Zebrahub: Illuminating Zebrafish Embryogenesis Through Light-Sheet Microscopy and scRNA Sequencing.
Posted by Loic Royer, on 25 July 2023
What is Zebrahub?
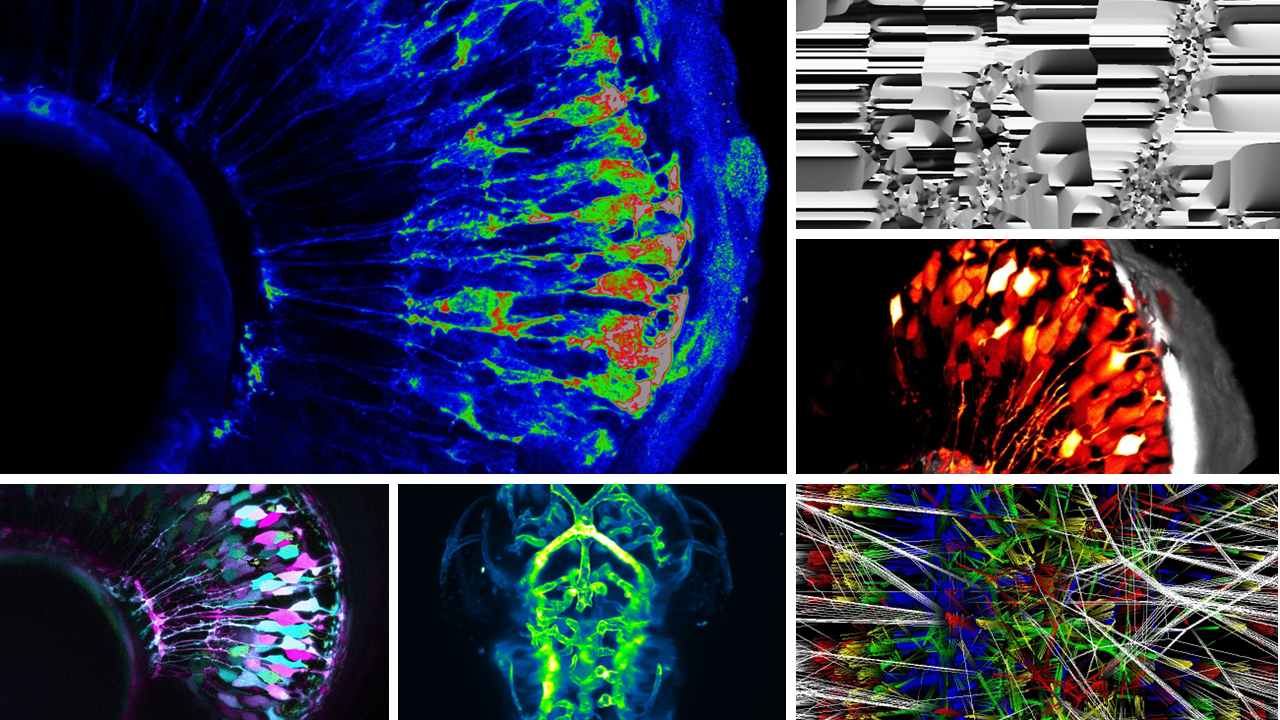
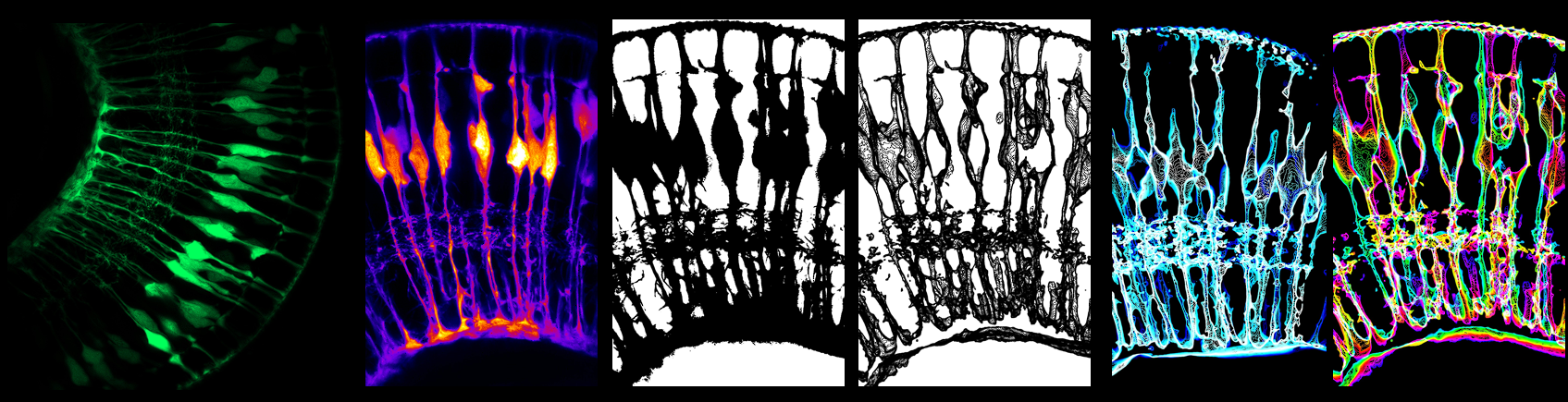
Zebrahub is a web-based platform that offers an integrated approach to studying and analyzing cellular lineages during zebrafish embryogenesis at both transcriptional and spatiotemporal levels. It combines single-cell sequencing time course data with lineage reconstructions facilitated by light-sheet microscopy. The scRNAseq gene expression across tissues and time points can be directly visually explored in a web browser. Additionally, researchers can navigate the complex cellular flows and lineages derived from light-sheet microscopy data using Zebrahub’s online web viewer and desktop interactive tool, enabling exploration of whole timelapse datasets and virtual fate mapping experiments.
What inspired the development of Zebrahub?
When we initiated the project, it was clear that the community was missing a database combining systematic live-imaging-based cell lineages with single-cell sequencing during development. Traditionally these approaches have been used separately, thereby limiting the full understanding of development. We wanted to use the most advanced microscopy and sequencing techniques available to establish a multimodal baseline of zebrafish development.
How can scientists utilize Zebrahub in their research?
Many important discoveries in developmental biology originated from observations; therefore, from the beginning, we wanted to have a fully interactive website to empower scientists by making our data accessible and, more importantly, directly explorable, even to scientists without coding skills. Thus, concerning our terabyte-scale high-resolution light-sheet imaging time-lapse dataset, we provide both an neuroglancer-based interactive viewer for entire multi-color time-lapse datasets and a napari plugin to perform in silico fate mapping experiments. Similarly, we allow researchers to effortlessly search for specific gene expressions and the emergence of cell types using CZI’s cell-by-gene platform. Our commitment to accessibility led us to invest substantial effort in ensuring that our data, code, and blueprints are readily available and open source. By doing so, we anticipate an immediate impact on a wide-ranging community, spanning disciplines such as developmental biology, single-cell biology, advanced microscopy, computer vision, tissue morphodynamics, and computational biology.
Who are the people behind this resource?

Supported by the CZ Biohub San Francisco (CZB-SF), Zebrahub is led by Merlin Lange, senior scientist in the lab of Loic Royer, and results from an interdisciplinary collaboration with CZB’s Data Science and Genomics Platforms, with key contributors among others: Angela Oliveira Pisco, Norma Neff, and Alejandro Granados. Overall, Zebrahub has more than 30 collaborators at CZB-SF collaborating institutions (see picture above). Many are early-career scientists who played a key role in the project. In the following, these young scientists introduce themselves and their contributions:
Shruthi : I am Shruthi VijayKumar, a research associate in the Royer lab. A significant part of my role involved developing and optimizing the single embryo single cell dissociation protocol for the various developmental stages in zebrahub along with Michael Borja (Genomics) and Merlin Lange. Additionally, I contributed to preparing the libraries required for sequencing and helped with cell annotations. This project has been an incredible experience, as it has provided me with the opportunity to work in a multidisciplinary and collaborative environment. This experience has not only given me a better understanding of the entire pipeline but also broadened my expertise through extensive interactions with different teams. The fact that Zebrahub is an expanding resource for the community makes this project even more special and rewarding to be a part of.
Sarah: My name is Sarah Ancheta, and I am an associate Data Scientist. As part of the Zebrahub team, I worked on data processing and analysis of inter-individual transcriptomic variability. Since Zebrahub has single-cell transcriptomic datasets from individually resolved embryos, I was able to dissect the embryo-to-embryo variability by developing a framework to investigate the differences in gene expression between sibling individuals over time. I feel fortunate to work with data of such high quality and resolution and excited to have been among the first to analyze Zebrahub’s data and explore new biological insights into zebrafish development. It has been a wonderful experience to work with such an interdisciplinary team and learn to conduct analyses from both a biological and data-driven perspective.
Mike: My name is Michael Borja, and I am a Senior Research Associate for the Genomics platform. My main role and responsibility is to oversee lab activities regarding Single Cell and Spatial Transcriptomic experiments. When Zebrahub initially started, I was tasked with selecting which single-cell transcriptomic assay to use for our initial experiments. From there, I led the early efforts of processing cell dissociations and library preparation of individual embryos. In addition and in partnership with Shruthi VijayKumar, we developed a robust zebrafish single-cell dissociation protocol from single embryos that is independent of the developmental stage. Being part of this multidisciplinary and innovative team has helped me see how large-scale projects within the realm of science can be idealized and completed. As I continue with my personal growth and path in the field of Genomics, I will absolutely continue to seek large-scale collaborations such as Zebrahub so that I may be able to apply what I’ve learned through this experience.
Jordao: My name is Jordao Bragantini. I am a software engineer in the Royer lab. My primary focus revolves around image processing, cell segmentation, and tracking. The Zebrahub project exposed me to the fascinating world of terabyte-scale microscopy images as well as to the mysteries of Developmental Biology. Handling this vast amount of data acquired on our advanced light-sheet microscopes required implementing specialized algorithms, ranging from GPU-friendly approximate morphological operators to our own distributed tracking software. These analyses require the full capacity of our available computational resources. I could not have wished for a better way to engage with machine learning in the biology domain than working with this multidisciplinary team, where we can learn from each other’s expertise.
What are the next steps for Zebrahub?
We want to expand Zebrahub with more developmental stages, novel multiomic modalities, and more lineage-specific imaging to ultimately reconstruct a digital multimodal embryo. We are also working on integrating existing datasets, including those from diverse species, to create a comprehensive atlas of vertebrate embryogenesis. This ambitious endeavor heralds a transformative era for the fields of developmental and evolutionary biology.
You can read more about Zebrahub in the BioRxiv preprint.


 (No Ratings Yet)
(No Ratings Yet)

 (3 votes)
(3 votes)