In Development this week (Vol. 141, Issue 14)
Posted by Seema Grewal, on 8 July 2014
Here are the highlights from the current issue of Development:
Prime time for pluripotency
 Embryonic stem cell (ESC) cultures display a marked heterogeneity in the expression of Nanog, one of several core pluripotency factors required for proper development in vivo. In addition, Nanog levels have also been shown to fluctuate in individual ESCs in vitro; however, the extent and functional consequences of these fluctuations in different pluripotency states has not been fully established. Now, on p. 2770, Domingos Henrique and colleagues take a closer look at Nanog expression in mouse (m) ESCs grown in both 2i/LIF and serum/LIF conditions, which promote naïve versus primed pluripotency states, respectively. Using confocal time-lapse imaging of individual Nanog-reporting mESCs, the authors show that the amplitude of Nanog fluctuation in individual mESCs is similar regardless of culture conditions. The authors also show that divergent Nanog levels exist even between sister cells, and that Nanog levels do not correlate in any way to the cell cycle. In both conditions, cells that expressed low levels of Nanog showed decreased clonogenic capacity and increased lineage priming, whereas the opposite was true when high levels of Nanog were observed. The authors conclude that fluctuating Nanog expression is a cell-intrinsic property that allows ESCs to explore available lineage options in the pluripotent space.
Embryonic stem cell (ESC) cultures display a marked heterogeneity in the expression of Nanog, one of several core pluripotency factors required for proper development in vivo. In addition, Nanog levels have also been shown to fluctuate in individual ESCs in vitro; however, the extent and functional consequences of these fluctuations in different pluripotency states has not been fully established. Now, on p. 2770, Domingos Henrique and colleagues take a closer look at Nanog expression in mouse (m) ESCs grown in both 2i/LIF and serum/LIF conditions, which promote naïve versus primed pluripotency states, respectively. Using confocal time-lapse imaging of individual Nanog-reporting mESCs, the authors show that the amplitude of Nanog fluctuation in individual mESCs is similar regardless of culture conditions. The authors also show that divergent Nanog levels exist even between sister cells, and that Nanog levels do not correlate in any way to the cell cycle. In both conditions, cells that expressed low levels of Nanog showed decreased clonogenic capacity and increased lineage priming, whereas the opposite was true when high levels of Nanog were observed. The authors conclude that fluctuating Nanog expression is a cell-intrinsic property that allows ESCs to explore available lineage options in the pluripotent space.How to make excitatory versus inhibitory neurons
 The balance between excitatory versus inhibitory neuron specification during development is crucial for sensory information processing in later life. The basic helix-loop-helix (bHLH) transcription factors Ascl1 and Ptf1a are crucial for establishing this specificity in the dorsal spinal cord, but how these two factors, which recognise a similiar DNA motif, can have opposite downstream effects is unclear. Now, on p. 2803, Jane Johnson and colleagues use chromatin immuno-precipitation sequencing (ChIP-Seq) and RNA-sequencing (RNA-Seq) to identify the precise target genes and DNA-binding motifs for both proteins in vivousing tissue from the developing mouse neural tube. The authors show that the downstream targets of Ascl1 and Ptf1a include many known homeodomain neuronal specification factors, but that the targets differ widely between the two proteins. Despite this distinction, Ascl1 and Ptf1a bind the same E-box motif; however, the authors show that Ptf1a also binds an additional motif where Ascl1 is not detected. This may explain, at least in part, some of the specificity achieved by Ptf1a. The authors also show that non-E-box motifs are enriched in Ascl1- and Ptf1a-bound regions, suggesting that cooperation from other transcription factors may further enhance this specificity.
The balance between excitatory versus inhibitory neuron specification during development is crucial for sensory information processing in later life. The basic helix-loop-helix (bHLH) transcription factors Ascl1 and Ptf1a are crucial for establishing this specificity in the dorsal spinal cord, but how these two factors, which recognise a similiar DNA motif, can have opposite downstream effects is unclear. Now, on p. 2803, Jane Johnson and colleagues use chromatin immuno-precipitation sequencing (ChIP-Seq) and RNA-sequencing (RNA-Seq) to identify the precise target genes and DNA-binding motifs for both proteins in vivousing tissue from the developing mouse neural tube. The authors show that the downstream targets of Ascl1 and Ptf1a include many known homeodomain neuronal specification factors, but that the targets differ widely between the two proteins. Despite this distinction, Ascl1 and Ptf1a bind the same E-box motif; however, the authors show that Ptf1a also binds an additional motif where Ascl1 is not detected. This may explain, at least in part, some of the specificity achieved by Ptf1a. The authors also show that non-E-box motifs are enriched in Ascl1- and Ptf1a-bound regions, suggesting that cooperation from other transcription factors may further enhance this specificity.
Polarisation mediates lineage specification
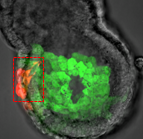
 During blastocyst development, asymmetric cell divisions generate polar and apolar daughter cells, which organise into outer and inner positions, respectively, to form the trophectoderm (TE) and inner cell mass (ICM) lineages. The Hippo signaling pathway is crucial for setting up this early lineage specification, but how Hippo signaling relates to cell position and polarity remains unclear. In this issue (p. 2813), Yojiro Yamanaka and colleagues carefully analyze the initiation process of the first lineage specifications in the 16-cell mouse embryo. The authors count the number of polar/apolar cells and outer/inner cells in intact embryos and find that many apolar cells are located in outer positions with only one or two cells fully internalized. Notably, many of these outer apolar cells have high cytoplasmic phosphorylated YAP, an early marker for inner cells. Further analysis of isolated 8-cell blastomeres confirms that polar and apolar cells have intrinsic differences in the regulation of phosphorylated YAP prior to setting up the outer/inner configurations in the embryo. The authors suggest that polarity regulates the outer/inner cell positioning, as well as Hippo signaling, in order to activate TE and ICM lineage specification.
During blastocyst development, asymmetric cell divisions generate polar and apolar daughter cells, which organise into outer and inner positions, respectively, to form the trophectoderm (TE) and inner cell mass (ICM) lineages. The Hippo signaling pathway is crucial for setting up this early lineage specification, but how Hippo signaling relates to cell position and polarity remains unclear. In this issue (p. 2813), Yojiro Yamanaka and colleagues carefully analyze the initiation process of the first lineage specifications in the 16-cell mouse embryo. The authors count the number of polar/apolar cells and outer/inner cells in intact embryos and find that many apolar cells are located in outer positions with only one or two cells fully internalized. Notably, many of these outer apolar cells have high cytoplasmic phosphorylated YAP, an early marker for inner cells. Further analysis of isolated 8-cell blastomeres confirms that polar and apolar cells have intrinsic differences in the regulation of phosphorylated YAP prior to setting up the outer/inner configurations in the embryo. The authors suggest that polarity regulates the outer/inner cell positioning, as well as Hippo signaling, in order to activate TE and ICM lineage specification.Scribble gets done for intracellular trafficking
 Cell polarity is fundamental for biological activity across many varied cell types within different animal species. Intracellular trafficking regulates the differential distribution of proteins that is fundamental to establishing cell polarity, but how cell polarity regulators exert their effects on trafficking machinery is largely unknown. Now, on p. 2796, David Bilder and colleagues identify a specific and unexpected role for Scribble, a conserved core polarity protein, in controlling cargo sorting during intracellular trafficking in several Drosophila epithelial tissues. The authors show that Scribble mutants phenocopy endocytic internalization mutants but that they themselves are not defective in endolysosomal trafficking. Instead, Scribble controls cargo sorting within the retromer pathway, a system used to recycle proteins from endosomes to the trans-Golgi network. Depletion of the Scribble module affects the localisation of canonical retromer-dependent cargo, such as Wntless and Crumbs, but does not affect cargos that are retromer independent. This work brings a new aspect to the interplay between membrane traffic and polarity, and will help to decipher how these two essential pathways interact to establish and maintain epithelial polarity.
Cell polarity is fundamental for biological activity across many varied cell types within different animal species. Intracellular trafficking regulates the differential distribution of proteins that is fundamental to establishing cell polarity, but how cell polarity regulators exert their effects on trafficking machinery is largely unknown. Now, on p. 2796, David Bilder and colleagues identify a specific and unexpected role for Scribble, a conserved core polarity protein, in controlling cargo sorting during intracellular trafficking in several Drosophila epithelial tissues. The authors show that Scribble mutants phenocopy endocytic internalization mutants but that they themselves are not defective in endolysosomal trafficking. Instead, Scribble controls cargo sorting within the retromer pathway, a system used to recycle proteins from endosomes to the trans-Golgi network. Depletion of the Scribble module affects the localisation of canonical retromer-dependent cargo, such as Wntless and Crumbs, but does not affect cargos that are retromer independent. This work brings a new aspect to the interplay between membrane traffic and polarity, and will help to decipher how these two essential pathways interact to establish and maintain epithelial polarity.
PLUS…
Regeneration, morphogenesis and self-organization
 In March 2014, the RIKEN Center for Developmental Biology in Kobe, Japan, hosted a meeting entitled ‘Regeneration of Organs: Programming and Self-Organization’. Scientists from across the globe met to discuss current research on regeneration, organ morphogenesis and self-organization – and the links between these fields. As discussed by Daniel Goldman, a diverse range of experimental models and organ systems was presented at the meeting, and the speakers aptly illustrated the unique power of each. See the Meeting Review on p 2745.
In March 2014, the RIKEN Center for Developmental Biology in Kobe, Japan, hosted a meeting entitled ‘Regeneration of Organs: Programming and Self-Organization’. Scientists from across the globe met to discuss current research on regeneration, organ morphogenesis and self-organization – and the links between these fields. As discussed by Daniel Goldman, a diverse range of experimental models and organ systems was presented at the meeting, and the speakers aptly illustrated the unique power of each. See the Meeting Review on p 2745.
Cellular and physical mechanisms of branching morphogenesis
 Branching morphogenesis is the developmental program that builds the epithelial trees of various organs, including the airways of the lung, the collecting ducts of the kidney, and the ducts of the mammary and salivary glands. Recent advances in cell lineage analysis and real-time imaging have uncovered surprising differences in the mechanisms that build these diverse tissues. Victor Varner and Celeste Nelson review these studies and discuss the cellular and physical mechanisms that can contribute to branching morphogenesis. See the Review on p. 2750.
Branching morphogenesis is the developmental program that builds the epithelial trees of various organs, including the airways of the lung, the collecting ducts of the kidney, and the ducts of the mammary and salivary glands. Recent advances in cell lineage analysis and real-time imaging have uncovered surprising differences in the mechanisms that build these diverse tissues. Victor Varner and Celeste Nelson review these studies and discuss the cellular and physical mechanisms that can contribute to branching morphogenesis. See the Review on p. 2750.
Harnessing developmental processes for vascular engineering and regeneration
 The formation of the vasculature is essential for tissue maintenance and regeneration, and understanding how vascular formation is coordinated in vivo can offer valuable insights into engineering approaches for therapeutic vascularization and angiogenesis. Here, Kyung Min Park and Sharon Gerecht discuss how the process of vascular development can be used to guide approaches to engineering vasculature. See the Review on p. 2760.
The formation of the vasculature is essential for tissue maintenance and regeneration, and understanding how vascular formation is coordinated in vivo can offer valuable insights into engineering approaches for therapeutic vascularization and angiogenesis. Here, Kyung Min Park and Sharon Gerecht discuss how the process of vascular development can be used to guide approaches to engineering vasculature. See the Review on p. 2760.


 (No Ratings Yet)
(No Ratings Yet) (3 votes)
(3 votes)











 This post is part of a series on a day in the life of developmental biology labs working on different model organisms. You can read the introduction to the series
This post is part of a series on a day in the life of developmental biology labs working on different model organisms. You can read the introduction to the series