Homeward bound
Posted by Kim Cooper, on 10 May 2012
Posted by Kim Cooper, on 10 May 2012
Posted by ama, on 10 May 2012
It is a time of gene counting, mapping, function guessing in a narrow way: a gene for this or a gene for that. If one reads the indexes of journals one might believe that by adding genes one gets an organism. It is for this reason that reading a book like “Wetware” by Dennis Bray is a stimulating exercise and a gift to the mind. Dennis Bray is a pioneer of what some of us call Systems Biology, not the one related to genomics and proteomics but ‘the other one’, the one that aims to understand Biology from the perspective of organized assemblies of molecules that perform specific tasks. For a long time he has been interested in the idea that proteins perform computations and this notion lies at the heart of his view of life. In Wetware, distilling thoughts gathered over a career probing the engineering of cells, Dennis Bray presents a personal view of a cell as a biomolecular device that senses its environment, whether a specific milieu or other cells, and through a computation of sorts, responds by changing itself and the environment. Sure, one can read his primary papers on many of the topics developed here but there is beauty and insight in the way the argument is put together in the book, its accessibility and the inspired way in which notions of what is life flow- from the reactions of Stentor to human intervention to the information processing capacities of biochemical networks.
The book can be construed as a popular work but this will betray what I perceive as an ambition to open ways of thinking which, if taken seriously, should lead many cell and developmental biologists to pose questions about calculations and dynamics rather than about genes and their function as revealed by more or less complex genetic experiments.
Dennis Bray has written before an excellent book on cell movement (Cell movement: from molecules to motility) where he explores in detail some of the issue raised in Wetware and which has guided many of us on our trips into the structure and function of the cytoskeleton. But Wetware has a lot more because here he develops a panoramic view of Life from the inside of the cell. The pervasive metaphor of the computer will irritate some computational buffs but this will only reveal that they miss the point: a cell is not a piece of hardware in the human sense and the book makes this clear that any analogy one might draw will have to bear in mind the peculiar characteristics of the molecular make up of a cell. One example: the notion that Diffusion of molecules within a cell might be a correlate of the cables that bring together the pieces of a machine, is a far reaching notion that deserves to be explored, as is the implicit notion that a signature of life is the molecular make up of sensory experiences.
I shall not go on about the book because the main object of this posting is to encourage you to read it. I believe that it should be required reading for graduate students in cell and developmental biology. Towards the end there is a quotation from Andre Gide which is an enticement to act: “one doesn’t discover new lands without consenting to lose sight of the shore for a very long time”. Wetware is a gust of wind that should encourage you to sail into the current of the unknown, without fear, with the imagination that is denied by the current interest in publications rather than Discovery.
Posted by Barbara Jennings, on 9 May 2012
Closing Date: 15 March 2021
Full Time : The appointment will be on UCL Grade 6. The salary range will be £25,927 – £30,384 per annum., inclusive of London Allowance.
The successful applicant will use molecular biology, Drosophila genetics and immunohistochemistry to study the molecular mechanisms regulating gene expression during development. They will also be responsible for maintenance of the laboratory’s collection of Drosophila stocks and for supporting the day-to-day running of the laboratory.
The post is intially funded till September 2013.
The candidate must hold a BSc (Hon) in biological sciences or equilvelant. Experience in basic molecular biology techniques along with good organisational and communication skills are essential for this post. While experience working with Drosophila would be an advantage, training will be provided as required.
For further details about the vacancy and how to apply on line please go to http://www.ucl.ac.uk/hr/jobs/ and search on Reference Number 1250187.
If you have any queries regarding the vacancy or application process please contact Cancer Vacancies Team e-mail: vacancies@cancer.ucl.ac.uk
Please quote the Reference Number with all correspondence.
Closing Date: 23/5/2012
We particularly welcome applications from black and minority ethnic candidates as they are under-represented within UCL at this level.
Posted by the Node, on 9 May 2012
Winners of the student poster competition at the annual BSDB meeting get an amazing prize: they receive a registration for the annual SDB meeting in North America. And, vice versa, winners of the SDB poster competition get to go to the BSDB meeting in the UK.
Sadly, the winner of last year’s SDB poster competition, Tracy Chong, was unable to make it to the UK for the BSDB meeting last month. She did write a great post on the Node about her work, so you can read that to find out what her poster was about.
In Tracy’s place, Steffen Biechele attended the BSDB meeting. He was a runner-up in the poster competition, and not at all expecting to be attending the BSDB meeting until he was contacted a few weeks beforehand!
As the substitute SDB poster winner, Steffen interviewed this year’s BSDB poster winner. The poster award went to Stephen Fleenor, a PhD student in Jo Begbie’s lab at Oxford University.
Interview:
Steffen Biechele: You won yesterday’s poster competition. Which lab do you work for?
Stephen Fleenor: I work in Jo Begbie’s lab. I’ve been there as a PhD student for about six months. Prior to that I did a rotation stint for five months, so I’ve been there about a year in total.
SB: Wow, you won a post award after six months – that’s fantastic.
SF: I’m equally surprised!
SB: What was the poster about?
SF: It was largely an introduction to the system that we’re studying, and the phenotype of a knockdown that I did. I knocked down a molecule known to be a catalytic regulator of G-protein signaling, but its regulation in this manner hasn’t been characterized in our system.
SB: What system is that?
SF: Developmental cranial sensory ganglia in the chick embryo. We’re looking at the generation of neuroblasts and migration of those neuroblasts toward the site of the ganglia proper. We’re trying to figure out what’s guiding them, what’s cueing them to begin differentiation and guiding the kinetics of their migration.
SB: Have you presented a poster at a meeting before?
SF: No. This is my first conference, as well. This has all been a whirlwind of excitement!
SB: What’s next?
SF: Well, apparently I’m going to the SDB Meeting in Montreal!
At the SDB meeting, Stephen is, in turn, going to try to interview the SDB poster winner, so we should hear from him again in a few months. The poster winners at the SDB meeting are announced at the very end of the conference, so the logistics are a bit more difficult than at the BSDB, where there is still half a day of talks after the announcements. Consider it an experiment. As it goes with all experiments, we hope it will be successful, but we won’t know if it works until we try!
Posted by Elisa Martí, on 8 May 2012
Closing Date: 15 March 2021
PhD student position at the IBMB-CSIC, Barcelona
Laboratory of Developmental Neurobiology
http://www.ibmb.csic.es/index.php?pg=laboratorio&idLaboratorio=18&tab=lab_home
We are looking for: Enthusiastic researchers with a BSc or Masters Degree in biomedical sciences with interest in Developmental Neurobiology Good academic records are required Good spoken and written command of English
We offer: A highly multidisciplinary and competitive training programme in biomedical research. Access to state-of-the-art infrastructures.
The selected candidate will investigate the role of extracellular signals and the genetic networks that control cell numbers, cell identity and cell shape changes during the embryonic development of the neural tube, using live-imaging, cell- and molecular biology in two animal model chick and zebrafish embryos
Those interested please send CV, a cover letter justifying the interest of the applicant in the project to emgbmc@ibmb.csic.es
Posted by Katherine Brown, on 8 May 2012
 (The following editorials all appear in Development vol. 139, issue 11. Image: the very first Development cover from 1987, with images from a paper by Jim Smith, our second Editor in Chief )
(The following editorials all appear in Development vol. 139, issue 11. Image: the very first Development cover from 1987, with images from a paper by Jim Smith, our second Editor in Chief )
When I arrived at Development in late 2011, I realised that the journal was about to celebrate its 25th birthday. Or perhaps its 59th, depending on how you look at things. The journal that is now Development was founded in 1953 as the Journal of Embryology and Experimental Morphology (JEEM). But in 1987, in a bold move by The Company of Biologists and the journal’s editors, it was rebranded, restyled and relaunched with a succinct one-word title and a mission to publish the best papers in the burgeoning field of developmental biology. That field has blossomed, matured and expanded over the last 25 years, and today’s cutting edge is remarkably far from those early days of molecular genetics.
We have been extremely fortunate to have had three visionary and dedicated Editors in Chief over the years: Chris Wylie, who oversaw the transition from JEEM to Development and remained at the helm until 2002; Jim Smith, who steered the ship from 2003 to 2009; and Olivier Pourquié, who now controls the tiller. And it has not been all smooth sailing over these 25 years, with challenges ranging from remaining a high-quality community journal in the face of stiff competition, through the transition from a paper-based to an online world, to the new scientific directions in which the field is moving. To look back on the trials and triumphs, and to contemplate the future, we have invited all three Editors in Chief to share their memories and thoughts on their time in charge, presented in the following editorials.
We are also celebrating our quarter-century by revisiting some of Development’s classic papers. It’s been very interesting to look back over the articles published in our pages; although the approaches and techniques may have changed drastically, many of the fundamental questions remain the same. Perhaps it’s also comforting to see that, while we have answered many problems, there’s still plenty to discover. Over the coming months, we will be showcasing some of Development’s most influential papers in our Spotlight section, so look out to see whether your favourite paper makes it in! Finally, we hope to be able to celebrate in person with some of you later in the year. Plans are underway for a one-day symposium to be held in Cambridge in the autumn; more details will be announced shortly.
For now, though, we invite you to step back in time and to follow Chris, Jim and Olivier through Development’s birth, youth and coming of age, and to look forward to the next 25 years of what I’m sure you will all agree is still the fascinating and fast-moving field of developmental biology.
Posted by Seema Grewal, on 8 May 2012
Here are the research highlights from the current issue of Development:
During the development of many animal organs, mesenchymal cells co-ordinately polarize to form epithelial sheets or tubes. In vitro studies have suggested that the extracellular matrix component laminin functions as a polarity cue during this mesenchymal to epithelial transition. Here (p. 2050), Jeffrey Rasmussen and colleagues provide in vivo evidence for laminin’s involvement in polarization by studying the development of the C. elegans pharynx, an epithelial tube that forms from pharyngeal precursor cells (PPCs). The researchers show that cell fate regulators, including the transcription factor PHA-4, cause the PPCs to form a bilaterally symmetric, rectangular array of cells called the ‘double plate’. PPC polarization and apical PAR localisation begin in the double plate cells, which then undergo apical constriction to form a cylindrical cyst. Notably, laminin provides an essential cue that orients the apical localisation of the PAR-3 complex in the double plate but not in the developing C. elegans intestine. Thus, the researchers conclude, laminin is an early polarizing cue for some but not all epithelia.
During mitosis, the spindle assembly checkpoint (SAC) coordinates proper bipolar chromosome attachment with the anaphase-promoting complex/cyclosome (APC/C)-mediated destruction of cyclin B1 that is required for anaphase onset, thereby avoiding chromosome mis-segregation and aneuploidy. The generation of a Mad2-based signal at kinetochores is central to current models of SAC-based APC/C inhibition: during mitosis, the kinetochores of polar chromosomes (non-aligned bivalents), which are at the greatest risk of mis-segregating, preferentially recruit Mad2, which couples SAC activation to aneuploidy risk. Paradoxically, although an SAC operates in mammalian oocytes, meiosis I is notoriously error prone. Two papers in this issue investigate this long-standing puzzle.
On p. 1947, Keith Jones and colleagues examine the timing of Mad2 loss from mouse oocyte kinetochores. The formation of stable kinetochore-microtubule attachments in mid-prometaphase (3-4 hours before anaphase), they report, coincides with the loss of Mad2 from the kinetochores and the start of APC/C-mediated cyclin B1 destruction. Thus, SAC inhibition of the APC/C ends in mid-prometaphase. However, in a third of oocytes examined, this timing did not coincide with bivalent congression. Notably, the presence of non-aligned bivalents (which were weakly positive for Mad2, under less tension than congressed bivalents, and in the process of establishing correct bi-orientation) did not affect the time between APC/C activation and anaphase onset, and non-aligned bivalents sometimes persisted until anaphase, resulting in homologue non-disjunction. Thus, in oocytes, a few erroneous microtubule-kinetochore attachments may go uncorrected because they do not generate a sufficient SAC ‘wait anaphase’ signal to inhibit the APC/C.
On p. 1941, Liming Gui and Hayden Homer investigate how the SAC responds to polar chromosomes during meiosis I in oocytes. They show that Mad2 is not preferentially recruited to the kinetochores of the rare polar chromosomes that occur in wild-type mouse oocytes or to the kinetochores of the more abundant polar chromosomes that are found in oocytes depleted of the kinesin-7 motor CENP-E. Moreover, in CENP-E-depleted oocytes all the kinetochores eventually become devoid of Mad2, even though the capacity of the chromosomes to form stable attachments to the spindle is severely compromised. These and other findings suggest that SAC signalling is uncoupled from chromosomal position during meiosis I in mouse oocytes, thereby predisposing oocytes to aneuploidy.
The regulation of gene expression by transcription factors drives cell fate specification during development. In Arabidopsis, transcription factors encoded by four classes of homeotic genes control floral organ identity. The A-class gene APETALA2 (AP2) promotes sepal and petal identity and restricts expression of the C-class gene AGAMOUS (AG), which specifies stamen and carpel identity, but how does AP2 perform these functions? On p. 1978, Xuemei Chen and co-workers report that AP2 recognises and acts through an AT-rich sequence element. The researchers show that AP2R2 (one of two DNA-binding domains in AP2) binds a non-canonical AT-rich target sequence in vitro and that the presence of this sequence in the second intron of AG is important for the restriction of AG expression in vivo. Other experiments indicate that AP2 directly regulates AG expression in young flowers through this sequence element, which is highly conserved in Brassicaceae. Together, these findings shed light on the molecular mechanism underlying AP2 action and provide a missing link in the mechanisms controlling flower development.
Biological tubes composed of polarized epithelial cells perform many functions in multicellular organisms. The establishment and maintenance of epithelial polarity depend on polarized trafficking of membrane components to the apical or basolateral domains of epithelial cells, but exactly how trafficking regulates epithelial polarity is unclear. In this issue, two papers describe a new and unexpected role for the post-Golgi vesicle coat clathrin and its adaptor AP-1 in apical sorting and lumen formation in the C. elegans intestine.
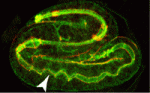
On p. 2061, Grégoire Michaux and colleagues report that the clathrin adaptor AP-1 is required for epithelial polarity in the C. elegans intestine. Depletion of AP-1 subunits does not affect the establishment of epithelial polarity or the formation of the intestinal lumen, the researchers report. However, they show that AP-1 is essential for the apical localisation of the oligopeptide transporter PEPT-1 and the polarity proteins PAR-6 and CDC-42, and for the basolateral distribution of the monocarboxylate transporter SLCF-1. They also show that AP-1 depletion triggers the formation of ectopic apical lumens between intestinal cells along the lateral membranes later during embryogenesis.
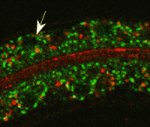
On p. 2071, Verena Gobel and colleagues perform an unbiased RNAi screen for apicobasal polarity and tubulogenesis defects in the C. elegans intestine and identify clathrin and AP-1 as being required for apical polarity and lumen formation. The researchers show that clathrin/AP-1-mediated polarized transport co-operates with a sphingolipid-dependent apical sorting process. Furthermore, they report, the depletion of clathrin, AP-1 or glycosphingolipid biosynthetic enzymes causes a set of apical membrane proteins (including PAR-6) to mislocalise basolaterally and generate ectopic lateral lumens. Finally, they show that clathrin-coated and sphingolipid-rich vesicles assemble at polarized plasma membrane domains in a co-dependent and AP-1-dependent manner.
Together, these findings suggest that clathrin/AP-1 controls both basolateral and apical sorting, an unexpected finding given that, until now, clathrin and its AP-1 adaptor had been thought to regulate only basolateral sorting in mammalian epithelia. Importantly, these findings indicate that this newly discovered clathrin/AP-1 function in apical sorting is required to regulate epithelial polarity in vivo in a tubular epithelium and that the clathrin/AP-1 apical sorting pathway converges with a sphingolipid-dependent apical trafficking path.
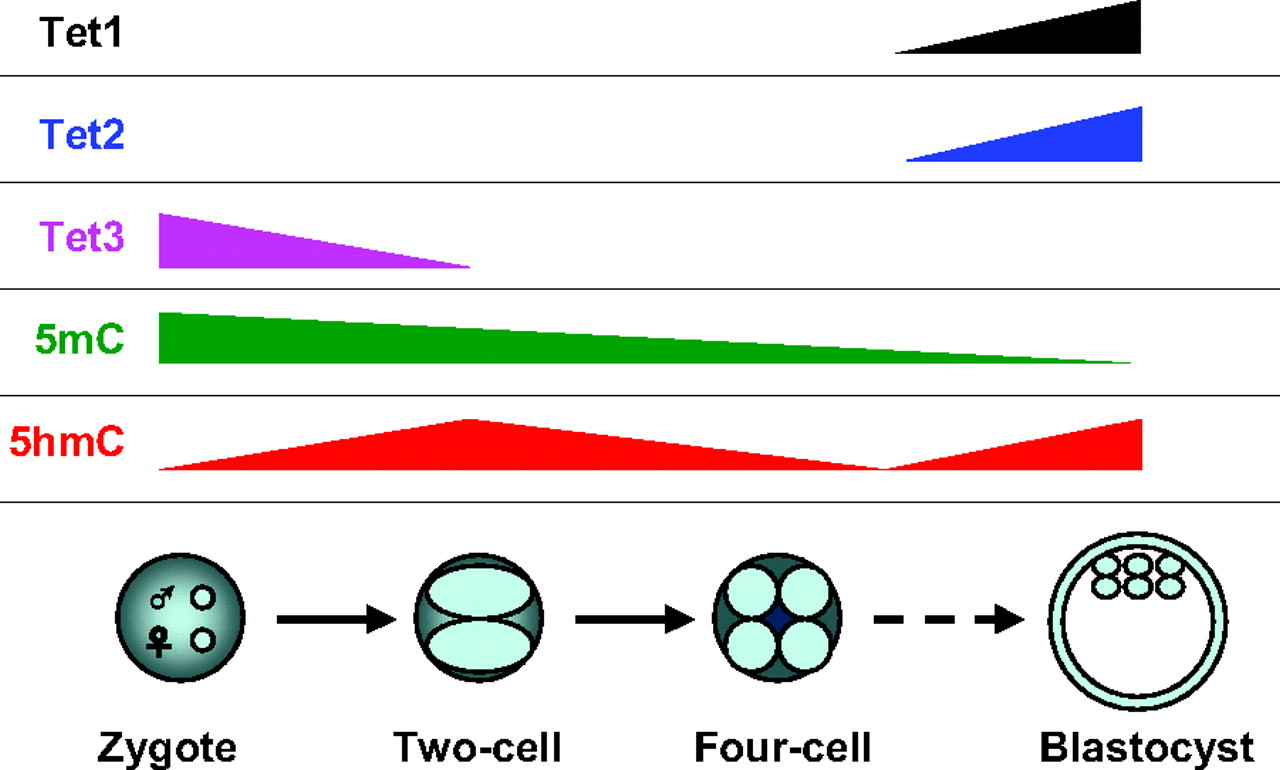
Over the past few decades, DNA methylation at the 5-position of cytosine (5-methylcytosine, 5mC) has emerged as an important epigenetic modification that plays essential roles in development, aging and disease. In this Issue, Tan and Shi provide an overview of the role of Tet family proteins and 5hmC in development and cancer. See the Primer article on p. 1895
The Journal of Embryology and Experimental Morphology (JEEM) was founded in 1953, but it wasn’t until 1987, in a bold move by The Company of Biologists and the journal’s editors, that the journal was rebranded, restyled and relaunched as the journal we now know as Development.
We’ll be celebrating celebrating our quarter-century throughout the year, but to kick-start the celebrations we’ve invited past and present Editors in Chief of Development to share their memories and thoughts on their time in charge.
See the Editorials from Chris Wylie, Jim Smith and Olivier Pourquie.
Posted by Guillermo Oliver, on 5 May 2012
Closing Date: 15 March 2021
Postdoctoral Positions, St. Jude Children’s Research Hospital, Memphis, TN, USA
Postdoctoral Positions in Molecular and Developmental Biology are available to study the cellular and molecular mechanisms controlling vertebrate organogenesis. We are particularly interested in the development of the lymphatic vasculature and the visual system. We use available mouse models to understand the mechanisms regulating these processes in normal and pathological conditions.
Highly motivated individuals who recently obtained a PhD. or MD degree and have a strong background in molecular and developmental biology are encouraged to apply. Interested individuals should send their curriculum vitae, a brief description of their research interests, and the names of three references to:
Guillermo Oliver, Ph.D
guillermo.oliver@stjude.org
Department of Genetics
St. Jude Children’s Research Hospital
332 N. Lauderdale
Memphis, TN 38105 USA
www.stjude.org/departments/oliver.htm
Posted by Treasa Creavin, on 3 May 2012
Abstract & Bursary deadline: 11 May | Registration deadline: 31 May
Now in its third year, this Wellcome Trust meeting will focus on the biology of sub-nuclear structures including the nucleolus, cajal and PML bodies. These structures have key roles in normal and diseased cells and they interact in a dynamic way. A fundamental understanding of these sub-nuclear structures can lead to advances in our understanding of infectious disease and cancer.
The conference will bring together cell biologists, microbiologists and virologists working on normal and tumor cells and researchers interested in how these structures are affected by infectious and acquired disease across all eukaryotic systems.
Scientific Organisers:
Susan Baserga Yale University, USA
Julian Hiscox University of Leeds, UK
David Matthews University of Bristol, UK
Brian McStay NUI Galway, Ireland
Invited Speakers include:
Susan Baserga Yale University, USA
Richard Gardner University of Washington, USA
Ingrid Grummt Deutsches Krebsforschungszentrum, Germany
Ross Hannan Peter MacCallum Cancer Centre, Australia
Valerie Lallemand-Breitenbach Hopital Saint-Louis, France
Angus Lamond University of Dundee, UK
Greg Matera University of North Carolina, USA
Brian McStay NUIG, Ireland
Karla Neugebauer Max Planck Institute, Germany
Craig S. Pikaard Indiana University, USA
Michael Taliansky The James Hutton Institute, UK
David Tollervey University of Edinburgh, UK
Adrian Whitehouse University of Leeds, UK
Abstract submission is strongly encouraged as several talks will be selected from abstracts.
For more information: https://registration.hinxton.wellcome.ac.uk/display_info.asp?id=294
Posted by the Node, on 3 May 2012
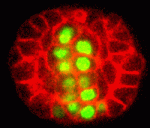
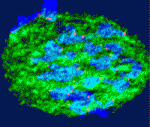
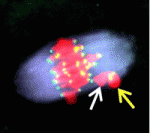
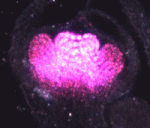
One of the covers of Development this year is reserved for an image from the International Course on Developmental Biology, UNAB, Quintay-Chile. This course ran from 5-17 January in Chile. You can read a report from the course on the Node.
Which image will get the coveted cover spot is up to you to decide. Choose your favourite before May 21st, noon GMT. There are 8 images to choose from. Click any of them to see a bigger version.