Using the mouse to model human disease: increasing validity and reproducibility
Posted by Disease Models and Mechanisms, on 3 February 2016
ABSTRACT
Experiments that use the mouse as a model for disease have recently come under scrutiny because of the repeated failure of data, particularly derived from preclinical studies, to be replicated or translated to humans. The usefulness of mouse models has been questioned because of irreproducibility and poor recapitulation of human conditions. Newer studies, however, point to bias in reporting results and improper data analysis as key factors that limit reproducibility and validity of preclinical mouse research. Inaccurate and incomplete descriptions of experimental conditions also contribute. Here, we provide guidance on best practice in mouse experimentation, focusing on appropriate selection and validation of the model, sources of variation and their influence on phenotypic outcomes, minimum requirements for control sets, and the importance of rigorous statistics. Our goal is to raise the standards in mouse disease modeling to enhance reproducibility, reliability and clinical translation of findings.
Model validity: making sure the mouse is right
It seems an obvious point, but the model used should be appropriate for the question being addressed. An ideal disease model accurately mimics the human condition, genetically, experimentally and/or physiologically. At DMM, we require that the similarities to human disease be rigorously validated, preferably by proof-of-principle experiments demonstrating response to treatment. The controversial study described above compared microarray gene expression data from humans and mice. In one example, data from human blunt-trauma patients were analyzed together with data from a mouse inbred strain that had been exsanguinated. Losing a large amount of blood does not equate to blunt trauma, and so this could be perceived as comparing apples to oranges. Furthermore, inbred mouse strains represent limited genetic diversity and might not reflect the responses generated in a genetically polymorphic human population. The conclusions drawn in this manuscript did not take into account these potential sources of experimental differences between the mouse and human, and raise the possibility of bias in data analysis.
In a different study, a mouse model was reported to display the key motor symptoms seen in humans with amyotrophic lateral sclerosis (ALS) (Wegorzewska et al., 2009). On the basis of this, the model was used in preclinical studies and promising drug candidates were tested in clinical trials; however, these drugs ultimately failed in humans (Perrin, 2014). It was then shown that this mouse is a poor genetic and phenotypic model of the human condition (Hatzipetros et al., 2014). This example illustrates how relevance to the human disease being studied, supported by strong data to validate the use of the model, is crucial for clinical translation. Humanized models – mice expressing human transgenes or engrafted with functional human cells or tissues – can provide important tools to bridge the gap between animals and humans in preclinical research.
References
- Bersell, K., Choudhury, S., Mollova, M., Polizzotti, B. D., Ganapathy, B., Walsh, S., Wadugu, B., Arab, S. and Kuhn, B. (2013). Moderate and high amounts of tamoxifen in alphaMHC-MerCreMer mice induce a DNA damage response, leading to heart failure and death. Dis. Model. Mech. 6, 1459-1469. doi:10.1242/dmm.010447
- Collins, F. S. and Tabak, L. A. (2014). Policy: NIH plans to enhance reproducibility. Nature 505, 612-613. doi:10.1038/505612a
- Fenwick, N., Griffin, G. and Gauthier, C. (2009). The welfare of animals used in science: how the “Three Rs” ethic guides improvements. Can. Vet. J. 50, 523-530.
- Han, H. J., Allen, C. C., Buchovecky, C. M., Yetman, M. J., Born, H. A., Marin, M. A., Rodgers, S. P., Song, B. J., Lu, H.-C., Justice, M. J. et al. (2012). Strain background influences neurotoxicity and behavioral abnormalities in mice expressing the tetracycline transactivator. J. Neurosci. 32, 10574-10586. doi:10.1523/JNEUROSCI.0893-12.2012
- Hatzipetros, T., Bogdanik, L. P., Tassinari, V. R., Kidd, J. D., Moreno, A. J., Davis, C., Osborne, M., Austin, A., Vieira, F. G., Lutz, C. et al. (2014). C57BL/6J congenic Prp-TDP43A315T mice develop progressive neurodegeneration in the myenteric plexus of the colon without exhibiting key features of ALS. Brain Res. 1584, 59-72. doi:10.1016/j.brainres.2013.10.013
- Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. and Altman, D. G. (2010). Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 8, e1000412. doi:10.1371/journal.pbio.1000412
- Lexow, J., Poggioli, T., Sarathchandra, P., Santini, M. P. and Rosenthal, N. (2013). Cardiac fibrosis in mice expressing an inducible myocardial-specific Cre driver. Dis. Model. Mech. 6, 1470-1476. doi:10.1242/dmm.010470
- Nguyen, T. L. A., Vieira-Silva, S., Liston, A. and Raes, J. (2015). How informative is the mouse for human gut microbiota research? Dis. Model. Mech. 8, 1-16. doi:10.1242/dmm.017400
- Perrin, S. (2014). Preclinical research: make mouse studies work. Nature 507, 423-425. doi:10.1038/507423a
- Rosenthal, N. and Brown, S. (2007). The mouse ascending: perspectives for human-disease models. Nat. Cell Biol. 9, 993-999. doi:10.1038/ncb437
- Seok, J., Warren, H. S., Cuenca, A. G., Mindrinos, M. N., Baker, H. V., Xu, W., Richards, D. R., McDonald-Smith, G. P., Gao, H., Hennessy, L. et al. (2013). Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA 110, 3507-3512. doi:10.1073/pnas.1222878110
- Simon, M. M., Greenaway, S., White, J. K., Fuchs, H., Gailus-Durner, V., Wells, S., Sorg, T., Wong, K., Bedu, E., Cartwright, E. J. et al. (2013). A comparative phenotypic and genomic analysis of C57BL/6J and C57BL/6N mouse strains. Genome Biol. 14, R82. doi:10.1186/gb-2013-14-7-r82
- Simpson, E. M., Linder, C. C., Sargent, E. E., Davisson, M. T., Mobraaten, L. E. and Sharp, J. J. (1997). Genetic variation among 129 substrains and its importance for targeted mutagenesis in mice. Nat. Genet. 16, 19-27. doi:10.1038/ng0597-19
- Takao, K. and Miyakawa, T. (2015). Genomic responses in mouse models greatly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA 112, 1167-1172. doi:10.1073/pnas.1401965111
- Wegorzewska, I., Bell, S., Cairns, N. J., Miller, T. M. and Baloh, R. H. (2009). TDP-43 mutant transgenic mice develop features of ALS and frontotemporal lobar degeneration. Proc. Natl. Acad. Sci. USA 106, 18809-18814. doi:10.1073/pnas.0908767106



 (2 votes)
(2 votes) (No Ratings Yet)
(No Ratings Yet)




 The Hedgehog (Hh) signalling pathway is one of the key regulators of metazoan development. Here,
The Hedgehog (Hh) signalling pathway is one of the key regulators of metazoan development. Here, 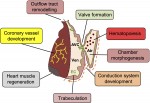 Endocardial cells are cardiac endothelial cells that line the interior of the heart tube. Here,
Endocardial cells are cardiac endothelial cells that line the interior of the heart tube. Here, 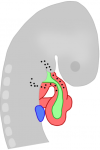 Nadia Rosenthal and co-workers discuss the role of cardiac fibroblasts in scarring and regeneration, as well as how these cells are specified during development and the unique characteristics that define them. See the Review on p.
Nadia Rosenthal and co-workers discuss the role of cardiac fibroblasts in scarring and regeneration, as well as how these cells are specified during development and the unique characteristics that define them. See the Review on p.  The award is named after
The award is named after  The BSDB is proud to announce the first winner of the Cheryll Tickle Medal, which will be awarded at the 2016 BSDB-BSCB Spring meeting in Warwick to
The BSDB is proud to announce the first winner of the Cheryll Tickle Medal, which will be awarded at the 2016 BSDB-BSCB Spring meeting in Warwick to 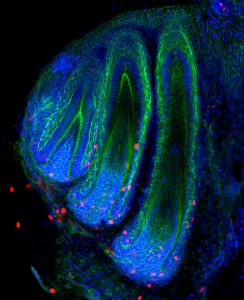
 –
–
 -We featured this
-We featured this