Catch up on Development presents… February webinar on organogenesis
Posted by the Node, on 6 March 2024
In the second 2024 webinar, Development Editor Liz Robertson (University of Oxford) hosted two early career researchers studying organogenesis.
Posted by the Node, on 6 March 2024
In the second 2024 webinar, Development Editor Liz Robertson (University of Oxford) hosted two early career researchers studying organogenesis.
Posted by the Node, on 5 March 2024
Welcome to our monthly trawl for developmental and stem cell biology (and related) preprints.
The preprints this month are hosted on bioRxiv – use these links below to get to the section you want:
Research practice and education
Rbm8a deficiency causes hematopoietic defects by modulating Wnt/PCP signalingv
Agnese Kocere, Elena Chiavacci, Charlotte Soneson, Harrison H. Wells, Kevin Manuel Méndez-Acevedo, Jacalyn S. MacGowan, Seth T. Jacobson, Max S. Hiltabidle, Azhwar Raghunath, Jordan A. Shavit, Daniela Panáková, Margot L. K. Williams, Mark D. Robinson, Christian Mosimann, Alexa Burger
Hedgehog signaling is required for the maintenance of mesenchymal nephron progenitors
Eunah Chung, Patrick Deacon, Yueh-Chiang Hu, Hee-Woong Lim, Joo-Seop Park
Kim Moorwood, Florentia M. Smith, Alastair S. Garfield, Michael Cowley, Lowenna J. Holt, Roger J. Daly, Andrew Ward
Qin Pan, Moritz Mercker, Alexander Klimovich, Jörg Wittlieb, Anna Marciniak-Czochra, Angelika Böttger
Youngmin Hwang, Yuko Shimamura, Junichi Tanaka, Akihiro Miura, Anri Sawada, Hemanta Sarmah, Dai Shimizu, Yuri Kondo, Zurab Ninish, Kazuhiko Yamada, Munemasa Mori
Geoffrey Soussi, Ausra Girdziusaite, Shalu Jhanwar, Victorio Palacio, Rushikesh Sheth, Rolf Zeller, Aimée Zuniga
Kendall R. Branham, Chhavi Sood, Susan E. Doyle, Matt C. Pahl, Sarah E. Siegrist
Sandra de Haan, Agustin A. Corbat, Christopher R. Cederroth, Lisa G. Autrum, Simona Hankeova, Elizabeth C. Driver, Barbara Canlon, Matthew W. Kelley, Emma R. Andersson
Suppression of ERK signalling promotes pluripotent epiblast in the human blastocyst
Claire S. Simon, Afshan McCarthy, Laura Woods, Desislava Staneva, Qiulin Huang, Madeleine Linneberg-Agerholm, Alex Faulkner, Athanasios Papathanasiou, Kay Elder, Phil Snell, Leila Christie, Patricia Garcia, Valerie Shaikly, Mohamed Taranissi, Meenakshi Choudhary, Mary Herbert, Joshua M. Brickman, Kathy K. Niakan
A TBX5-dependent compartment boundary patterns the cardiac interventricular septum
Irfan S. Kathiriya, Martin H. Dominguez, Kavitha S. Rao, Jonathon M. Muncie-Vasic, W. Patrick Devine, Kevin M. Hu, Swetansu K. Hota, Bayardo I. Garay, Diego Quintero, Piyush Goyal, Megan N. Matthews, Reuben Thomas, Tatyana Sukonnik, Dario Miguel-Perez, Sarah Winchester, Emily F. Brower, André Forjaz, Pei-Hsun Wu, Denis Wirtz, Ashley L. Kiemen, Benoit G. Bruneau
Temporal dynamics of BMP/Nodal ratio drive tissue-specific gastrulation morphogenesis
Alyssa A Emig, Megan Hansen, Sandra Grimm, Cristian Coarfa, Nathan D Lord, Margot Kossmann Williams
Simaran Johal, Randa Elsayed, Kristen A. Panfilio, Andrew C. Nelson
Connor J Sproston, Julia E Rak, Shu Kondo, Darren W Williams
Fengzhu Xiong, Andrea R Tentner, Sandy Nandagopal, Tom W Hiscock, Peng Huang, Sean Megason
The deubiquitylating enzyme Fat Facets promotes Fat signalling and restricts tissue growth
Lauren E Dawson, Alexander D Fulford, Aashika Sekar, Paulo S Ribeiro
NOTUM-MEDIATED WNT SILENCING DRIVES EXTRAVILLOUS TROPHOBLAST CELL LINEAGE DEVELOPMENT
Vinay Shukla, Ayelen Moreno-Irusta, Kaela M. Varberg, Marija Kuna, Khursheed Iqbal, Anna M. Galligos, John D. Aplin, Ruhul H. Choudhury, Hiroaki Okae, Takahiro Arima, Michael J. Soares
Lineage-specific intersection of endothelin and GDNF signaling in enteric nervous system development
Denise M. Poltavski, Alexander T. Cunha, Jaime Tan, Henry M. Sucov, Takako Makita
Duplication of the anteroposterior body axis in postembryos of the Eratigena atrica spider
Teresa Napiórkowska, Julita Templin, Paweł Napiórkowski
Diverse Fgfr1 signaling pathways and endocytic trafficking regulate early mesoderm development
James F. Clark, Philippe Soriano
Mohammad Alhashmi, Abdulrahman ME Gremida, Santosh K Maharana, Marco Antonaci, Amy Kerr, Noor A Al-Maslamani, Ke Liu, Maria M Meschis, Hazel Sutherland, Peter Wilson, Peter Clegg, Grant N Wheeler, Robert J van ’t Hof, George Bou-Gharios, Kazuhiro Yamamoto
Edgar M. Pera, Josefine Nilsson-De Moura, Yuriy Pomeshchik, Laurent Roybon, Ivana Milas
Spatiotemporal dynamics of NF-κB/Dorsal inhibitor IκBα/Cactus in Drosophila blastoderm embryos
Allison E. Schloop, Sharva Hiremath, Razeen Shaikh, Cranos M. Williams, Gregory T. Reeves
Spatial Multiomics Reveal the Role of Wnt Modulator, Dkk2, in Palatogenesis
Jeremie Oliver Pina, Resmi Raju, Daniela M Roth, Emma Wentworth Winchester, Cameron Padilla, James Iben, Fabio R Faucz, Justin L Cotney, Rena N D’Souza
Shruti S. Tophkhane, Katherine Fu, Esther M. Verheyen, Joy M. Richman
The ciliary protein C2cd3 is required for mandibular musculoskeletal tissue patterning
Evan C. Brooks, Simon J.Y. Han, Christian Louis Bonatto Paese, Amya A. Lewis, Megan Aarnio-Peterson, Samantha A. Brugmann
Coupling and uncoupling of midline morphogenesis and cell flow in amniote gastrulation
Rieko Asai, Vivek N. Prakash, Shubham Sinha, Manu Prakash, Takashi Mikawa
Role of Fibronectin in Postnatal Skeletal Development
Neha E. H. Dinesh, Nissan Baratang, Justine Rosseau, Ronit Mohapatra, Ling Li, Ramshaa Mahalingam, Kerstin Tiedemann, Philippe M. Campeau, Dieter P. Reinhardt
Compressive stress drives morphogenetic apoptosis
Tatiana Merle, Martine Cazales, Ronan Bouzignac, Brice Ronsin, Christian Rouvière, Magali Suzanne
A tension-induced morphological transition shapes the avian extra-embryonic territory
Arthur Michaut, Alexander Chamolly, Aurélien Villedieu, Francis Corson, Jérôme Gros
Susannah B.P. McLaren, Shi-Lei Xue, Siyuan Ding, Alex Winkel, Oscar Baldwin, Shreya Dwarakacherla, Kristian Franze, Edouard Hannezo, Fengzhu Xiong
AnnJosette Ramirez, Christina Vyzas, Kevin Eng, Karl Degenhardt, Sophie Astrof
Mechanotransductive feedback control of endothelial cell motility and vascular morphogenesis
Devon E. Mason, Paula Camacho, Megan E. Goeckel, Brendan R. Tobin, Sebastián L. Vega, Pei-Hsun Wu, Dymonn Johnson, Su-Jin Heo, Denis Wirtz, Jason A. Burdick, Levi Wood, Brian Y. Chow, Amber N. Stratman, Joel D. Boerckel
Signatures of structural disorder in developing epithelial tissues
Christian Cupo, Cole Allan, Vikram Ailiani, Karen E. Kasza
Eph/ephrin signalling in the developing brain is regulated by tissue stiffness
Jana Sipkova, Kristian Franze
Claudio Araya, Raegan Boekemeyer, Francesca Farlie, Lauren Moon, Freshta Darwish, Chris Rookyard, Leanne Allison, Gema Vizcay-Barrena, Roland Fleck, Millaray Aranda, Masa Tada, Jonathan D W Clarke
Eman Hijaze, Tsvia Gildor, Ronald Seidel, Majed Layous, Mark Winter, Luca Bertinetti, Yael Politi, Smadar Ben-Tabou de-Leon
Michele Romanos, Tasha Salisbury, Samuel Stephan, Rusty Lansford, Pierre Degond, Ariane Trescases, Bertrand Benazeraf
Lens Placode Modulates Extracellular Matrix Formation During Early Eye Development
Cecilia Gallottini de Magalhaes, Ales Cvekl, Ruy G. Jaeger, Chao Yun Irene Yan
Casey Griffin, Kelsey Coppenrath, Doha Khan, Ziyan Lin, Marko Horb, Jean-Pierre Saint-Jeannet
Yuuta Moriyama, Toshiyuki Mitsui, Carl-Philipp Heisenberg
Lisa-Marie Legault, Thomas Dupas, Mélanie Breton-Larrivée, Fannie Filion-Bienvenue, Anthony Lemieux, Alexandra Langford-Avelar, Serge McGraw
miR-203 controls timing of developmental transitions during early preimplantation embryogenesis
José González-Martínez, Estefanía Ayala, Agustín Sanchez-Belmonte, Alejandro García, Enrique Nogueira, Anna Melati, Daniel Gimenez, Ana Losada, Sagrario Ortega, Marcos Malumbres
Grigorios Fanourgakis, Laura Gaspa-Toneu, Pavel A. Komarov, Evgeniy A. Ozonov, Sebastien A. Smallwood, Antoine H.F.M. Peters
Developmental pathways underlying sexual differentiation in a U/V sex chromosome system
Daniel Liesner, Guillaume Cossard, Min Zheng, Olivier Godfroy, Josue Barrera-Redondo, Fabian B. Haas, Susana M Coelho

X-chromosome loss rescues Sertoli cell maturation and spermatogenesis in Klinefelter syndrome
Sofia Boeg Winge, Niels E. Skakkebaek, Lise Aksglaede, Guelizar Saritas, Ewa Rajpert-De Meyts, Ellen Goossens, Anders Juul, Kristian Almstrup
Fengmei Xu, Ritsuko Suyama, Toshifumi Inada, Shinichi Kawaguchi, Toshie Kai
Stephenson Chea, Jesse Kreger, Martha E. Lopez-Burks, Adam L. MacLean, Arthur D. Lander, Anne L. Calof
Adriana Karina Alexander, Karina F Rodriguez, Yu-Ying Chen, Ciro Amato, Martin A Estermann, Xin Xu, Humphrey Yao
Bernard K. van der Veer, Lehua Chen, Spyridon Champeris Tsaniras, Wannes Brangers, Qiuying Chen, Mariana Schroiff, Colin Custers, Harm H.M. Kwak, Rita Khoueiry, Robert Cabrera, Steven S. Gross, Richard H. Finnell, Yunping Lei, Kian Peng Koh
Escape from X inactivation is directly modulated by levels of Xist non-coding RNA
Antonia Hauth, Jasper Panten, Emma Kneuss, Christel Picard, Nicolas Servant, Isabell Rall, Yuvia A. Pérez-Rico, Lena Clerquin, Nila Servaas, Laura Villacorta, Ferris Jung, Christy Luong, Howard Y. Chang, Oliver Stegle, Duncan T. Odom, Agnese Loda, Edith Heard
Spatial patterning of the epigenome during vertebrate gastrulation
Ana Paula Azambuja, Megan Rothstein, Tatiane Y. Kanno, Marcos Simoes-Costa
Rapid response of fly populations to gene dosage across development and generations
Xueying C. Li, Lautaro Gandara, Måns Ekelöf, Kerstin Richter, Theodore Alexandrov, Justin Crocker
Haploidy-linked cell proliferation defects limit larval growth in Zebrafish
Kan Yaguchi, Daiki Saito, Triveni Menon, Akira Matsura, Takeomi Mizutani, Tomoya Kotani, Sreelaja Nair, Ryota Uehara
Juliana Moreira Marques, Claudia Veiga Chang, Nicholas Silvestre Souza Trigueiro, Ricardo Vieira Araujo, Cinthya dos Santos Cerqueira, Lilian Cristina Russo, Bruna Viscardi Azevedo, Berenice Bilharinho de Mendonca, Nicolas Hoch, Luciani Renata Silveira de Carvalho
Liangdao Li, Xinbao Ding, Alex P. Sheft, John C. Schimenti
Maria Ivanova, Eric G. Moss
Aging impairs cold-induced beige adipogenesis and adipocyte metabolic reprogramming
Corey D. Holman, Alexander P. Sakers, Ryan P. Calhoun, Lan Cheng, Ethan C. Fein, Christopher Jacobs, Linus Tsai, Evan D. Rosen, Patrick Seale
Sophie T Raterman, Frank A D T G Wagener, Jan Zethof, Vincent Cuijpers, Peter H M Klaren, Juriaan R Metz, Johannes W. Von den Hoff
Notch is Required for Neural Progenitor Proliferation During Embryonic Eye Regrowth
Dylan J. Guerin, Belen Gutierrez, Baoyi Zhang, Kelly Ai-Sun Tseng
Somite-independent regeneration of the axolotl primary body axis
Wouter Masselink, Tobias Gerber, Francisco Falcon, Tom Deshayes, Sofia-Christina Papadopoulos, Marko Pende, Vijayishwer Singh Jamwal, Yuka Taniguchi-Sugiura, Tzi-Yang Lin, Thomas Kurth, Jingkui Wang, Detlev Arendt, Ji-Feng Fei, Barbara Treutlein, Elly M. Tanaka, Prayag Murawala
Mona Steichele, Lara Sauermann, Qin Pan, Jasmin Moneer, Alexandra de la Porte, Martin Heß, Moritz Mercker, Catharina Strube, Marcell Jenewein, Angelika Böttger
Substrate Stiffness Reshapes Layer Architecture and Biophysical Features of Human Induced Pluripotent Stem Cells to Modulate their Differentiation Potential
Jack Llewellyn, Anne Charrier, Emmanuèle Helfer, Rosanna Dono
Margarida Viola, Maarten P. Bebelman, Renee G. C. Maas, Frederik J. Verweij, Cor S. Seinen, Saskia C. A. de Jager, Pieter Vader, D. Michiel Pegtel, Joost P. G. Sluijter
Yifei Miao, Cheng Tan, Nicole M. Pek, Zhiyun Yu, Kentaro Iwasawa, Daniel O. Kechele, Nambirajan Sundaram, Victor Pastrana-Gomez, Keishi Kishimoto, Min-Chi Yang, Cheng Jiang, Jason Tchieu, Jeffrey A. Whitsett, Kyle W. McCracken, Robbert J. Rottier, Darrell N. Kotton, Michael A. Helmrath, James M. Wells, Takanori Takebe, Aaron M. Zorn, Ya-Wen Chen, Minzhe Guo, Mingxia Gu
Microtubule perturbations of neurite extension and nucleokinesis in an iPSC-derived model system
Muriel Sébastien, Alexandra L. Paquette, Adam G. Hendricks, Gary J. Brouhard
Progressive cell fate specification in morphallactic regeneration
Clara Nuninger, Panagiotis Papasaikas, Jacqueline Ferralli, Sebastien Smallwood, Charisios D. Tsiairis
Novel 3D Approach to Model Non-Alcoholic Fatty Liver Disease using human Pluripotent Stem Cells
Carola Maria Morell, Samantha Grace Tilson, Rute Alexandra Tomaz, Arash Shahsavari, Andi Munteanu, Giovanni Canu, Brandon Tyler Wesley, Marion Perrin, Imbisaat Geti, Subhankar Mukhopadhyay, Francesca Mazzacuva, Paul Gissen, Jose Garcia-Bernardo, Martin Bachman, Casey Allison Rimland, Fotios Sampaziotis, Irina Mohorianu, Ludovic Vallier
Léa Torcq, Sara Majello, Catherine Vivier, Anne A. Schmidt
Pauline Garcia, William Jarassier, Caroline Brun, Lorenzo Giordani, Fany Agostini, Wai Hing Kung, Cécile Peccate, Jade Ravent, Sidy Fall, Valentin Petit, Tom H Cheung, Slimane Ait-Si-Ali, Fabien Le Grand
Loss of UBE3A impacts both neuronal and non-neuronal cells in human cerebral organoids
R. Chris Estridge, Z. Begum Yagci, Dilara Sen, Travis S. Ptacek, Jeremy M. Simon, Albert J. Keung
Selective Translation Orchestrates Key Signaling Pathways in Primed Pluripotency
Chikako Okubo, Michiko Nakamura, Masae Sato, Yuichi Shichino, Mari Mito, Yasuhiro Takashima, Shintaro Iwasaki, Kazutoshi Takahashi
Developmental emergence of quiescent-like neural progenitor cells in the zebrafish embryonic brain
Yuanyuan Liu, Carla-Sophie Lembke, Judith TML Paridaen
Velinda Vidaurre, Annabelle Song, Taibo Li, Wai Lim Ku, Keji Zhao, Jiang Qian, Xin Chen
Characterization of Pro-Fibrotic Signaling Pathways using Human Hepatic Organoids
Yuan Guan, Zhuoqing Fang, Angelina Hu, Sarah Roberts, Meiyue Wang, Wenlong Ren, Patrik K. Johansson, Sarah C. Heilshorn, Annika Enejder, Gary Peltz
Julia Kramer, Rita Aires, Sean D. Keeley, Tom Alexander Schröder, Günter Lauer, Tatiana Sandoval-Guzmán
Resistance to Naïve and Formative Pluripotency Conversion in RSeT Human Embryonic Stem Cells
Kevin G. Chen, Kory R. Johnson, Kyeyoon Park, Dragan Maric, Forest Yang, Wen Fang Liu, Yang C. Fann, Barbara S. Mallon, Pamela G. Robey
Licensing and competition of stem cells at the niche combine to regulate tissue maintenance
Rodrigo Garcia-Tejera, Marc Amoyel, Ramon Grima, Linus Schumacher
Olivia Stonehouse, Christine Biben, Tom S Weber, Alexandra Garnham, Katie Fennell, Alison Farley, Antoine Terreaux, Warren Alexander, Mark Dawson, Shalin Naik, Samir Taoudi
DDR1 regulates RUNX1-CBFβ to control breast stem cell differentiation
Colin Trepicchio, Gat Rauner, Nicole Traugh, Meadow Parrish, Daniel E.C. Fein, Youssof Mal, Charlotte Kuperwasser
George C. Gabriel, Hisato Yagi, Tuantuan Tan, Abha S. Bais, Benjamin J. Glennon, Margaret C. Stapleton, Lihua Huang, William T. Reynolds, Marla G. Shaffer, Madhavi Ganapathiraju, Dennis Simon, Ashok Panigrahy, Yijen L. Wu, Cecilia W. Lo
TASOR expression in naive embryonic stem cells safeguards their developmental potential
Carlos A. Pinzon-Arteaga, Ryan O’Hara, Alice Mazzagati, Emily Ballard, Yingying Hu, Alex Pan, Daniel A. Schmitz, Yulei Wei, Masahiro Sakurai, Peter Ly, Laura Banaszynski, Jun Wu
Stereotyped cell lineage trees support robust development
Xiaoyu Zhang, Zizhang Li, Jingyu Chen, Wenjing Yang, Peng Wu, Feng Chen, Xingxing He, Xiaoshu Chen, Jian-Rong Yang
The Mayo Clinic Salivary Tissue-Organoid Biobanking: A Resource for Salivary Regeneration Research
Syed Mohammed Musheer Aalam, Ana Rita Varela, Aalim Khaderi, Ronsard J Mondesir, Dong-Gi Mun, Andrew Ding, Isabelle M.A. Lombaert, Rob P. Coppes, Chitra Priya Emperumal, Akhilesh Pandey, Jeffrey R. Janus, Nagarajan Kannan
Douglas Terry, Colby Schweibenz, Kenneth Moberg
Himanshu Chhillar, Pei-Min Yeh, Hoang Hung Nguyen, Jonathan DG Jones, Pingtao Ding
Kevin Bellande, David Roujol, Josiane Chourré, Sophie Le Gall, Yves Martinez, Alain Jauneau, Vincent Burlat, Elisabeth Jamet, Hervé Canut
Twilight length alters growth and flowering time in Arabidopsis via LHY/CCA1
Devang Mehta, Sabine Scandola, Curtis Kennedy, Christina Lummer, Maria Camila Rodriguez Gallo, Lauren E. Grubb, Maryalle Tan, Enrico Scarpella, R. Glen Uhrig
Ashleigh Edwards, Maurizio Junior Chiurazzi, Anko Blaakmeer, Ylenia Vittozzi, Ashish Sharma, Sanne Matton, Valdeko Kruusvee, Daniel Straub, Giovanna Sessa, Monica Carabelli, Giorgio Morelli, Stephan Wenkel
Maria Florencia Ercoli, Alexandra M. Shigenaga, Artur Teixeira de Araujo Jr, Rashmi Jain, Pamela C. Ronald
Alan O. Marron, Marius Rebmann, Mihails Delmans, Jim Haseloff
OsNF-YB7 inactivates OsGLK1 to inhibit chlorophyll biosynthesis in rice embryo
Zongju Yang, Tianqi Bai, E Zhiguo, Baixiao Niu, Chen Chen
Chiara Perico, Maricris Zaidem, Olga Sedelnikova, Samik Bhattacharya, Christian Korfhage, Jane A. Langdale
Geometry and cell wall mechanics guide early pollen tube growth in Arabidopsis thaliana
Lucie Riglet, Catherine Quilliet, Christophe Godin, Karin John, Isabelle Fobis-Loisy
Tomasz Jamruszka, Joanna Banasiak, Aleksandra Pawela, Karolina Jarzyniak, Jian Xia, Wanda Biała-Leonhard, Lenka Plačková, Tashi Tsering, Francesca Romana Iacobini, Ondřej Novák, Markus Geisler, Michał Jasiński
ABCC10 roles in plant development and the transport of indole-3-butyric acid
Arielle L. Homayouni, Suresh Damodaran, Katherine Schreiber, Marta Michniewicz, Lauren K. Gunther, Lucia C. Strader
Ashleigh Edwards, Maurizio Junior Chiurazzi, Anko Blaakmeer, Ylenia Vittozzi, Ashish Sharma, Sanne Matton, Valdeko Kruusvee, Daniel Straub, Giovanna Sessa, Monica Carabelli, Giorgio Morelli, Stephan Wenkel
The ELF3-BBX24/BBX25-PIF4 module controls thermosensory growth in Arabidopsis
Bidhan Chandra Malakar, Shivani Singh, Vikas Garhwal, Gouranga Upadhyaya, Rajanesh Chandramohan, Sreeramaiah N. Gangappa
Noy Sadot Muzika, Tamir Kamai, Leor Eshed Williams, Maya Kleiman
Leila Jaafar, Yintong Chen, Sedighe Keynia, Joseph A Turner, Charles T Anderson
The DC1 domain protein Vacuoleless Gametophytes regulates stamen development in Arabidopsis
Natalia L. Amigo, Leonardo A. Arias, Fernanda Marchetti, Sebastián D’Ippólito, Jesica Frik, María Cristina Lombardo, María Cecilia Terrile, Claudia A. Casalongue, Gabriela C. Pagnussat, Diego F. Fiol
Stomatal patterning is differently regulated in adaxial and abaxial epidermis in Arabidopsis
Pirko Jalakas, Ingmar Tulva, Nele Malvīne Bērziņa, Hanna Hõrak
Overexpressing NRT2.7 induces nitrate export from the vacuole and increases growth of Arabidopsis
Patrick Armengaud, Alexis De Angeli, Patrick Berquin, Virginie Bréhaut, Mickaël Durand, Françoise Daniel-Vedele, Anne Krapp, Sophie Filleur
SHOOTMERISTEMLESS autoactivation: A prerequisite for fruit metamorphosis
Yang Dong, Zhi-Cheng Hu, Mateusz Majda, Hao-Ran Sun, Yao Zhang, Yi-Ning Ding, Quan Yuan, Tong-Bing Su, Tian-Feng Lü, Feng Gao, Gui-Xia Xu, Richard S. Smith, Lars Østergaard
Tathagata Biswas, Huzaifa Hassan, Nicolas Rohner
Lineage-resolved analysis of embryonic gene expression evolution in C. elegans and C. briggsae
Christopher R. L. Large, Rupa Khanal, LaDeana Hillier, Chau Huynh, Connor Kubo, Junhyong Kim, Robert H. Waterston, John I. Murray
Joel H. Gayford, Phillip C. Sternes, Scott G. Seamone, Hana Godfrey, Darren A. Whitehead
Pierre Galipot, Julie Zalko
Sydney K Decker, Kaiya L Provost, Bryan C Carstens
The ivory lncRNA regulates seasonal color patterns in buckeye butterflies
Richard A Fandino, Noah K Brady, Martik Chatterjee, Jeanne Marie Chan McDonald, Luca Livraghi, Karin van der burg, Anyi Mazo-Vargas, Robert D. Reed
Katie Pelletier, Megan Bilodeau, Isabella Pellizzari-Delano, M. Daniel Siemon, Yuheng Huang, John E. Pool, Ian E Dworkin
Eyespots originated multiple times independently across the Lepidoptera
Brian Hanotte, Beatriz Willink, Antónia Monteiro
Rapid growth and the evolution of complete metamorphosis in insects
Christin Manthey, C Jessica E Metcalf, Michael T Monaghan, Ulrich Karl Steiner, Jens Rolff
Lisa Hanna, Brendon E. Boudinot, Jürgen Liebig, Ehab Abouheif
mirror determines the far posterior domain in butterfly wings
Martik Chatterjee, Xin Y. Yu, Noah K. Brady, Gabriel C. Hatto, Robert D. Reed
Gabrielle C. Coffing, Silas Tittes, Scott T. Small, Jeremea O. Songco-Casey, Denise M. Piscopo, Judit R. Pungor, Adam C Miller, Cristopher Niell, Andrew D. Kern
Cell cycle variants during Drosophila accessory gland development
Allison M. Box, Navyashree A. Ramesh, Shyama Nandakumar, Samuel Jaimian Church, Dilan Prasad, Ariana Afrakhteh, Russell S. Taichman, Laura Buttitta
Elena D. Christofidou, Marios Tomazou, Chrysovalantis Voutouri, Christina Michael, Triantafyllos Stylianopoulos, George M. Spyrou, Katerina Strati
Mohammad Zeeshan, Ravish Rashpa, David J. Ferguson, George Mckeown, Raushan Nugmanova, Amit K. Subudhi, Raphael Beyeler, Sarah L. Pashley, Robert Markus, Declan Brady, Magali Roques, Andrew R. Bottrill, Andrew M. Fry, Arnab Pain, Sue Vaughan, Anthony A. Holder, Eelco C. Tromer, Mathieu Brochet, Rita Tewari
Ottavia Romoli, Javier Serrato-Salas, Chloé Gapp, Yanouk Epelboin, Pol Figueras Ivern, Frédéric Barras, Mathilde Gendrin
IL-1β disrupts blood-brain barrier development by inhibiting endothelial Wnt/β-catenin signaling
Audrey R. Fetsko, Dylan J. Sebo, Lilyana B. Budzynski, Alli Scharbarth, Michael R. Taylor
Loss of the mitochondrial citrate carrier, Slc25a1/CIC disrupts embryogenesis via 2-Hydroxyglutarate
Anna Kasprzyk-Pawelec, Mingjun Tan, Yu Leng Phua, Raneen Rahhal, Alec McIntosh, Harvey Fernandez, Rami Mosaoa, Michael Girgis, Amrita Cheema, Lei Jiang, Lawrence F. Kroemer, Anastas Popratiloff, Cheryl Clarkson, Brian M. Kirmsa, Gray W. Pearson, Eric Glasgow, Christopher Albanese, Jerry Vockley, Maria Laura Avantaggiati
Ruonan Zhao, Emma L. Moore, Madelaine M Gogol, Jay R. Unruh, Zulin Yu, Allison Scott, Yan Wang, Naresh Kumar Rajendran, Paul A. Trainor
Yanan Shan, Jacob A O’Brian, Lubna Nadeem, Caroline Dunk, Yan Chen, Jelena Brkic, Heyam Hayder, Stephen J Lye, Chun Peng
The transmembrane protein Syndecan regulates stem cell nuclear properties and cell maintenance
Buffy L. Eldridge-Thomas, Jerome G. Bohere, Chantal Roubinet, Alexandre Barthelemy, Tamsin J. Samuels, Felipe Karam Teixeira, Golnar Kolahgar
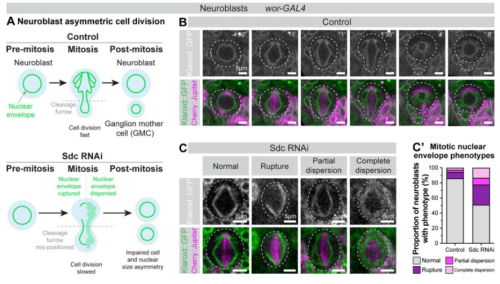
Nicholas W. Zinck, Danielle Stanton-Turcotte, Emily A. Witt, Marley Bloomers, Angelo Iulianella
Filamin A mediates embryonical palatal fusion by linking mechanotransduction with β-Catenin/Smad2
Ziyi Wang, Satoru Hayano, Yao Weng, Xindi Mu, Mitsuaki Ono, Jeremie Oliver Piña, Rena N. D’Souza, Takashi Yamashiro, Toshitaka Oohashi, Hiroshi Kamioka
Sanjay Kumar Sukumar, Vimala Antonydhason, Linnea Molander, Jawdat Sandakly, Malak Kleit, Ganesh Umapathy, Patricia Mendoza-Garcia, Tafheem Masudi, Andreas Schlossser, Dick R. Nässel, Christian Wegener, Margret Shirinian, Ruth H. Palmer
Antagonistic Roles of Tau and MAP6 in Regulating Neuronal Development
Xiaohuan Sun, Wenqian Yu, Peter W. Baas, Kazuhito Toyooka, Liang Qiang
Placental cytotrophoblast microvillar stabilization is required for cell-cell fusion
Wendy K. Duan, Sumaiyah Z. Shaha, Khushali J. Patel, Ivan K. Domingo, Meghan R. Riddell
Weihua Wang, Junqiao Xing, Xiqi Zhang, Hongni Liu, Haochen Jiang, Cheng Xu, Xue Zhao, Zhangfeng Hu
Pierre Ferrer, Srijana Upadhyay, James Cai, Tracy M Clement
Rapid growth and the evolution of complete metamorphosis in insects
Christin Manthey, Jessica CE Metcalf, Michael T Monaghan, Ulrich Karl Steiner, Jens Rolff
Modularity of the segmentation clock and morphogenesis
James E. Hammond, Ruth E. Baker, Berta Verd
Deep Learning Based Models for Preimplantation Mouse and Human Development
Martin Proks, Nazmus Salehin, Joshua M. Brickman
Daniel Seeler, Nastasja Grdseloff, Claudia Jasmin Rödel, Charlotte Kloft, Salim Abdelilah-Seyfried, Wilhelm Huisinga
A quantitative model for in vivo stem cell decisions in planarians
Tamar Frankovits, Prakash Varkey Cherian, Yarden Yesharim, Simon Dobler, Omri Wurtzel
A Comprehensive Human Embryogenesis Reference Tool using Single-Cell RNA-Sequencing Data
Cheng Zhao, Alvaro Plaza Reyes, John Paul Schell, Jere Weltner, Nicolás M. Ortega, Yi Zheng, Åsa K. Björklund, Laura Baqué-Vidal, Joonas Sokka, Ras Torokovic, Brian Cox, Janet Rossant, Jianping Fu, Sophie Petropoulos, Fredrik Lanner
A Genetic Screen for regulators of CNS morphology in Drosophila
Haluk Lacin, Yuqing Zhu, Jose DiPaola, Beth A. Wilson, Yi Zhu, James B. Skeath
Kelly J. Clark, Emily E. Lubin, Elizabeth M. Gonzalez, Annabel K. Sangree, Dana E. Layo-Carris, Emily L. Durham, Rebecca C. Ahrens-Nicklas, Tomoki T. Nomakuchi, Elizabeth J. Bhoj
Monique Mills, Chihiro Emori, Parveen Kumar, Zachary Boucher, Joshy George, Ewelina Bolcun-Filas
Single-cell and spatial transcriptomics of the avian embryo tailbud
GF Mok, S Turner, E Smith, L Mincarelli, A Lister, J Lipscombe, V Uzun, W Haerty, IC Macaulay, A Münsterberg
Taro Ichimura, Taishi Kakizuka, YuKi Sato, Keiko Itano, Kaoru Seiriki, Hitoshi Hashimoto, Hiroya Itoga, Shuichi Onami, Takeharu Nagai
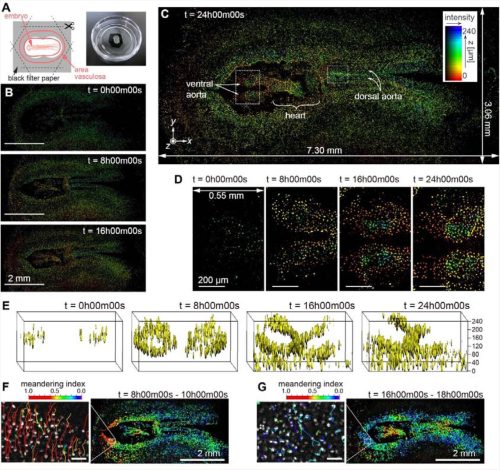
Félix Simon, Isabel Holguera, Yen-Chung Chen, Jennifer Malin, Priscilla Valentino, Ted Erclik, Claude Desplan
A single-cell 3D spatiotemporal multi-omics atlas from Drosophila embryogenesis to metamorphosis
Mingyue Wang, Qinan Hu, Zhencheng Tu, Lingshi Kong, Jiajun Yao, Rong Xiang, Zhan Chen, Yan Zhao, Yanfei Zhou, Tengxiang Yu, Yuetian Wang, Zihan Jia, Kang Ouyang, Xianzhe Wang, Yinqi Bai, Mingwei Lian, Zhenyu Yang, Tao Yang, Jing Chen, Yunting Huang, Ni Yin, Wenyuan Mo, Wenfu Liang, Chang Liu, Xiumei Lin, Chuanyu Liu, Ying Gu, Wei Chen, Longqi Liu, Xun Xu, Yuhui Hu
The Guinea Pig: A New Model for Human Preimplantation Development
Jesica Romina Canizo, Cheng Zhao, Sophie Petropoulos
Fátima Sanchís-Calleja, Akanksha Jain, Zhisong He, Ryoko Okamoto, Charlotte Rusimbi, Pedro Rifes, Gaurav Singh Rathore, Malgorzata Santel, Jasper Janssens, Makiko Seimiya, Jonas Simon Fleck, Agnete Kirkeby, J. Gray Camp, Barbara Treutlein
Chloe Moss, Barbara Vacca, Jo Arnold, Chantal Hubens, Dominic M. Lynch, James Pegge, Michael A.R. Green, Charlotte A. Hosie, Tessa E. Smith, Jeremy B.A. Green
Ashwin Bhaskar, Sophie Astrof
Nobuhiko Hamazaki, Wei Yang, Connor Kubo, Chengxiang Qiu, Beth K Martin, Riddhiman K Garge, Samuel G Regalado, Eva Nichols, Choli Lee, Riza M Daza, Sanjay Srivatsan, Jay Shendure
Scalable and Efficient Generation of Mouse Primordial Germ Cell-like Cells
Xinbao Ding, Liangdao Li, Jingyi Gao, Dain Yi, John C Schimenti
Kendra L. Stansak, Luke D. Baum, Sumana Ghosh, Punam Thapa, Vineel Vanga, Bradley J. Walters
Juliana Sánchez-Posada, Emily S Noël
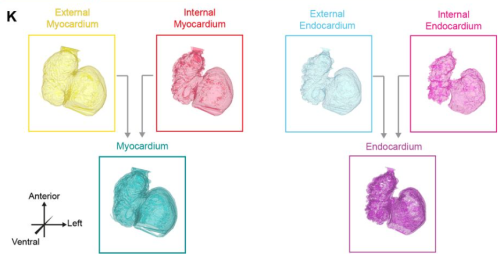
Nuclear instance segmentation and tracking for preimplantation mouse embryos
Hayden Nunley, Binglun Shao, David Denberg, Prateek Grover, Jaspreet Singh, Maria Avdeeva, Bradley Joyce, Rebecca Kim-Yip, Abraham Kohrman, Abhishek Biswas, Aaron Watters, Zsombor Gal, Alison Kickuth, Madeleine Chalifoux, Stanislav Shvartsman, Lisa M. Brown, Eszter Posfai
Stephen M J Pollo, Hongrui Liu, Aralia Leon Coria, Nicole Rosin, Elodie Labit, Jeff Biernaskie, Constance A M Finney, James D Wasmuth
Structured Peer Review: Pilot results from 23 Elsevier Journals
Mario Malički, Bahar Mehmani
Perry G Beasley-Hall, Pam Papadelos, Anne Hewitt, Kate D L Umbers, Michelle T Guzik
Nationwide Inclusive Facilitator Training: Mindsets, Practices and Growth
Diane Codding, Alexandria H. Yen, Haley Lewis, Vanessa Johnson-Ojeda, Regina F. Frey, Sarah Chobot Hokanson, Bennett B. Goldberg
Postdoctoral Scholar Recruitment and Hiring Practices in STEM: A Pilot Study
Meagan Heirwegh, Douglas C. Rees, Lindsey Malcom-Piqueux
Posted by felicity davie, on 5 March 2024
Royal Society Publishing has recently published a special issue from Philosophical Transactions B entitled – Causes and consequences of stochastic processes in development and disease.
The issue is organised and edited by Dagan Jenkins, Jonathan Chubb and Gabriel Galea.
The articles can be accessed directly at www.bit.ly/PTB1900
About this issue
Biology is inherently variable. Some differences between individuals are controlled by genetics and predictable: all Chihuahuas are smaller than Great Danes even though they are members of the same species. This issue presents studies of biological variability which is not predictable, emphasising ‘random’ differences between cells or organisms. Sources of random variability are identified using both statistical methods and cellular analyses, in controlled cultures and growing tissues. Some studies show that consequences of variability can be positive, for example by allowing a population to resist external stresses, like plants resisting unpredictable weather. In other situations, it can be detrimental, such as by allowing some cancer cells to resist treatment or resulting in error-prone embryo development which causes malformations at birth.
This issue arises from a Royal Society discussion meeting held in April 2023.
Purchase the print issue at the reduced price of £40 by contacting sales@royalsociety.org

Posted by Qingqing Wang, on 29 February 2024
In a new study, Joshua Gendron and colleagues find that plants can measure two different photoperiods to independently control seasonal flowering and growth, and the vegetative growth is partially dependent on the production of myo-inositol, a precursor required in many processes that control growth (1). First author Qingqing Wang takes us through the story behind the paper.
After earning my PhD in fish reproductive physiology from a laboratory in China, I ventured to the US and secured a postdoctoral position in the Gendron lab, initiating my journey into the field of botany. While transitioning from zoology to botany presented significant hurdles, my background in biology, which encompassed genes, molecules, and proteins, eased the process of assimilating knowledge into the botanical sphere. Fortunately, the steadfast support of my lab colleagues, along with Josh Gendron’s encouragement, fostered within me the belief that creativity and problem-solving acumen are indispensable in scientific research, igniting a fervent passion for botanical pursuits.
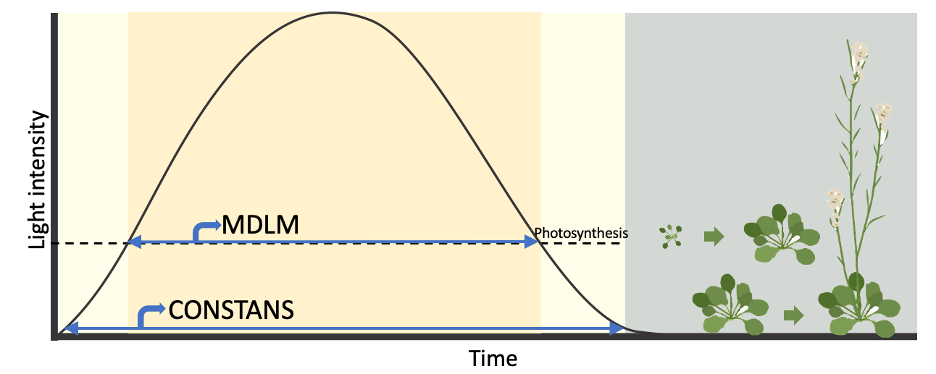
In our laboratory, we have elucidated a metabolic daylength measurement (MDLM) system that is crucial for regulating plant growth under short winter photoperiod conditions. Notably, the post-dusk induction of one gene, PHLOEM PROTEIN 2-A13 (PP2-A13), in short days is pivotal for sustaining plant growth in winter but not summer photoperiod (2). This led me to speculate about the existence of a contrasting pathway that is regulated by MDLM system controlling plant summer growth. My exploration of photoperiod-related literature primarily uncovered research focused on the CONSTANS- FLOWERING LOCUS T (CO-FT) module’s role in regulating flowering, leaving much unknown about other pathways controlling plants’ summer growth.
MIPS1 as a model to study photoperiod-controlled growth.
Delving into the RNA-seq database curated by Gendron lab (3), I meticulously examined gene expression profiles from RNA-seq, seeking candidates exhibiting heightened expression under summer long day conditions. Amidst the constraints of Covid-induced isolation, devoid of laboratory benchwork, I seized the opportunity to immerse myself in extensive literature reviews pertaining to each intriguing gene. I came across a gene called MYOINOSITOL-1-PHOSPHATE SYNTHASE 1 (MIPS1), which plays a pivotal role in myo-inositol biosynthesis. Myo-inositol is an essential sugar that governs various cellular processes crucial for growth regulation. Notably, MIPS1 displayed elevated expression under summer long-day photoperiods, and we discovered that its mutant counterpart, mips1, exhibited specific growth defects under summer long-day conditions but not winter short-day conditions, contrasting with the pp2-a13 mutant. We observed that MIPS1 has a long-day-specific growth effect, prompting us to investigate whether growth is photoperiod-controlled and how MIPS1 responds. To address this, I cultivated both wild type and mips1 mutant plants under critical photoperiod conditions and monitored their growth. As the daytime lengthened, wild type growth was minimal in photoperiods less than 12 hours light to 12 hours dark (12L:12D). However, in photoperiods of 12L:12D or longer, wild type growth accelerated significantly, while the mips1 mutant exhibited clear growth defects. Upon seeing these results, I was excited to share the findings with everyone. The two years of effort dedicated to growing plants under various conditions in the same growth chamber proved worthwhile. These results indicate that growth is indeed controlled by photoperiod and requires MIPS1.
Those findings prompted us to investigate the potential involvement of the CO-FT mechanism in regulating MIPS1 expression. Interestingly, genetic data indicated that MIPS1 expression is not controlled by the CO-FTpathway, which led us to wonder a pivotal question: What regulatory network governs MIPS1 expression, thereby determining plant summer growth rate?
Parallel control by MDLM for MIPS1 required for long day rapid growth, and CO-FT for photoperiod flowering.
We employed a starch-less pgm mutant with a dysfunctional MDLM system to examine the regulatory effect of MDLM on MIPS1 expression. As expected, the photoperiodic expression of MIPS1 was absent in the pgm mutant, confirming our hypothesis that MDLM system regulates MIPS1 expression. Then I think about how to design an experiment to decouple photoperiodic growth and photoperiodic flowering. Exposing the plant to low light intensity, nearing the compensation point where photosynthesis equals respiration, could exhaust starch and sucrose reserves and therefore impeding MDLM but not CO-FT system. I grew plants in low light intensity condition and found that the mips1 mutant phenotype disappeared, while flowering time remained consistent with long-day conditions, which is making us exciting. This sustained our model that MDLM regulates plant growth in parallel to CO-FT regulated flowering. To further test this model, I propose a classic experiment akin to the “night break” experiments used to test photoperiodic flowering. In “night break” flowering experiments, long day plant flowers in long day but not short day photoperiod. However, introducing short pulses of light during the night in short-day conditions promotes flowering. Similarly, to assess whether photoperiod regulates rapid growth, we propose dividing the daytime in long-day conditions into two segments: one exposed to high light intensity and the other to low intensity. We found that the mips1 mutant phenotype would manifest in the segment under intense light, while the wild-type size would exceed that of the segment under light exposure during the initial portion. Moreover, flowering timing in both scenarios would mirror that of long-day conditions. This suggests that plants can discern an absolute photoperiod using low light-sensing photoreceptor mechanisms to regulate flowering time. Concurrently, they can gauge the photosynthetic duration as a metabolic day length to govern growth.
In the end…
This journey has taught me that in scientific research, one must be passionate, diligent, possess problem-solving skills, embrace teamwork, and be willing to challenge and follow classic principles. This model has filled and expanded our understanding of plant regulation by photoperiod, which extends beyond just flowering time; there are many other processes for photoperiodic regulation. Moreover, by utilizing different durations and intensities of light exposure, we can achieve separate regulation of flowering and growth in crops, leading to conservation of energy resources for crops in the future.
References
Posted by Toby Andrews, on 29 February 2024
The Young Embryologist Network conference 2024 (YEN24) is the 16th iteration of the network’s renowned yearly Developmental Biology meeting. This hybrid conference will take place at the Francis Crick Institute on Tuesday 28thMay 2024, and will be streamed worldwide over Zoom. The YEN conference is a unique opportunity for early career researchers in Developmental Biology to share their research, network, and interact with peers and pioneers in the field.

This year, we will bring together a global audience. We are actively recruiting local representatives from universities and institutes worldwide to establish remote hubs for participants to gather, watch talks and engage in the conversation. We are also awarding travel grants, generously supported by the Company of Biologists.
YEN24 features a diverse line-up of invited speakers, covering a wide spectrum of topics in Developmental Biology. We will hear from Aydan Bulut Karslioglu (MPI-MG) on environmental regulation of embryonic gene expression and cell fate specification, and Kristian Franze (University of Cambridge, FAU Erlangen-Nürnberg) on the mechanical control of vertebrate neural development. This year, we are delighted to host Pavel Tomancak (MPI-CBG, CEITEC) as keynote speaker, whose lab uses an interdisciplinary toolkit and comparative approach to understand the evolution of tissue morphogenesis.
We are also excited to host three speakers engaging with the social, legal and ethical implications of biological research in ‘Science in Society’ perspectives talks. Naomi Moris (The Francis Crick Institute) uses synthetic embryos to understands fundamental principles of human development, and recently contributed to a re-evaluation of the term ‘embryo’, defining a roadmap for the use of in vitro embryo models in research. Laurence Lwoff (Council of Europe) is head of the Human Rights and Biomedicine Division at the Council of Europe, and works on an intergovernmental steering committee advising on the protection of human rights in biomedical research. Steve Crabtree (BBC Science Unit) is an award-winning executive producer at BBC Studios, and former series editor for the flagship science series Horizon, where he commissioned and produced over 70 episodes.
We are also inviting abstracts from early career researchers to highlight a cross-section of research in the field in short talks and posters.
Registration for the YEN Conference 2024 is now open! Use our google form to sign up and send in your abstract: tinyurl.com/YENMeeting24.
We hope to see you there!
Head to our website, and follow us on social media for the latest news and announcements
Website: www.youngembryologists.org
Twitter: @YEN_community
Instagram: @youngembryologistsnetwork
Posted by the Node, on 29 February 2024
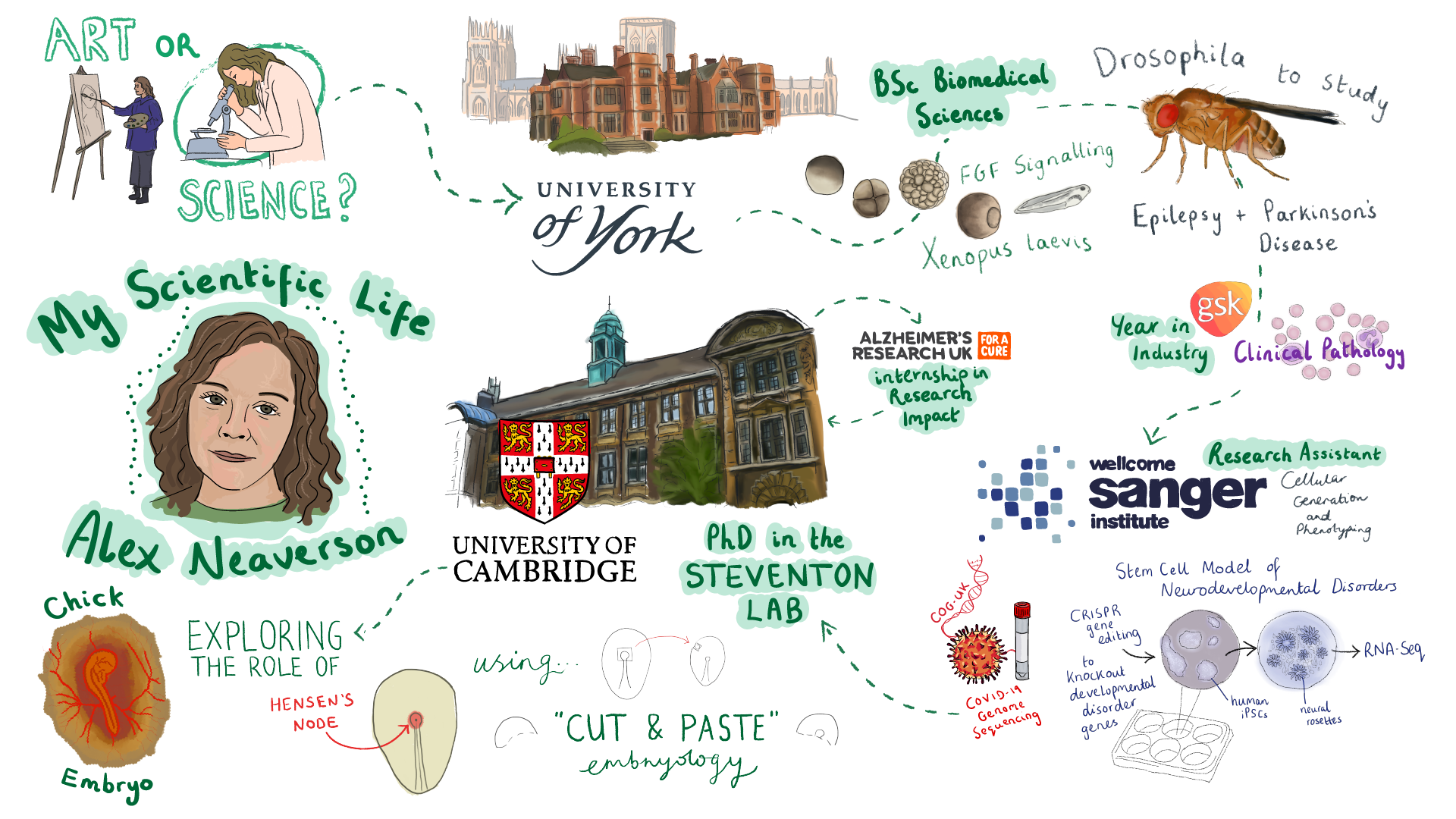
Alex Neaverson is a third-year PhD student at the University of Cambridge, studying regeneration of the Hensen’s Node in chick embryos. Alex is a keen artist and has recently got into scientific illustration while doing an internship at a research charity. As a Node correspondent, she plans to use her artistic skills to create illustrated career timelines of developmental biologists and draw graphical summaries to highlight research conducted by different teams around the world.

Congratulations on being selected as one of our new correspondents! What made you decide to apply to become a Node correspondent?
I’ve known about the Node since I started my PhD in 2021. The Node’s a great resource for bringing developmental biologists together. I really like how there’s a mixture of scientific articles and opinion pieces, and I like the ‘Lab meeting’ posts where you get to know all the different labs. I especially like the SciArt profiles. I love seeing my two big passions — biology and art — mixed together, sometimes in really unusual ways. I’ve always kept art and science separate in my life, where science has been my job and art has been my hobby. But I recently started getting into scientific illustration. I thought considering the Node publishes the SciArt profiles, you might be open to something slightly different from a Correspondent. Maybe you’d like to have someone who can create illustrations as a means of communicating science, instead of purely writing text-based articles. I deliberated over it for a while, but I spoke to my PI and he thought it was a great idea, so I took a chance and clearly it paid off!
How did you get into scientific illustration?
Scientific illustration is something that is very new to me. Art has always been a hobby. I love painting people and animal portraits. I have only started doing scientific illustration during my internship with Alzheimer’s Research UK (ARUK) last year. As part of my PhD course, I had to do a 3-month internship. There was an internship fair and while most organisations had very specific projects in mind, Jorge from ARUK’s Research team was very open to ideas and said that my project could depend on my interests.
My internship ended up being about translating research on dementia prevention into infographics that could be understood by a layperson. I wanted to do this in a more creative way. ARUK was very encouraging, and I was able to get a tablet to do digital illustration and attend online courses for Adobe Illustrator. The resource that I contributed to for ARUK is called the Impact Hub, which is used by the Science communication team and other teams in the charity to communicate the impact of the research funded by the charity. The infographics I created have to clearly communicate the science in a way that can be understood by anybody. That internship was where I started to meld science and art together.
Examples of illustrations Alex did during her internship at Alzheimer’s Research UK (click to enlarge image)


Apart from scientific illustration, have you done much science communication and public outreach?
Before I started my PhD, I worked as a research assistant at the Wellcome Sanger Institute. During the COVID lockdown, I had significantly less lab work to do. I started writing a blog together with a colleague, which was quite fun to do. I also completely revamped my department’s website because it was totally out of date and very boring to look at. I’d like to think I improved it! I’ve also taken part in several outreach activities for children, such as the Big Bang Fair at Birmingham NEC, and the Cambridge Festival last year.
What is your scientific background and what is your current research focus?
I did my undergrad in biomedical sciences in York. I was initially interested in human biology and disease. But it started to change over time as I did my degree. The programme was very open, so I was able to pick a lot of different modules. It was around second or third year when I started to show an interest in developmental biology, which probably had a lot to do with my lecturers. They were very passionate about their subject, and they transferred that passion to me.
In my final year, I did a project with Xenopus embryos, which I really enjoyed. When I finished at York, I thought the one experience that I lacked was with cell culture. I started looking for jobs where I could gain those skills, and ended up joining a core facility at the Sanger Institute where I primarily did stem cell differentiation projects. I learned how to work with iPSCs and differentiate them into neural lineages. But I started to miss working model organisms and I started to realise that I wanted to pursue my own research, which I didn’t have much freedom to do as a research assistant. That was when I decided to take on a PhD at Ben Stevenson’s lab in Cambridge.
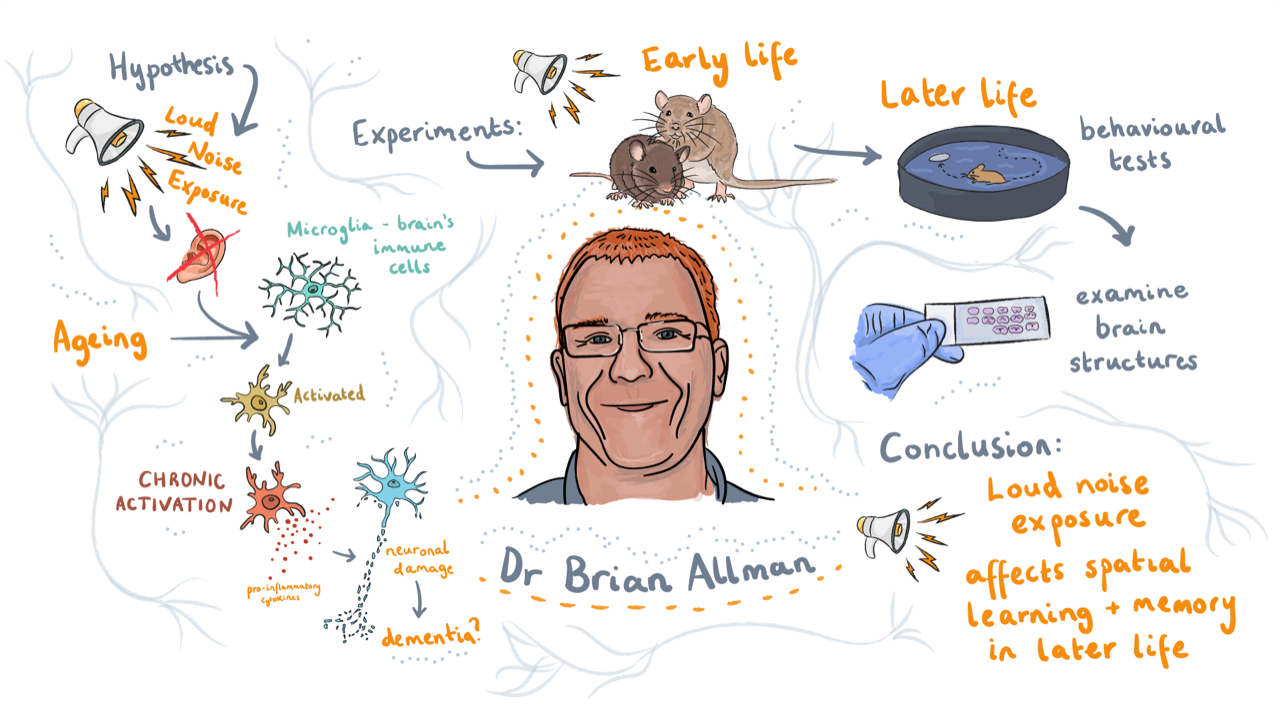
Now, I work with very early chick embryos, and I’m interested in the role of Hensen’s Node, the so-called organiser in the chick embryo, during neural development. The node is thought to be responsible for releasing signals that pattern the neural territory. If you cut the node out of the embryo it will completely regenerate itself and the embryo carries on developing. I’m studying this regenerative process and what the triggers for tissue re-specification might be and whether this has knock on effect on the development of the embryo.
You mentioned you plan to create illustrated content for the Node. Can you elaborate on your ideas?
I’d like to use my illustration skills to create new kinds of content. For example, I’d like to interview developmental biologists about their career paths and create illustrated timelines of their scientific life. I think this will be really fun to create and I think people will be interested to learn about how other people in the field got to where they are today.
I’m also thinking about doing graphical research summaries about an individual or a lab’s research, similar to a paper’s graphical abstract. I’m not sure who I’m going to interview yet, but I’ll probably start with my own lab and people that I know and then branch out.
Will you also write for the Node as well?
I’d like to gain skills in writing as well. One of the things I want to improve is how to adapt the way I write for different audiences, which I think is a really important skill as a scientist. I’d like to make content that blends both text writing and illustration.
Apart from gaining a bit more writing experience, what else do you hope to gain from being a correspondent?
I’d like to meet other people who have similar interests. I’m thinking about a potential career path into science communication after I finish my PhD. I’d like to get to know people and network and find out more about careers in this area.
Finally, what do you like to do in your spare time?
At the end of my undergraduate degree, I loved cooking and baking so much that I convinced myself that I didn’t want to do science anymore and that I actually wanted to be a recipe developer. I felt very conflicted about this because I just spent four years of my life working towards a career in science. I ended up going to my first job in science anyway at the Sanger Institute. I’m glad I did because it did revive my interest in science.
I still do a lot of baking in my spare time and my lab is more than happy to eat all of my creations. Some of my favourite bakes have been embryo themed. I’ve made some cupcakes for a friend’s viva with the stages of zebrafish development. Another friend is a fellow chick embryologist. When she finished her viva, I made some early chick culture cupcakes that were really realistic. So realistic that maybe they were slightly off putting to some people!



Posted by Margarida Cardoso Moreira, on 28 February 2024
Dear colleagues,
We are delighted to bring you a 2-day workshop at the Francis Crick Institute that will gather leading experts in the field to break new ground in cell-type evo-devo. This workshop is free and open to all, but you do need to register (link).
Cells are the fundamental building blocks of living systems. Understanding the differentiation of cell types as well as the origin of novel cell types remains a central problem in developmental and evolutionary biology. Today, advances in molecular techniques have enabled the molecular profiling of individual cells, providing fresh opportunities for new insights into cell type development and evolution. This meeting will be of interest to anyone working with single-cell data.
Speakers:
Pawel Burkhardt (University of Bergen): “The deep evolutionary origins of neurons and nervous systems”
Margarida Cardoso-Moreira (Francis Crick Institute): “Origins of cells and organs – the view from the placenta”
Douglas Erwin (Smithsonian Institute): “Alternative models for formation of cells”
Jacob Musser (Yale University): TBA
Joe Parker (Caltech): “The cellular substrate of evolutionary novelty”
Mihaela Pavlicev (University of Vienna): “Cell types as characters”
Arnau Sebé-Pedrós (CRG – Barcelona): “Early animal cell type diversity, evolution, and regulation”
Stefan Semrau (University of Leiden): “How many cell types are there?”
Francesca Spagnoli (Kings College London): “Mapping the emergence of lineage identities in time & space”
Uli Technau (University of Vienna): TBA
Gunter Wagner (Yale University): “How to distinguish cell types from mere similarity clusters? “
There will also be two panel discussions, and there is plenty of time allocated for discussion and mixing within the schedule.
More information can be found here
All the best,
James DiFrisco and Margarida Cardoso Moreira
Posted by Debora Bogani, on 27 February 2024
Approximately 1 in 20 babies are born with severe anatomical malformations. Each year this equates to 8 million affected newborns, of which 300,000 die within the first four weeks of life. With advances in sequencing technology, the identification of possible disease-causing changes in the genetic code of these patients has accelerated. However, it is a major challenge to prove which of these genetic changes, also called variants, cause these malformations, as well as to establish the cellular mechanisms by which these changes disrupt normal development. How do we distinguish problematic inherited or spontaneous variants in DNA from the many benign changes, and prove that they disrupt normal development? Can we better understand why some patients are more affected than others even though they carry similar, if not the same, genetic changes? How do important environmental influences, such as maternal health during pregnancy, modify how these genetic changes exhibit themselves in terms of severity and spectrum of patient presentations? Many genes that are implicated in congenital anomalies play multiple roles in different tissues during prenatal and postnatal development; thus, these genes are difficult to study in humans, even in stem cell ‘disease-in-a-dish’ models.


Working closely with clinicians and researchers who submit variants of uncertain significance (VUS) via our portal, the Congenital Anomalies Cluster will create precisely engineered mouse models of patient variants, which will:
Our cluster has broad developmental biology expertise and together with the MRC National Mouse Genetics Network (NMGN) we have built a pipeline to investigate the complex interactions that are disrupted during early life, across multiple organ systems. Through the detection of overlapping phenotypes in F0 embryo screening, we hope to provide clinicians with sufficient evidence to confirm a genetic diagnosis in their families. The generation of clinically relevant mouse lines to study pathogenic mechanisms allows exploration of the consequences of genetic mutations during the critical postnatal period (automated live monitoring), and during disease progression later in life. These models will serve as improved platforms for developing much-needed therapeutic interventions.
Part of the remit of the Congenital Anomalies Cluster is to help clinicians and researchers establish genetic diagnoses by modelling VUS in mice. Our submissions portal is open to clinicians and researchers worldwide.
Submitted VUS will be assessed by our Clinical Advisory Board who will meet at least twice a year.
They will consider and rank the submitted VUS based on the following Congenital Anomalies Cluster priorities (in order of importance): 1) New disease gene. 2) Known disease gene, but new phenotype association or novel allelic disorder. 3) Known disease gene with difficult-to-interpret VUS; e.g., nearby significant variants such as structural variants, or deep intronic single nucleotide variants. 4) Known disease genes where broader investigation of the mouse model could lead to new insights into pathogenic mechanism and/or therapy development.
Considerations:
Contributing teams are ideally clinically led or have strong clinical engagement, to efficiently return diagnostic information and enable further assessment of patient phenotypes. The clinical features of your patient should be present at birth and should overlap with the specialities of our developmental biology team: craniofacial, skeletal, heart, neural tube, kidney, and ciliopathies. More weight will be given to syndromic conditions to enable the simultaneous study of multiple systems in the mouse.
Please follow this link to the submission portal and feel free to download and share the flyer above.
For further information please email Karen Liu karen.liu@kcl.ac.uk or Stephen Twigg stephen.twigg@imm.ox.ac.uk
Posted by the Node, on 26 February 2024

In the last of the series of speakers from Development’s 2023 Pathway to Independence programme, we hear from two researchers studying the role of RNA biology in development.
Eric Cornes (ARNA Lab, Inserm/University of Bordeaux)
‘Germline gene regulation by non-coding small RNAs’
Maya Spichal (University of Massachusetts Chan Medical School)
‘Non-canonical germline RNA expression from repetitive DNA’
At the discretion of the speakers, the webinar will be recorded for viewing on demand. To see the other webinars scheduled in our series, and to catch up on previous talks, please visit: thenode.biologists.com/devpres
Posted by Joyce Yu, on 22 February 2024
A press release related to the Research Article in Development “Developmental regulation of cellular metabolism is required for intestinal elongation and rotation“.
How does our intestine, which can be at least 15 feet long, fit properly inside our bodies? As our digestive system grows, the gut tube goes through a series of dramatic looping and rotation to package the lengthening intestine. Failure of the gut to rotate properly during development results in a prevalent, but poorly understood, birth anomaly called intestinal malrotation. Now, in a study published in the journal Development, scientists from North Carolina State University have uncovered a potential cause of this life-threatening condition.
Intestinal malrotation affects 1 in 500 births but the underlying causes are not well understood. To find out why gut revolution could go amiss, scientists need to first understand intestinal rotation during normal development, a complex process that still baffles biologists.
The team of scientists, led by Dr Nanette Nascone-Yoder, decided to make use of a well-established system in frogs. “As vertebrates, frogs and humans share a common ancestor and have many similar anatomical features, including an intestine that rotates in a counter-clockwise direction,” explained Dr Nascone-Yoder. “Because frog embryos develop in only a few days and are highly experimentally accessible, they allow us to quickly test new hypotheses about how and why development goes awry during malrotation.”
“Frog embryos develop in a petri dish and are transparent when the intestine is developing, so they can be exposed to drugs or environmental chemicals to screen for substances capable of producing malrotation,” said Dr Nascone-Yoder. One of the compounds the team screened was the herbicide atrazine. They found that exposure to atrazine greatly increased the frequency at which frog intestines rotated in the reverse (clockwise) direction and decided to focus on atrazine to further investigate intestinal malrotation.
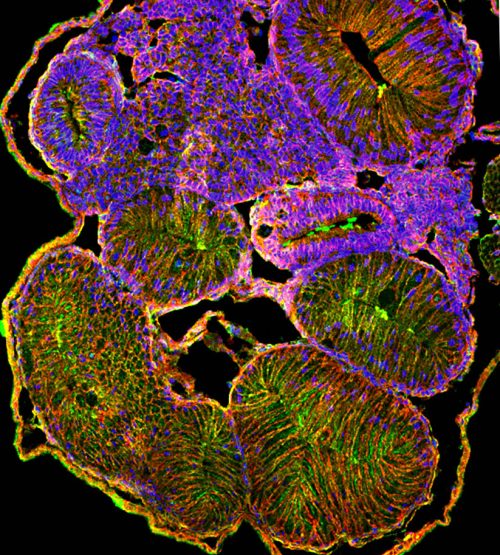
Dr Julia Grzymkowski, who led the experimental work of this study, found that exposure to atrazine disrupted metabolism (chemical reactions that provide energy for biological processes) in the frog embryos. Metabolic imbalance in the embryos derailed a series of cellular processes in the gut — cells could not grow, divide and rearrange appropriately to drive the proper intestinal elongation and rotation.
“Although we found that atrazine causes malrotation in frogs, these results do not necessarily mean that this herbicide causes malrotation in humans, because, in our screen, the tadpoles were exposed to 1000-fold higher levels than are typically found in the environment,” Dr Nascone-Yoder emphasised, “but our findings do strongly suggest that disturbing the same cellular metabolic processes affected by atrazine, for example, via exposure to other chemicals in the environment and/or genetic variations that affect metabolism, could contribute to intestinal malrotation in humans.”
This study is just beginning to unravel what happens during embryonic development that leads to intestinal malrotation. Dr Nascone-Yoder’s team hopes to extend this work: “Our results have provided new avenues to explore the underlying causes of this prevalent birth anomaly. We are now starting to dive deeper into the cellular events that coordinate the complicated process of intestinal elongation and rotation.”
———-
To find out more about the story behind the paper, check out this interview we did with first author Julia Grzymkowski and corresponding author Nanette Nascone-Yoder.
———-
Reference: Grzymkowski, J.K., Chiu, M.Y., Jima, D.D., Wyatt, B.H., Jayachandran, S.M., Stutts, W.L., Nascone-Yoder, N.M. (2024) Developmental regulation of cellular metabolism is required for intestinal elongation and rotation. Development, 151, dev202020. doi:10.1242/dev.202020