors (BPs) exist – including basal RGCs, with both self-renewal and proliferative capacity, and more restricted progenitors. The stem cell-like properties of RGCs are linked to the fact that these cells are connected to the basal lamina, from which they receive proliferative signals via integrins. Now, Denise Stenzel et al. investigate in rodents the role of integrin αvβ3 in regulating the proliferative capacity of BPs (
Within the developing neocortex, multiple progenitor cell types contribute to neuronal production, and the properties and relative abundance of these populations help to define the extent of neocortical growth. As well as apically localised radial glial cells (RGCs) that comprise the stem cell compartment, various populations of basal progenitors (BPs) exist – including basal RGCs, with both self-renewal and proliferative capacity, and more restricted progenitors. The stem cell-like properties of RGCs are linked to the fact that these cells are connected to the basal lamina, from which they receive proliferative signals via integrins. Now, Denise Stenzel et al. investigate in rodents the role of integrin αvβ3 in regulating the proliferative capacity of BPs (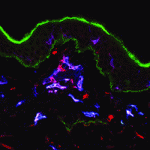 Langerhans cells (LCs) are antigen-presenting cells of the epidermis, and play a key role in detecting pathogens in the skin and coordinating the immune response. However, the precursors of LCs during embryonic development are poorly characterised, particularly in humans. Here (
Langerhans cells (LCs) are antigen-presenting cells of the epidermis, and play a key role in detecting pathogens in the skin and coordinating the immune response. However, the precursors of LCs during embryonic development are poorly characterised, particularly in humans. Here (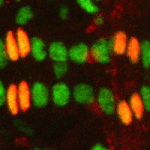 The Drosophila heart provides a relatively simple system for the analysis of gene regulatory networks (GRNs), as it comprises just two cell types – contractile cardial cells (CCs) and non-muscle pericardial cells (PCs). Moreover, many transcription factors (TFs) that regulate Drosophila heart development have been identified and shown to play conserved roles in mammals. On
The Drosophila heart provides a relatively simple system for the analysis of gene regulatory networks (GRNs), as it comprises just two cell types – contractile cardial cells (CCs) and non-muscle pericardial cells (PCs). Moreover, many transcription factors (TFs) that regulate Drosophila heart development have been identified and shown to play conserved roles in mammals. On  The shoot apical meristem (SAM) of higher plants contains the stem cell population that contributes to all above-ground organs. During vegetative growth, leaf primordia emerge from the periphery of the SAM, and the SAM transitions to an inflorescence meristem to initiate the reproductive phase. The WUSCHEL transcription factor specifies stem cell fate, and hence controls the size and activity of the SAM. A gene network involving the CLAVATA signalling pathway, the microRNA miR166g and HD-ZIPIII transcription factors is responsible for regulating WUSCHEL levels. On
The shoot apical meristem (SAM) of higher plants contains the stem cell population that contributes to all above-ground organs. During vegetative growth, leaf primordia emerge from the periphery of the SAM, and the SAM transitions to an inflorescence meristem to initiate the reproductive phase. The WUSCHEL transcription factor specifies stem cell fate, and hence controls the size and activity of the SAM. A gene network involving the CLAVATA signalling pathway, the microRNA miR166g and HD-ZIPIII transcription factors is responsible for regulating WUSCHEL levels. On 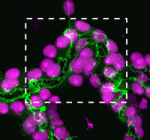 The Drosophila Mcr protein is a member of the thioester protein (TEP) family of proteins, which includes key regulators of innate immunity. Mcr is an unusual member of this family, possessing a putative transmembrane domain and lacking a key residue in the thioester motif. It has, nevertheless, been shown to be involved in phagocytic uptake in cultured Drosophila cells, although its putative immune functions have not been assessed in vivo. Two papers, from Stefan Luschnig and colleagues (
The Drosophila Mcr protein is a member of the thioester protein (TEP) family of proteins, which includes key regulators of innate immunity. Mcr is an unusual member of this family, possessing a putative transmembrane domain and lacking a key residue in the thioester motif. It has, nevertheless, been shown to be involved in phagocytic uptake in cultured Drosophila cells, although its putative immune functions have not been assessed in vivo. Two papers, from Stefan Luschnig and colleagues (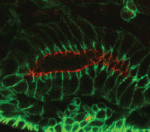 Both studies identify lethal EMS mutations inMcr in independent genetic screens, finding mutant phenotypes typical of SJ components. Consistent with a putative role in SJ formation, the protein colocalises with other SJ proteins at the lateral membrane. They further find that Mcr localisation is dependent on core SJ components and that, conversely, SJ proteins are mislocalised in Mcr mutants. At the morphological and ultrastructural level, SJs are disrupted in the absence of Mcr, and functional assays demonstrate that Mcr is required to form an effective paracellular barrier. As well as identifying a new SJ protein essential for barrier integrity, these two studies suggest an intriguing link between SJs and innate immunity. The epithelial barrier represents the first line of defence against pathogen invasion, andDrosophila haemocytes are known to undergo an epithelialisation-like process when encapsulating pathogens in the haemolymph. The identification of Mcr as a protein involved in both SJ formation and innate immunity now provides a molecular connection, and opens up new avenues for investigating potential functional links, between these two seemingly disparate processes.
Both studies identify lethal EMS mutations inMcr in independent genetic screens, finding mutant phenotypes typical of SJ components. Consistent with a putative role in SJ formation, the protein colocalises with other SJ proteins at the lateral membrane. They further find that Mcr localisation is dependent on core SJ components and that, conversely, SJ proteins are mislocalised in Mcr mutants. At the morphological and ultrastructural level, SJs are disrupted in the absence of Mcr, and functional assays demonstrate that Mcr is required to form an effective paracellular barrier. As well as identifying a new SJ protein essential for barrier integrity, these two studies suggest an intriguing link between SJs and innate immunity. The epithelial barrier represents the first line of defence against pathogen invasion, andDrosophila haemocytes are known to undergo an epithelialisation-like process when encapsulating pathogens in the haemolymph. The identification of Mcr as a protein involved in both SJ formation and innate immunity now provides a molecular connection, and opens up new avenues for investigating potential functional links, between these two seemingly disparate processes.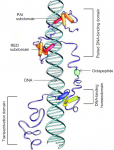 Pax genes encode a family of transcription factors that orchestrate complex processes of lineage determination in the developing embryo. Here, Judith Blake and Melanie Ziman review the molecular functions of Pax genes during development and detail the regulatory mechanisms by which they specify and maintain progenitor cells across various tissue lineages. See the Primer article on p.
Pax genes encode a family of transcription factors that orchestrate complex processes of lineage determination in the developing embryo. Here, Judith Blake and Melanie Ziman review the molecular functions of Pax genes during development and detail the regulatory mechanisms by which they specify and maintain progenitor cells across various tissue lineages. See the Primer article on p.  James Wells and Jason Spence review how recent advances in developmental and stem cell biology have made it possible to generate complex, three-dimensional, human intestinal tissues in vitro through directed differentiation of human pluripotent stem cells. See the Primer on p.
James Wells and Jason Spence review how recent advances in developmental and stem cell biology have made it possible to generate complex, three-dimensional, human intestinal tissues in vitro through directed differentiation of human pluripotent stem cells. See the Primer on p.