A study on cell migration provides insights into the movement of cancer cells
Posted by IRBBarcelona, on 20 November 2013
Using Drosophila melanogaster, researchers at IRB Barcelona discover that during multiple cell migrations a single cell can act as leader, dragging the others with it.
_____________
The migration of groups of cells in order to form tissues is common during the development of an organism. Discovering how these multiple movements are achieved is not only crucial to understand the basic principles of development but provides new information and insights for further research into processes associated with the spread of cancer.
Jordi Casanova, head of the “Morphogenesis in Drosophila” lab at IRB Barcelona and CSIC research professor, and Gaëlle Lebreton, postdoctoral fellow in the same group, have published a study performed using Drosophila melanogaster in the Journal of Cell Science. This work reveals that in a multiple movement, a single cell can act as the leader and can drag the rest with it. The scientists have studied the tracheal development of Drosophila in vivo and describe the morphological characteristics of the leading cell and provide molecular details about how it drives the movement.
“Cancer researchers are keen to know how cells are organized to achieve migration and to form new capillaries to feed an expanding cancerous tumor,” explains Gaëlle Lebreton, first author of the article. “Our study gives new data about how angiogenesis might arise,” comments the French scientist at IRB Barcelona. Angiogenesis or the formation of new blood vessels is a critical process in the context of cancer because it is one of the steps that mark the transformation of a benign tumour into a malignant one. The formation of new blood vessels involves the synchronized movements of groups of cells. In this regard, understanding how these groups work will open up new research lines on angiogenesis.
Over seven hours, the scientists tracked a group of seven cells that form one of the tracheal branches of the fly Drosophila melanogaster in its first hours of development. The leading cell is the only one that has receptors for the growth factor FGF. The FGF signal stimulates a cascade of reactions in this cell in order to generate sufficient energy and to turn it into the promoter of motility.
“This is a novel piece of work because we monitored the entire process in vivo and because it is the first time we have seen, in an experimental context, that a single cell can lead this multiple migration,” says Casanova.
It is important to note that the development of trachea in the Drosophila fly is similar to that of bronchia in humans. Consequently, this development is also of biomedical interest in order to unravel the basic processes involved in the formation of new tissue.
Reference article:
Specification of leading and trailing cell features during collective migration in the Drosophila trachea
Lebreton G, Casanova J.
J Cell Sci. 2013 Nov 8. [Epub ahead of print]
This article was first published on the 20th of November 2013 in the news section of the IRBBarcelona website.


 (2 votes)
(2 votes)
 (No Ratings Yet)
(No Ratings Yet)
 Mammalian cardiac regeneration is greatly impeded by the massive loss of cardiomyocytes that occurs following acute injury. The failure of the remaining cells to proliferate is a considerable challenge for the field, but the molecular mechanisms that control cardiomyocyte proliferation in the adult heart are largely unknown. Now, on
Mammalian cardiac regeneration is greatly impeded by the massive loss of cardiomyocytes that occurs following acute injury. The failure of the remaining cells to proliferate is a considerable challenge for the field, but the molecular mechanisms that control cardiomyocyte proliferation in the adult heart are largely unknown. Now, on  Precise orchestration of palate formation involves the complex interaction of signalling cascades and transcriptional networks in the developing craniofacial region. Pax9 and Osr2 have previously been implicated in palate formation, but little is known about how these molecular components interact within the greater regulatory network. Now, on
Precise orchestration of palate formation involves the complex interaction of signalling cascades and transcriptional networks in the developing craniofacial region. Pax9 and Osr2 have previously been implicated in palate formation, but little is known about how these molecular components interact within the greater regulatory network. Now, on  Contact inhibition of locomotion (CIL) is a fundamental regulatory mechanism that ensures correct cell movement and migration. During CIL, cells form transient contacts but the molecular nature of such contacts is unknown. In this issue, Roberto Mayor and colleagues (
Contact inhibition of locomotion (CIL) is a fundamental regulatory mechanism that ensures correct cell movement and migration. During CIL, cells form transient contacts but the molecular nature of such contacts is unknown. In this issue, Roberto Mayor and colleagues ( The immense power of Drosophila genetics has allowed invaluable insight into developmental biology. Despite these advances, a significant limitation has always been the lack of an efficient method for modifying select genetic loci. Now, on
The immense power of Drosophila genetics has allowed invaluable insight into developmental biology. Despite these advances, a significant limitation has always been the lack of an efficient method for modifying select genetic loci. Now, on  Proper control of cell size is vital to ensure the correct growth and development of any organism. The Myc family of proteins are key regulators of growth, but the mechanisms that control Myc protein levels are complex. Now, on
Proper control of cell size is vital to ensure the correct growth and development of any organism. The Myc family of proteins are key regulators of growth, but the mechanisms that control Myc protein levels are complex. Now, on  Dendrite complexity determines the functional properties of neurons and the overall connectivity of neuronal circuits. The bone morphogenetic protein (BMP) family is known to regulate a myriad of developmental processes, but the extent to which different members of the family are involved in dendrite growth remains unclear. In this issue (
Dendrite complexity determines the functional properties of neurons and the overall connectivity of neuronal circuits. The bone morphogenetic protein (BMP) family is known to regulate a myriad of developmental processes, but the extent to which different members of the family are involved in dendrite growth remains unclear. In this issue (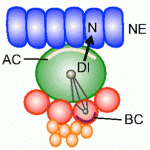 Stem cells and their progenitors are maintained within a microenvironment, termed the niche, but it is known that systemic signals originating outside the niche also affect stem cell and progenitor behavior. Here, Utpal Banerjee and colleagues review recent studies of nutritional effects on stem and progenitor cell maintenance and proliferation in Drosophila. See the Review article on p.
Stem cells and their progenitors are maintained within a microenvironment, termed the niche, but it is known that systemic signals originating outside the niche also affect stem cell and progenitor behavior. Here, Utpal Banerjee and colleagues review recent studies of nutritional effects on stem and progenitor cell maintenance and proliferation in Drosophila. See the Review article on p.  The proper formation and morphogenesis of dendrites is fundamental to the establishment of neural circuits in the brain. In this issue, Sidharth Puram and Azad Bonni review cell-intrinsic drivers of dendrite patterning and discuss how the characterization of such regulators advances our understanding of normal brain development and pathogenesis of diverse cognitive disorders. See the Review on p.
The proper formation and morphogenesis of dendrites is fundamental to the establishment of neural circuits in the brain. In this issue, Sidharth Puram and Azad Bonni review cell-intrinsic drivers of dendrite patterning and discuss how the characterization of such regulators advances our understanding of normal brain development and pathogenesis of diverse cognitive disorders. See the Review on p. 






 This post is part of a series on a day in the life of developmental biology labs working on different model organisms. You can read the introduction to the series
This post is part of a series on a day in the life of developmental biology labs working on different model organisms. You can read the introduction to the series  (8 votes)
(8 votes)