Slidecasts and videos from conference on congenital melanocytic nevus
Posted by Heather, on 5 February 2014
Limited time offer! Read on.

As a developmental biologist, I have found my calling in applying what I have learned about normal embryogenesis to better understanding the pathophysiology of various human congenital malformations. Often these are rare diseases, and I work closely with patient associations devoted to those conditions and learning to live with them (or with the death of the child who had been afflicted).
Among these, the congenital melanocytic nevus (CMN) is one in which I have been interested for the longest time. I organized a conference last fall in Marseille specifically devoted to the basic biology, epidemiology and medical and psychological considerations around the largest and rarest forms of CMN. I have also assisted the existing worldwide patient groups to federate, which will help us help them build a prospective registry for further research.
I didn’t do it alone, of course. Mark Beckwith from Nevus Outreach, Inc. was the most active, and the only non-physician, member of my organizing committee. We innovated by asking all the speakers to make their presentations available online, behind an inexpensive but secure paywall, and by dubbing the presentations with the sound of their actual delivery. In some settings, this is known as a slidecast. The slides advance automatically in sync with the sound.
We also placed the videos, when relevant, of the Q&A periods following the talks. The titles of the talks in the programme are at this link, and the PDF with the abstracts and titles can be downloaded directly from this one.
The idea was to make a resource that would slowly garner hits over time, as I have done with some of my teaching in the past. However, the company hosting the slidecasts has decided to eliminate the slidecast offering for good, after seven years, in the next three months. After I paid for a subscription for a full year, of course.
So: 33 presentations from the ESPCR-sponsored International Expert Meeting on Large Congenital Melanocytic Nevi and Neurocutaneous Melanocytosis in September, 2013, are currently available online. They are web-viewable “slidecasts” (author-approved slideshows with synchronized sound from the live presentations). In addition, there are 29 videos of the corresponding question and answer sessions. The slidecasts disappear at the end of April, 2014.
You can virtually “attend” this conference for only 25 euros (approximately 35 USD or 21 GBP) by navigating to this webpage. All proceeds will directly support building a prospective patient registry by the Naevus Global international federation of advocacy groups. I’m writing this to ask you to support a worthy cause and consider learning about the direct result of the developmental biology of a neural crest derivative, gone wrong in one particular molecular way (I’ll let you discover which).
After secure payment through PayPal using either a PayPal account or a credit card, we hand-distribute unique identifiers and the address of a restricted part of the Naevus Global site. There, the order of the program is reproduced, with hyperlinks to the slidecasts and videos.
Thank you for your interest in current research on congenital melanocytic nevi & neurocutaneous melanocytosis! I hope you find this resource beneficial and informative, and that you will support the patient registry initiative in this manner.
Sincerely yours,
Heather C. Etchevers, Ph.D.


 (3 votes)
(3 votes)
 I think that one of the scariest parts of being President of the Society for Developmental Biology is coming up with topics for these editorials in the Newsletter.
I think that one of the scariest parts of being President of the Society for Developmental Biology is coming up with topics for these editorials in the Newsletter. (25 votes)
(25 votes)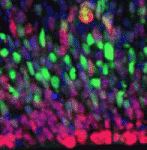 Within the developing neocortex, multiple progenitor cell types contribute to neuronal production, and the properties and relative abundance of these populations help to define the extent of neocortical growth. As well as apically localised radial glial cells (RGCs) that comprise the stem cell compartment, various populations of basal progenitors (BPs) exist – including basal RGCs, with both self-renewal and proliferative capacity, and more restricted progenitors. The stem cell-like properties of RGCs are linked to the fact that these cells are connected to the basal lamina, from which they receive proliferative signals via integrins. Now, Denise Stenzel et al. investigate in rodents the role of integrin αvβ3 in regulating the proliferative capacity of BPs (
Within the developing neocortex, multiple progenitor cell types contribute to neuronal production, and the properties and relative abundance of these populations help to define the extent of neocortical growth. As well as apically localised radial glial cells (RGCs) that comprise the stem cell compartment, various populations of basal progenitors (BPs) exist – including basal RGCs, with both self-renewal and proliferative capacity, and more restricted progenitors. The stem cell-like properties of RGCs are linked to the fact that these cells are connected to the basal lamina, from which they receive proliferative signals via integrins. Now, Denise Stenzel et al. investigate in rodents the role of integrin αvβ3 in regulating the proliferative capacity of BPs (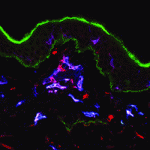 Langerhans cells (LCs) are antigen-presenting cells of the epidermis, and play a key role in detecting pathogens in the skin and coordinating the immune response. However, the precursors of LCs during embryonic development are poorly characterised, particularly in humans. Here (
Langerhans cells (LCs) are antigen-presenting cells of the epidermis, and play a key role in detecting pathogens in the skin and coordinating the immune response. However, the precursors of LCs during embryonic development are poorly characterised, particularly in humans. Here (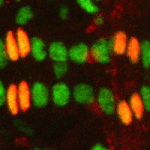 The Drosophila heart provides a relatively simple system for the analysis of gene regulatory networks (GRNs), as it comprises just two cell types – contractile cardial cells (CCs) and non-muscle pericardial cells (PCs). Moreover, many transcription factors (TFs) that regulate Drosophila heart development have been identified and shown to play conserved roles in mammals. On
The Drosophila heart provides a relatively simple system for the analysis of gene regulatory networks (GRNs), as it comprises just two cell types – contractile cardial cells (CCs) and non-muscle pericardial cells (PCs). Moreover, many transcription factors (TFs) that regulate Drosophila heart development have been identified and shown to play conserved roles in mammals. On  The shoot apical meristem (SAM) of higher plants contains the stem cell population that contributes to all above-ground organs. During vegetative growth, leaf primordia emerge from the periphery of the SAM, and the SAM transitions to an inflorescence meristem to initiate the reproductive phase. The WUSCHEL transcription factor specifies stem cell fate, and hence controls the size and activity of the SAM. A gene network involving the CLAVATA signalling pathway, the microRNA miR166g and HD-ZIPIII transcription factors is responsible for regulating WUSCHEL levels. On
The shoot apical meristem (SAM) of higher plants contains the stem cell population that contributes to all above-ground organs. During vegetative growth, leaf primordia emerge from the periphery of the SAM, and the SAM transitions to an inflorescence meristem to initiate the reproductive phase. The WUSCHEL transcription factor specifies stem cell fate, and hence controls the size and activity of the SAM. A gene network involving the CLAVATA signalling pathway, the microRNA miR166g and HD-ZIPIII transcription factors is responsible for regulating WUSCHEL levels. On 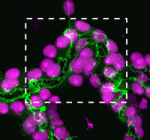 The Drosophila Mcr protein is a member of the thioester protein (TEP) family of proteins, which includes key regulators of innate immunity. Mcr is an unusual member of this family, possessing a putative transmembrane domain and lacking a key residue in the thioester motif. It has, nevertheless, been shown to be involved in phagocytic uptake in cultured Drosophila cells, although its putative immune functions have not been assessed in vivo. Two papers, from Stefan Luschnig and colleagues (
The Drosophila Mcr protein is a member of the thioester protein (TEP) family of proteins, which includes key regulators of innate immunity. Mcr is an unusual member of this family, possessing a putative transmembrane domain and lacking a key residue in the thioester motif. It has, nevertheless, been shown to be involved in phagocytic uptake in cultured Drosophila cells, although its putative immune functions have not been assessed in vivo. Two papers, from Stefan Luschnig and colleagues (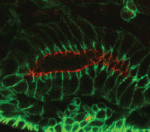 Both studies identify lethal EMS mutations inMcr in independent genetic screens, finding mutant phenotypes typical of SJ components. Consistent with a putative role in SJ formation, the protein colocalises with other SJ proteins at the lateral membrane. They further find that Mcr localisation is dependent on core SJ components and that, conversely, SJ proteins are mislocalised in Mcr mutants. At the morphological and ultrastructural level, SJs are disrupted in the absence of Mcr, and functional assays demonstrate that Mcr is required to form an effective paracellular barrier. As well as identifying a new SJ protein essential for barrier integrity, these two studies suggest an intriguing link between SJs and innate immunity. The epithelial barrier represents the first line of defence against pathogen invasion, andDrosophila haemocytes are known to undergo an epithelialisation-like process when encapsulating pathogens in the haemolymph. The identification of Mcr as a protein involved in both SJ formation and innate immunity now provides a molecular connection, and opens up new avenues for investigating potential functional links, between these two seemingly disparate processes.
Both studies identify lethal EMS mutations inMcr in independent genetic screens, finding mutant phenotypes typical of SJ components. Consistent with a putative role in SJ formation, the protein colocalises with other SJ proteins at the lateral membrane. They further find that Mcr localisation is dependent on core SJ components and that, conversely, SJ proteins are mislocalised in Mcr mutants. At the morphological and ultrastructural level, SJs are disrupted in the absence of Mcr, and functional assays demonstrate that Mcr is required to form an effective paracellular barrier. As well as identifying a new SJ protein essential for barrier integrity, these two studies suggest an intriguing link between SJs and innate immunity. The epithelial barrier represents the first line of defence against pathogen invasion, andDrosophila haemocytes are known to undergo an epithelialisation-like process when encapsulating pathogens in the haemolymph. The identification of Mcr as a protein involved in both SJ formation and innate immunity now provides a molecular connection, and opens up new avenues for investigating potential functional links, between these two seemingly disparate processes.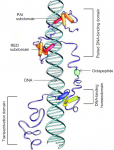 Pax genes encode a family of transcription factors that orchestrate complex processes of lineage determination in the developing embryo. Here, Judith Blake and Melanie Ziman review the molecular functions of Pax genes during development and detail the regulatory mechanisms by which they specify and maintain progenitor cells across various tissue lineages. See the Primer article on p.
Pax genes encode a family of transcription factors that orchestrate complex processes of lineage determination in the developing embryo. Here, Judith Blake and Melanie Ziman review the molecular functions of Pax genes during development and detail the regulatory mechanisms by which they specify and maintain progenitor cells across various tissue lineages. See the Primer article on p.  James Wells and Jason Spence review how recent advances in developmental and stem cell biology have made it possible to generate complex, three-dimensional, human intestinal tissues in vitro through directed differentiation of human pluripotent stem cells. See the Primer on p.
James Wells and Jason Spence review how recent advances in developmental and stem cell biology have made it possible to generate complex, three-dimensional, human intestinal tissues in vitro through directed differentiation of human pluripotent stem cells. See the Primer on p.  (No Ratings Yet)
(No Ratings Yet)