The people behind the papers – Alok Javali, Aritra Misra & Ramkumar Sambasivan
Posted by the Node Interviews, on 20 December 2017
Neuromesoderm progenitors are a population of stem cells that contribute to the neural tube and somite-forming paraxial mesoderm, and promote axial growth of the vertebrate embryo. In the latest issue of Development, a new paper addresses the transcriptional control of fate determination in this fascinating cell lineage. We caught up with co-first authors Alok Javali and Aritra Misra and their supervisor Ramkumar Sambasivan of the Institute for Stem Cell Biology and Regenerative Medicine in Bengaluru, India, to hear the story behind the work.

Ramkumar, can you give us your scientific biography and the main questions your lab is trying to answer?
RS It all started with the question how do skeletal muscle stem cells stay mitotically dormant and then wake up to proliferate and differentiate in order to repair damaged muscle. I had worked on this question for my Ph.D. thesis research and identified genes that play a role in return of dormant cells into active mitotic cycling. From addressing the cell biology of muscle stem cells, my interest broadened and I studied skeletal muscle development as well as regeneration in mice as a postdoctoral researcher. Currently, in my laboratory, we study the how the myogenic mesoderm develops and diversifies in vertebrates. Another major interest is on neural crest development, a new addition to my long-standing interest in muscle.
What is the current standing and future prospects for developmental biology research in India?
RS Developmental biologists make a small community in India and there is a need for expansion to derive the benefits of a strong community. However, things appear to move in the right direction. The number of developmental biologists in India has seen a significant spurt in the recent past, which I could gauge from the fantastic biennial meetings of the Indian Society of Developmental Biology. This augurs well.
Alok and Aritra – how did you come to join Ramkumar’s lab?
AJ I joined the graduate program of NCBS and InStem in 2013. Having prior interest in developmental biology, I did my lab rotation in Ramkumar’s lab. Being the first grad student in the lab, I had unique opportunity to be a part of multiple projects, which were still at their inception, ranging from studies on mesoderm patterning to mechanisms of fate choice in neural crest cells. This really excited me to join the lab for pursuing my PhD.
AM Prior to joining NCBS-inStem graduate program, I had trained in evolutionary biology and comparative morphology while studying zoology at bachelors and Masters level. This drove me to pursue PhD in either Evo-Devo or cell communication. Since Ramkumar’s work overlapped with my interest, I chose to join his lab.
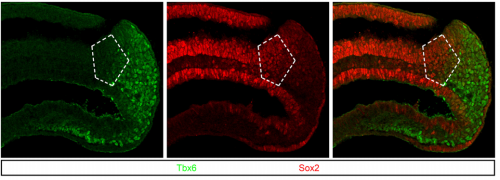
What’s so intriguing about neuromesodermal progenitors, and what were the questions you were hoping to answer in your study?
RS Neuromesodermal progenitors (NMPs) are key for axial growth in amniotes. I wonder about snakes and whales and the changes in the regulation of these progenitors that enabled such axially extended forms of life. Two different observations by Alok and Aritra took us in the direction of NMP biology and emphasized previous findings that Tbx6 is a key regulator of NMPs. The broad question is Tbx6 function in these progenitors. In this work we laid the foundation to tackle this question by addressing the timing of Tbx6 induction in NMP lineage.
Can you give us key results of the paper in a paragraph?
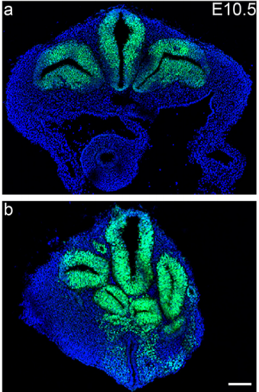
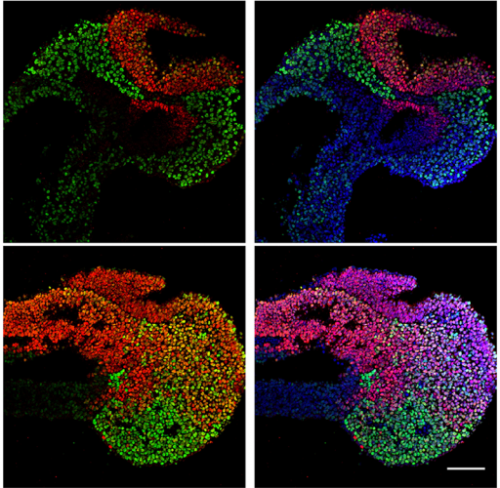
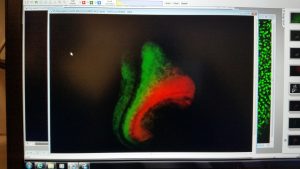
RS, AJ & AM We show that Tbx6 is expressed in a subset of neuromesoderm progenitors based on spatiotemporal expression pattern in the NMP niche in early mouse embryos. Co-expression of Tbx6 with Sox2, a neurogenic factor in this subset, as well as genetic tracing of Tbx6-expressing cells corroborated our findings of Tbx6 in NMPs. Expression of Tbx6 in NMPs provides a key missing link in favour of the current model in the field, which invokes Tbx6 as mesoderm switch in NMP lineage. In addition, we show that Tbx6 mutant mouse embryos have five developing spinal cords in the tail. This novel aspect of Tbx6 mutant phenotype in the tail further supports the model. Thus, our results strongly support the role of Tbx6 in promoting mesoderm fate choice of NMPs.
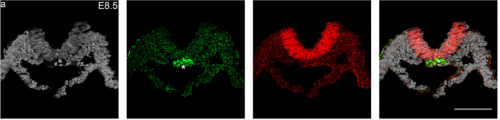
Your work supports the notion that Tbx6 as a bistable fate switch – how exactly do you think it plays this role?
AJ & RS Tbx6, being a transcription factor, functions by activating or repressing its transcriptional targets. Published literature already suggests that Tbx6 indirectly supresses the expression of Sox2, a key neurogenic factor, to promote mesodermal fate. An interesting aspect revealed by our study is the existence of a regulatory cell state, wherein Tbx6 and Sox2 are co-expressed. This indicates a biphasic function of Tbx6 in NMP lineage; early role in braking neural fate and subsequent function in mesoderm differentiation. We are currently trying to identify the spectrum of Tbx6 transcriptional targets to elucidate the mechanistic details of its function in NMP fate choice.
When doing the research, did you have any particular result or eureka moment that has stuck with you?
AJ: Looking at five distinct neural tubes arranged beautifully in a transverse section of mutant mouse embryo was special. Not everyday one gets to see such dramatic unreported phenotype. This was not really a Eureka moment as we were not anticipating this phenotype. This observation was accidental. We were trying to get transverse sections of a mutant embryo post in situ hybridization as the staining that we had performed was not very clear. We were not particularly looking into the tail region. Since the embryo is ‘C’ shaped, along with the transverse section of the interlimb region, we got the section of truncated tail of the mutant. And there we saw five distinct neural tubes.
AM: Tbx6 is viewed uniquely as paraxial mesoderm factor, but while analysing the confocal images of immunostaining in E8.5 mouse embryo tail regions, I observed specific subset co-expressing Tbx6 and Sox2, a neural marker, within NMP niche. This was indeed a eureka moment for me, because in mouse gastrulation, this expression precedes paraxial mesoderm formation in NMP lineage. This implied that there is more to Tbx6 function than had been appreciated.
And what about the flipside: any moments of frustration or despair?
AM: The most frustrating moment was trying to obtain serial transverse sections of a very specific anatomy of tiny E8.5 mouse embryos for control experiments to demonstrate that our novel Tbx6Cre transgenic line reports for Tbx6 faithfully. I had to ensure that I do not lose any section in the series. This experiment also provided crucial support to our observation of Tbx6 expression in the NMP niche.
AJ The most frustrating time was when I was trying to get non-oblique transverse section of primitive streak and node regions of e8.5 mouse embryos and saggital section of tailbud of e9.5 mouse embryos. The region of interest in both these cases is extremely small. Getting the ideal sections with all the necessary anatomical information to identify the region of interest took multiple attempts.
What are your career plans following this work?
AM: I want to continue in academia. My interest is to work on the evolution of bilateral symmetry and I want to start by looking into reptiles. But I will also be excited to explore opportunities in facilities such as mouse genome engineering facilities or Jackson Laboratories.
AJ: I am currently working on my PhD project where I am trying to understand the role of micro-RNAs in the development of another interesting cell type called neural crest. As much as I am attracted to academic research, along with it, I would also like to explore a career path in science education, which I believe is a need of the hour in India.
And what next for the Sambasivan lab?
I will address what next for my lab in the context of NMPs. We are curious about the evolutionary origin of NMPs and tracing this in the chordate lineage.
Finally, what do you like to do when you are not in the lab?
AM: I always wanted to be a historian and study the evolution of human civilization. I have not had the opportunity before joining for Ph.D., however, now I use my time out of lab to fulfil this dream. In the past 2 years I have researched on how evolution of Indian civilization can be traced to have more connections to the development of agrarian systems rather than changes in ruling dynasties. Recently I started to work on socio-economic changes in pre-British era driving changes in Indian demography. This began with an amazing book: Rise of Islam and the Bengal Frontiers by Richard Eaton. Currently I have started probing into the concept of Sacred Feminine and evolution of Egyptian society.
AJ: There are few things that I usually do. Swimming or running is my stress buster. I love to play Cricket (famous sport in India), though I don’t get much time for it anymore. I also love to read popular science books
RS: This used to be an easier question to answer.
Alok Javali, Aritra Misra, Karolis Leonavicius, Debalina Acharyya, Bhakti Vyas, Ramkumar Sambasivan. Co-expression of Tbx6 and Sox2 identifies a novel transient neuromesoderm progenitor cell state. 2017. Development,
This is #34 in our interview series. Browse the archive here.


 (17 votes)
(17 votes) (No Ratings Yet)
(No Ratings Yet)




 (3 votes)
(3 votes)
 The Manchester Fly Facility hosts
The Manchester Fly Facility hosts 

 The Arp2/3 complex is responsible for the assembly of branched actin filaments. Although its cellular functions are well understood, less is known about the consequence of its disruption in developing animals. To investigate the function of Arp2/3 in the skin, Metello Innocenti and colleagues (p.
The Arp2/3 complex is responsible for the assembly of branched actin filaments. Although its cellular functions are well understood, less is known about the consequence of its disruption in developing animals. To investigate the function of Arp2/3 in the skin, Metello Innocenti and colleagues (p.  Neuromesoderm progenitors (NMPs) are a distinct stem cell population that express both the mesoderm marker brachyury (T) and the neural marker Sox2, and are required for axial growth in vertebrates. They are capable of binary fate choice, differentiating as either neural or mesodermal progenitors. Tbx6, a downstream target of T, has been proposed to act as a fate switch, stimulating the mesodermal differentiation of NMPs, but the timing of this fate commitment is unclear. On p.
Neuromesoderm progenitors (NMPs) are a distinct stem cell population that express both the mesoderm marker brachyury (T) and the neural marker Sox2, and are required for axial growth in vertebrates. They are capable of binary fate choice, differentiating as either neural or mesodermal progenitors. Tbx6, a downstream target of T, has been proposed to act as a fate switch, stimulating the mesodermal differentiation of NMPs, but the timing of this fate commitment is unclear. On p.  Alveologenesis – the repeated division and growth of alveoli to expand the surface area in the lung – is one of the least understood stages of postnatal lung development. Defective alveologenesis results in bronchopulmonary dysplasia, a disorder often observed in premature infants. FGF signalling and the extracellular matrix (ECM) protein elastin are known to be important for alveologenesis, but the underlying mechanisms are poorly understood. On p.
Alveologenesis – the repeated division and growth of alveoli to expand the surface area in the lung – is one of the least understood stages of postnatal lung development. Defective alveologenesis results in bronchopulmonary dysplasia, a disorder often observed in premature infants. FGF signalling and the extracellular matrix (ECM) protein elastin are known to be important for alveologenesis, but the underlying mechanisms are poorly understood. On p.  Although mammals have internal mechanisms for regulating body temperature, the vast majority of organisms are ectotherms, meaning that their body temperature is dictated by the external environment. Temperature fluctuations significantly affect cellular homeostasis, but the molecular mechanisms underlying these effects are currently poorly understood. Multiple lines of evidence suggest that microtubules are sensitive to cold, with suboptimal temperatures causing their disassembly. Christian Lehner and colleagues (p.
Although mammals have internal mechanisms for regulating body temperature, the vast majority of organisms are ectotherms, meaning that their body temperature is dictated by the external environment. Temperature fluctuations significantly affect cellular homeostasis, but the molecular mechanisms underlying these effects are currently poorly understood. Multiple lines of evidence suggest that microtubules are sensitive to cold, with suboptimal temperatures causing their disassembly. Christian Lehner and colleagues (p.  Claudio Stern is the J. Z. Young Professor of Anatomy at University College London (UCL), UK. His lab studies the processes that regulate patterning and cell diversity in the early embryos of vertebrates, mostly in chick. Claudio, an elected fellow of the Royal Society, the UK Academy of Medical Sciences, and the Latin-American Academy of Medical Sciences, was awarded the 2006 Waddington Medal by the British Society of Developmental Biology, and he also served as President of the International Society for Developmental Biology (ISDB) from 2010-2013. At the 18th Congress of the ISDB (Singapore, June 2017), Claudio was awarded the ISDB’s Ross Harrison Prize, which recognises an individual’s outstanding contributions to developmental biology. We met with Claudio to ask him more about his career, his thoughts on the field, and his advice for early career researchers. Read the Spotlight article on p.
Claudio Stern is the J. Z. Young Professor of Anatomy at University College London (UCL), UK. His lab studies the processes that regulate patterning and cell diversity in the early embryos of vertebrates, mostly in chick. Claudio, an elected fellow of the Royal Society, the UK Academy of Medical Sciences, and the Latin-American Academy of Medical Sciences, was awarded the 2006 Waddington Medal by the British Society of Developmental Biology, and he also served as President of the International Society for Developmental Biology (ISDB) from 2010-2013. At the 18th Congress of the ISDB (Singapore, June 2017), Claudio was awarded the ISDB’s Ross Harrison Prize, which recognises an individual’s outstanding contributions to developmental biology. We met with Claudio to ask him more about his career, his thoughts on the field, and his advice for early career researchers. Read the Spotlight article on p.  The 10th FASEB meeting ‘The TGFβ Superfamily: Signaling in Development and Disease’ took place in Lisbon, Portugal, in July 2017. Here,
The 10th FASEB meeting ‘The TGFβ Superfamily: Signaling in Development and Disease’ took place in Lisbon, Portugal, in July 2017. Here,  Cell-cell fusion is essential for fertilization and organ development. Dedicated proteins known as fusogens are responsible for mediating membrane fusion. However, until recently, these proteins either remained unidentified or were poorly understood at the mechanistic level. Here,
Cell-cell fusion is essential for fertilization and organ development. Dedicated proteins known as fusogens are responsible for mediating membrane fusion. However, until recently, these proteins either remained unidentified or were poorly understood at the mechanistic level. Here,  While the principles that establish and regulate pluripotency have been well defined in the mouse, it has been difficult to extrapolate these insights to the human system due to species-specific differences and the distinct developmental identities of mouse versus human embryonic stem cells. In their Review,
While the principles that establish and regulate pluripotency have been well defined in the mouse, it has been difficult to extrapolate these insights to the human system due to species-specific differences and the distinct developmental identities of mouse versus human embryonic stem cells. In their Review,